graduate+school+of+medical+science+and+engineering
-
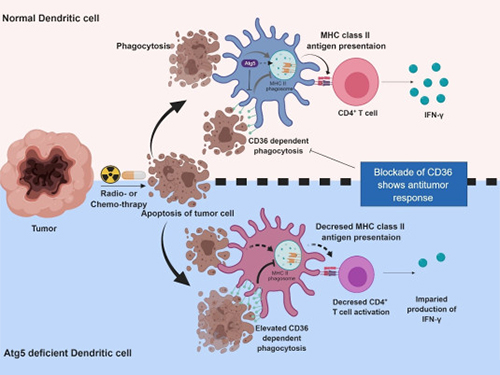 Autophagy in Dendritic Cells Helps Anticancer Activity
Autophagy contributes to the homeostasis of a cell and recently another function of autophagy has been reported. A KAIST research team found that the autophagy of dendritic cells supports T-cell anticancer activity.
Autophagy is a process of maintaining cell homeostasis by removing cellular waste and damaged cellular organelles; nevertheless, its role in the presentation of phagocytized tumor-associated antigens remains vague.
Meanwhile, dendritic cells are the ones that recognize pathogens or cancer antigens, and induce immune responses in T cells. When cancer cells are killed by radiation or an anticancer drug, dendritic cells absorb and remove them and present antigens on their surface to transfer them to T-cells.
Professor Heung Kyu Lee from the Graduate School of Medical Science and Engineering and his team found that the autophagy of dendritic cells plays a key role in T-cell activation and they proposed the principles of enhancing anti-cancer effects.
Their experiments showed that T-cell activation of dendritic cells as well as anticancer immune response dropped when there is a deficiency of Atg5 (autophagy-related) in dendritic cells.
Interestingly, Atg5-deficient dendritic cells significantly elevated receptor CD36 on the surface of the cells, which increased the phagocytosis of apoptotic tumor cells yet restricted the activation of T-cells.
At this time, when introducing antibodies into the system in order to block the receptor CD36, the anti-tumor T-cell response increased substantially while tumor growth declined.
Professor Lee said, “This study allowed us to explore the role of autophagy in the anti-cancer immune response of T-cells. We look forward to developing targeted anti-cancer therapies using the receptor CD36.”
This research was published in Autophagy (10.1080/15548627.2019.1596493) on March 22, 2019.
Figure 1.Mechanism of autophagy in dendritic cells
Figure 2. A role of autophagy in dendritic cells
2019.05.13 View 49959
Autophagy in Dendritic Cells Helps Anticancer Activity
Autophagy contributes to the homeostasis of a cell and recently another function of autophagy has been reported. A KAIST research team found that the autophagy of dendritic cells supports T-cell anticancer activity.
Autophagy is a process of maintaining cell homeostasis by removing cellular waste and damaged cellular organelles; nevertheless, its role in the presentation of phagocytized tumor-associated antigens remains vague.
Meanwhile, dendritic cells are the ones that recognize pathogens or cancer antigens, and induce immune responses in T cells. When cancer cells are killed by radiation or an anticancer drug, dendritic cells absorb and remove them and present antigens on their surface to transfer them to T-cells.
Professor Heung Kyu Lee from the Graduate School of Medical Science and Engineering and his team found that the autophagy of dendritic cells plays a key role in T-cell activation and they proposed the principles of enhancing anti-cancer effects.
Their experiments showed that T-cell activation of dendritic cells as well as anticancer immune response dropped when there is a deficiency of Atg5 (autophagy-related) in dendritic cells.
Interestingly, Atg5-deficient dendritic cells significantly elevated receptor CD36 on the surface of the cells, which increased the phagocytosis of apoptotic tumor cells yet restricted the activation of T-cells.
At this time, when introducing antibodies into the system in order to block the receptor CD36, the anti-tumor T-cell response increased substantially while tumor growth declined.
Professor Lee said, “This study allowed us to explore the role of autophagy in the anti-cancer immune response of T-cells. We look forward to developing targeted anti-cancer therapies using the receptor CD36.”
This research was published in Autophagy (10.1080/15548627.2019.1596493) on March 22, 2019.
Figure 1.Mechanism of autophagy in dendritic cells
Figure 2. A role of autophagy in dendritic cells
2019.05.13 View 49959 -
 KAIST Identifies the Cause of Sepsis-induced Lung Injury
(Professor Pilhan Kim from the Graduate School of Medical Science and Engineering)
A KAIST research team succeeded in visualizing pulmonary microcirculation and circulating cells in vivo with a custom-built 3D intravital lung microscopic imaging system. They found a type of leukocyte called neutrophils aggregate inside the capillaries during sepsis-induced acute lung injury (ALI), leading to disturbances and dead space in blood microcirculation.
According to the researchers, this phenomenon is responsible for tissue hypoxia causing lung damage in the sepsis model, and mitigating neutrophils improves microcirculation as well as hypoxia.
The lungs are responsible for exchanging oxygen with carbon dioxide gases during the breathing process, providing an essential function for sustaining life. This gas exchange occurs in the alveoli, each surrounded by many capillaries containing the circulating red blood cells.
Researchers have been making efforts to observe microcirculation in alveoli, but it has been technically challenging to capture high-resolution images of capillaries and red blood cells inside the lungs that are in constant breathing motion.
Professor Pilhan Kim from the Graduate School of Medical Science and Engineering and his team developed an ultra-fast laser scanning confocal microscope and an imaging chamber that could minimize the movement of a lung while preserving its respiratory state. They used this technology to successfully capture red blood cell circulation inside the capillaries of animal models with sepsis.
During the process, they found that hypoxia was induced by the increase of dead space inside the lungs of a sepsis model, a space where red blood cells do not circulate. This phenomenon is due to the neutrophils aggregating and trapping inside the capillaries and the arterioles. It was also shown that trapped neutrophils damage the lung tissue in the sepsis model by inhibiting microcirculation as well as releasing reactive oxygen species.
Further studies showed that the aggregated neutrophils inside pulmonary vessels exhibit a higher expression of the Mac-1 receptor (CD11b/CD18), which is a receptor involved in intercellular adhesion, compared to the neutrophils that normally circulate. Additionally, they confirmed that Mac-1 inhibitors can improve inhibited microcirculation, ameliorate hypoxia, while reducing pulmonary edema in the sepsis model.
Their high-resolution 3D intravital microscope technology allows the real-time imaging of living cells inside the lungs. This work is expected to be used in research on various lung diseases, including sepsis.
The research team’s pulmonary circulation imaging and precise analytical techniques will be used as the base technology for developing new diagnostic technologies, evaluating new therapeutic agents for various diseases related to microcirculation.
Professor Kim said, “In the ALI model, the inhibition of pulmonary microcirculation occurs due to neutrophils. By controlling this effect and improving microcirculation, it is possible to eliminate hypoxia and pulmonary edema – a new, effective strategy for treating patients with sepsis.”
Their 3D intravital microscope technology was commercialized through IVIM Technology, Inc., which is a faculty startup at KAIST. They released an all-in-one intravital microscope model called ‘IVM-CM’ and ‘IVM-C’. This next-generation imaging equipment for basic biomedical research on the complex pathophysiology of various human diseases will play a crucial role in the future global bio-health market.
This research, led by Dr. Inwon Park from the Department of Emergency Medicine at Seoul National University Bundang Hospital and formally the Graduate School of Medical Science and Engineering at KAIST, was published in the European Respiratory Journal (2019, 53:1800736) on March 28, 2019.
Figure 1. Custom-built high-speed real-time intravital microscope platform
Figure 2. Illustrative schematic and photo of a 3D intravital lung microscopic imaging system
Figure 3. Aggregation of neutrophils and consequent flow disturbance in pulmonary arteriole in sepsis-induced lung injury
2019.05.07 View 43497
KAIST Identifies the Cause of Sepsis-induced Lung Injury
(Professor Pilhan Kim from the Graduate School of Medical Science and Engineering)
A KAIST research team succeeded in visualizing pulmonary microcirculation and circulating cells in vivo with a custom-built 3D intravital lung microscopic imaging system. They found a type of leukocyte called neutrophils aggregate inside the capillaries during sepsis-induced acute lung injury (ALI), leading to disturbances and dead space in blood microcirculation.
According to the researchers, this phenomenon is responsible for tissue hypoxia causing lung damage in the sepsis model, and mitigating neutrophils improves microcirculation as well as hypoxia.
The lungs are responsible for exchanging oxygen with carbon dioxide gases during the breathing process, providing an essential function for sustaining life. This gas exchange occurs in the alveoli, each surrounded by many capillaries containing the circulating red blood cells.
Researchers have been making efforts to observe microcirculation in alveoli, but it has been technically challenging to capture high-resolution images of capillaries and red blood cells inside the lungs that are in constant breathing motion.
Professor Pilhan Kim from the Graduate School of Medical Science and Engineering and his team developed an ultra-fast laser scanning confocal microscope and an imaging chamber that could minimize the movement of a lung while preserving its respiratory state. They used this technology to successfully capture red blood cell circulation inside the capillaries of animal models with sepsis.
During the process, they found that hypoxia was induced by the increase of dead space inside the lungs of a sepsis model, a space where red blood cells do not circulate. This phenomenon is due to the neutrophils aggregating and trapping inside the capillaries and the arterioles. It was also shown that trapped neutrophils damage the lung tissue in the sepsis model by inhibiting microcirculation as well as releasing reactive oxygen species.
Further studies showed that the aggregated neutrophils inside pulmonary vessels exhibit a higher expression of the Mac-1 receptor (CD11b/CD18), which is a receptor involved in intercellular adhesion, compared to the neutrophils that normally circulate. Additionally, they confirmed that Mac-1 inhibitors can improve inhibited microcirculation, ameliorate hypoxia, while reducing pulmonary edema in the sepsis model.
Their high-resolution 3D intravital microscope technology allows the real-time imaging of living cells inside the lungs. This work is expected to be used in research on various lung diseases, including sepsis.
The research team’s pulmonary circulation imaging and precise analytical techniques will be used as the base technology for developing new diagnostic technologies, evaluating new therapeutic agents for various diseases related to microcirculation.
Professor Kim said, “In the ALI model, the inhibition of pulmonary microcirculation occurs due to neutrophils. By controlling this effect and improving microcirculation, it is possible to eliminate hypoxia and pulmonary edema – a new, effective strategy for treating patients with sepsis.”
Their 3D intravital microscope technology was commercialized through IVIM Technology, Inc., which is a faculty startup at KAIST. They released an all-in-one intravital microscope model called ‘IVM-CM’ and ‘IVM-C’. This next-generation imaging equipment for basic biomedical research on the complex pathophysiology of various human diseases will play a crucial role in the future global bio-health market.
This research, led by Dr. Inwon Park from the Department of Emergency Medicine at Seoul National University Bundang Hospital and formally the Graduate School of Medical Science and Engineering at KAIST, was published in the European Respiratory Journal (2019, 53:1800736) on March 28, 2019.
Figure 1. Custom-built high-speed real-time intravital microscope platform
Figure 2. Illustrative schematic and photo of a 3D intravital lung microscopic imaging system
Figure 3. Aggregation of neutrophils and consequent flow disturbance in pulmonary arteriole in sepsis-induced lung injury
2019.05.07 View 43497 -
 Optimal Immuno-Therapeutic Strategies for Liver Cancer
KAIST medical scientists have presented a heterogeneity of immune cell exhaustion in the cancer environment, providing evidence and rationale for designing optimal strategies for immune checkpoint inhibitors in liver cancer patients.
They succeeded in distinguishing the hepatocellular carcinoma group from the exhausted tumor infiltrating immune cell composition of liver cancer patients. The study, conducted in collaboration with Asan Medical Center, confirmed the applicability for liver cancer patients, providing a new path for personalized precision medicine as well as a new model for translational research.
Our immune system is able to destroy cancerous cells in our body, however sometimes cancer cells can adapt and mutate, effectively hiding from our immune system. One of the mechanisms that has evolved to prevent eradication by the immune system is to functionally silence effector T cells, termed T-cell exhaustion, that is mainly mediated by immune checkpoint molecules such as PD-1, TIM-3, and LAG-3.
Recent breakthroughs and encouraging clinical results with various immune checkpoint inhibitors (ICIs), such as anti-PD-1 monoclonal antibodies (mAbs) and anti-CTLA-4 mAbs, have demonstrated tremendous potential to cure cancers through the immune activation of exhausted T cells. Immune checkpoint inhibitors showed significant clinical benefits for several types of cancers, leading to their wide application in clinical practice.
Anti-PD1 blocking antibodies are one of the most representative agents in this class of drug. However, it has been challenging to precisely understand the biological and clinical significance of T-cell exhaustion in cancer. A KAIST research team led by Professor Su-Hyung Park reported the heterogeneity of T-cell exhaustion in hepatocellular carcinoma (HCC) and its potential clinical implications in Gastroenterology on December 4.
The team revealed that heterogeneous T-cell exhaustion status is determined by the differential PD-1 expression levels in CD8+ T cells in liver cancer patients. The authors found that tumor-infiltrating CD8+ T cells with high PD-1 expression from liver cancer patients are functionally impaired and co-express other immune checkpoint receptors such as TIM-3 and/or LAG3, compared to those with low PD-1 expression.
Moreover, based on these results, the authors suggested that liver cancer patients can be classified into two distinct subgroups. Patients having high PD-1 expression levels in the tumor microenvironment showed more aggressive tumor features and biomarkers predicting a favorable response to anti-PD1 therapy. The research team also demonstrated that only liver cancer patients having high PD-1 expression are susceptible to combined immune checkpoint blockade-based therapies.
Prof. Park said, “The new classification of liver cancer patients identified by this study can be utilized as a biomarker to predict the response of current cancer immunotherapy targeting the PD-1 pathway.” He also said they will continue to conduct research on T-cell exhaustion and activation in various types of cancer, which could lead to a better understanding of T-cell response against cancer, thereby providing evidence for future cancer immunotherapy to achieve the ultimate goal to prolong the survival of cancer patients.
2018.12.18 View 4514
Optimal Immuno-Therapeutic Strategies for Liver Cancer
KAIST medical scientists have presented a heterogeneity of immune cell exhaustion in the cancer environment, providing evidence and rationale for designing optimal strategies for immune checkpoint inhibitors in liver cancer patients.
They succeeded in distinguishing the hepatocellular carcinoma group from the exhausted tumor infiltrating immune cell composition of liver cancer patients. The study, conducted in collaboration with Asan Medical Center, confirmed the applicability for liver cancer patients, providing a new path for personalized precision medicine as well as a new model for translational research.
Our immune system is able to destroy cancerous cells in our body, however sometimes cancer cells can adapt and mutate, effectively hiding from our immune system. One of the mechanisms that has evolved to prevent eradication by the immune system is to functionally silence effector T cells, termed T-cell exhaustion, that is mainly mediated by immune checkpoint molecules such as PD-1, TIM-3, and LAG-3.
Recent breakthroughs and encouraging clinical results with various immune checkpoint inhibitors (ICIs), such as anti-PD-1 monoclonal antibodies (mAbs) and anti-CTLA-4 mAbs, have demonstrated tremendous potential to cure cancers through the immune activation of exhausted T cells. Immune checkpoint inhibitors showed significant clinical benefits for several types of cancers, leading to their wide application in clinical practice.
Anti-PD1 blocking antibodies are one of the most representative agents in this class of drug. However, it has been challenging to precisely understand the biological and clinical significance of T-cell exhaustion in cancer. A KAIST research team led by Professor Su-Hyung Park reported the heterogeneity of T-cell exhaustion in hepatocellular carcinoma (HCC) and its potential clinical implications in Gastroenterology on December 4.
The team revealed that heterogeneous T-cell exhaustion status is determined by the differential PD-1 expression levels in CD8+ T cells in liver cancer patients. The authors found that tumor-infiltrating CD8+ T cells with high PD-1 expression from liver cancer patients are functionally impaired and co-express other immune checkpoint receptors such as TIM-3 and/or LAG3, compared to those with low PD-1 expression.
Moreover, based on these results, the authors suggested that liver cancer patients can be classified into two distinct subgroups. Patients having high PD-1 expression levels in the tumor microenvironment showed more aggressive tumor features and biomarkers predicting a favorable response to anti-PD1 therapy. The research team also demonstrated that only liver cancer patients having high PD-1 expression are susceptible to combined immune checkpoint blockade-based therapies.
Prof. Park said, “The new classification of liver cancer patients identified by this study can be utilized as a biomarker to predict the response of current cancer immunotherapy targeting the PD-1 pathway.” He also said they will continue to conduct research on T-cell exhaustion and activation in various types of cancer, which could lead to a better understanding of T-cell response against cancer, thereby providing evidence for future cancer immunotherapy to achieve the ultimate goal to prolong the survival of cancer patients.
2018.12.18 View 4514 -
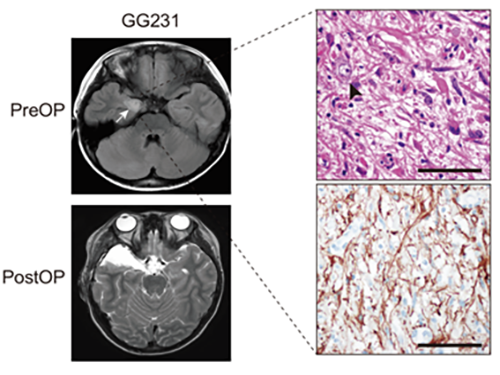 Understanding Epilepsy in Pediatric Tumors; New Therapeutic Target of Intractable Epilepsy Identified
Pediatric brain tumors are characterized by frequent complications due to intractable epilepsy compared to adult brain tumors. However, the genetic cause of refractory epilepsy in pediatric brain tumors has not been elucidated yet, and it is difficult to treat patients because the tumors do not respond to existing antiepileptic drugs and debilitate children’s development.
A research team led by Professor Jeong Ho Lee of the Graduate School of Medical Science and Engineering has recently identified a neuronal BRAF somatic mutation that causes intrinsic epileptogenicity in pediatric brain tumors. Their research results were published online in Nature Medicine on September 17.
The research team studied patients’ tissue diagnosed with ganglioglioma (GG), one of the main causes of tumor-associated intractable epilepsy, and found that the BRAF V600E somatic mutation is involved in the development of neural stem cells by using deep DNA sequencing. This mutation was carried out in an animal model to reproduce the pathology of GG and to observe seizures to establish an animal model for the treatment of epileptic seizures caused by pediatric brain tumors.
Using immunohistochemical and transcriptome analysis, they realized that the BRAF V600E mutation that arose in early progenitor cells during embryonic brain formation led to the acquisition of intrinsic epileptogenic properties in neuronal lineage cells, whereas tumorigenic properties were attributed to a high proliferation of glial lineage cells exhibiting the mutation. Notably, researchers found that seizures in mice were significantly alleviated by intraventricular infusion of the BRAF V600E inhibitor, Vemurafenib, a clinical anticancer drug.
The authors said, “Our study offers the first direct evidence that the BRAF somatic mutation arising from neural stem cells plays a key role in epileptogenesis in the brain tumor. This study also showed a new therapeutic target for tumor-associated epileptic disorders.”
In collaboration with the KAIST startup company, SoVarGen, the research team is currently developing innovative therapeutics for epileptic seizures derived from pediatric brain tumors. This study was supported by the Suh Kyungbae Foundation (SUHF) and the Citizens United for Research in Epilepsy.
(Figure: Preoperative and postoperative brain MRI (left panel), tumor H&E (right upper panel) and GFAP immunohistochemical (right lower panel) staining images from a patient with ganglioglioma (GG231) carrying the BRAFV600E mutation. The white arrow and the black arrowhead indicate the brain tumor and a dysplastic neuron, respectively.)
2018.09.19 View 5197
Understanding Epilepsy in Pediatric Tumors; New Therapeutic Target of Intractable Epilepsy Identified
Pediatric brain tumors are characterized by frequent complications due to intractable epilepsy compared to adult brain tumors. However, the genetic cause of refractory epilepsy in pediatric brain tumors has not been elucidated yet, and it is difficult to treat patients because the tumors do not respond to existing antiepileptic drugs and debilitate children’s development.
A research team led by Professor Jeong Ho Lee of the Graduate School of Medical Science and Engineering has recently identified a neuronal BRAF somatic mutation that causes intrinsic epileptogenicity in pediatric brain tumors. Their research results were published online in Nature Medicine on September 17.
The research team studied patients’ tissue diagnosed with ganglioglioma (GG), one of the main causes of tumor-associated intractable epilepsy, and found that the BRAF V600E somatic mutation is involved in the development of neural stem cells by using deep DNA sequencing. This mutation was carried out in an animal model to reproduce the pathology of GG and to observe seizures to establish an animal model for the treatment of epileptic seizures caused by pediatric brain tumors.
Using immunohistochemical and transcriptome analysis, they realized that the BRAF V600E mutation that arose in early progenitor cells during embryonic brain formation led to the acquisition of intrinsic epileptogenic properties in neuronal lineage cells, whereas tumorigenic properties were attributed to a high proliferation of glial lineage cells exhibiting the mutation. Notably, researchers found that seizures in mice were significantly alleviated by intraventricular infusion of the BRAF V600E inhibitor, Vemurafenib, a clinical anticancer drug.
The authors said, “Our study offers the first direct evidence that the BRAF somatic mutation arising from neural stem cells plays a key role in epileptogenesis in the brain tumor. This study also showed a new therapeutic target for tumor-associated epileptic disorders.”
In collaboration with the KAIST startup company, SoVarGen, the research team is currently developing innovative therapeutics for epileptic seizures derived from pediatric brain tumors. This study was supported by the Suh Kyungbae Foundation (SUHF) and the Citizens United for Research in Epilepsy.
(Figure: Preoperative and postoperative brain MRI (left panel), tumor H&E (right upper panel) and GFAP immunohistochemical (right lower panel) staining images from a patient with ganglioglioma (GG231) carrying the BRAFV600E mutation. The white arrow and the black arrowhead indicate the brain tumor and a dysplastic neuron, respectively.)
2018.09.19 View 5197 -
 Potential Drug to Cure Ciliopathies
(from left: Professor Joon Kim and PhD candidate Yong Joon Kim)
Ciliopathies are rare disorders involving functional and structural abnormalities of cilia. Although they are rare, they may reach 1 in 1,000 births. Unfortunately, there are no small-molecule drugs for treating ciliary defects. A KAIST research team conducted successful research that introduces a potential treatment that will be a foundation for developing drugs to treat the disease as well as a platform for developing small-molecule drugs for similar genetic disorders.
It was found that mutations in genes required for the formation or function of primary cilia cause ciliopathies and they result in cerebellar disorders, kidney dysfunction, and retinal degeneration.
Primary cilia are cell organelles playing a crucial role in the human body. They participate in intercellular signal transduction during embryonic development and allow retinal photoreceptor cells to function.
Currently, there are no approved drugs available for treating most ciliopathies. In fact, this is the case for most of the rare genetic disorders involving functional abnormalities through genetic mutation, and gene therapy is usually the only treatment available.
To tackle this issue, a team led by Professor Joon Kim from the Graduate School of Medical Science and Engineering and Ho Jeong Kwon from Yonsei University constructed a cell that mimics a gene-mutated CEP290, one of the main causes of ciliopathies, through genome editing. They then used cell-based compound library screening to obtain a natural small-molecule compound capable of relieving defects in ciliogenesis, the production of cilia.
The CEP290 protein forms a complex with a ciliopathy protein called NPHP5 to support the function of the ciliary transition zone. In cases where the CEP290 protein is not formed due to a genetic mutation, NPHP5 will not function normally. Here, the compound was confirmed to partially restore the function of the complex by normalizing the function of NPHP5.
The team also identified that the compound is capable of retarding retinal degeneration by injecting the compound into animal models.
As a result, they discovered a lead compound for developing medication to treat ciliopathy patients involving retinal degeneration. Hence, the findings imply that chemical compounds that target other proteins interacting with the disease protein can mitigate shortages of a disease protein in recessive genetic disorders.
PhD candidate Yong Joon Kim stated, “This study shows how genetic disorders caused by genetic mutation can be treated with small-molecule drugs.”
Professor Kim said, “Since the efficacy of the candidate drug has been verified through animal testing, a follow-up study will also be conducted to demonstrate the effect on humans.”
This research was published in the Journal of Clinical Investigation on July 23.
Figure 1. Identification of compounds that rescue ciliogenesis defects caused by CEP290 knockout
Figure 2. Eupatilin injection ameliorates M-opsin trafficking and electrophysiological response of cone photoreceptors in rd16 mice
2018.08.30 View 7388
Potential Drug to Cure Ciliopathies
(from left: Professor Joon Kim and PhD candidate Yong Joon Kim)
Ciliopathies are rare disorders involving functional and structural abnormalities of cilia. Although they are rare, they may reach 1 in 1,000 births. Unfortunately, there are no small-molecule drugs for treating ciliary defects. A KAIST research team conducted successful research that introduces a potential treatment that will be a foundation for developing drugs to treat the disease as well as a platform for developing small-molecule drugs for similar genetic disorders.
It was found that mutations in genes required for the formation or function of primary cilia cause ciliopathies and they result in cerebellar disorders, kidney dysfunction, and retinal degeneration.
Primary cilia are cell organelles playing a crucial role in the human body. They participate in intercellular signal transduction during embryonic development and allow retinal photoreceptor cells to function.
Currently, there are no approved drugs available for treating most ciliopathies. In fact, this is the case for most of the rare genetic disorders involving functional abnormalities through genetic mutation, and gene therapy is usually the only treatment available.
To tackle this issue, a team led by Professor Joon Kim from the Graduate School of Medical Science and Engineering and Ho Jeong Kwon from Yonsei University constructed a cell that mimics a gene-mutated CEP290, one of the main causes of ciliopathies, through genome editing. They then used cell-based compound library screening to obtain a natural small-molecule compound capable of relieving defects in ciliogenesis, the production of cilia.
The CEP290 protein forms a complex with a ciliopathy protein called NPHP5 to support the function of the ciliary transition zone. In cases where the CEP290 protein is not formed due to a genetic mutation, NPHP5 will not function normally. Here, the compound was confirmed to partially restore the function of the complex by normalizing the function of NPHP5.
The team also identified that the compound is capable of retarding retinal degeneration by injecting the compound into animal models.
As a result, they discovered a lead compound for developing medication to treat ciliopathy patients involving retinal degeneration. Hence, the findings imply that chemical compounds that target other proteins interacting with the disease protein can mitigate shortages of a disease protein in recessive genetic disorders.
PhD candidate Yong Joon Kim stated, “This study shows how genetic disorders caused by genetic mutation can be treated with small-molecule drugs.”
Professor Kim said, “Since the efficacy of the candidate drug has been verified through animal testing, a follow-up study will also be conducted to demonstrate the effect on humans.”
This research was published in the Journal of Clinical Investigation on July 23.
Figure 1. Identification of compounds that rescue ciliogenesis defects caused by CEP290 knockout
Figure 2. Eupatilin injection ameliorates M-opsin trafficking and electrophysiological response of cone photoreceptors in rd16 mice
2018.08.30 View 7388 -
 A Breakthrough for Understanding Glioblastoma: Origin Cells for Deadly Brain Tumors Identified
Figure 1. The pattern of GBM genesis is similar to that of firework. The bottom canon represents the first occurrence of the SVZ mutated cell.
A new study by KAIST researchers identified where the mutation causing glioblastoma starts. According to the study, neural stem cells away from the tumor mass are the cells of origin that contain mutation drivers for glioblastoma, one of the most aggressive brain tumor. This breakthrough research, reported in Nature on August 1, gives insights for understanding why glioblastomas almost always grow back, even after surgery, and suggests novel ways to treat glioblastoma, which was previously thought to be incurable.
Like most cancers, glioblastoma is treated with surgery to remove as much of the tumor as possible, then radiation and chemotherapy. However, it almost always returns in less than a year and its median survival time is only 15 months. Precision therapeutic approaches targeting tumors themselves didn’t lead to any breakthroughs.
Professor Jeong Ho Lee’s team at the Graduate School of Medical Science and Engineering described direct genetic evidence through the deep sequencing of all triple-matched samples: normal SVZ tissue away from the tumor mass, tumor tissue, and normal cortical tissue. The research team studied 28 patients with glioblastomas and other types of brain tumors who underwent supra-total resection or other surgical resections of tumors, providing access to normal subventricular zone (SVZ) tissue (where neural stem cells are located) away from the tumor mass. The researchers used various deep and single cell sequencing technologies to conduct comparative DNA analysis on the samples from the patient’s SVZ tissue and tumors.
They reported that normal SVZ tissue away from the tumor in 56.3% of patients with glioblastoma already contained low-level glioblastoma driver mutations that were observed at high levels in their matching tumors. Furthermore, the research team generated a genome edited mouse carrying glioblastoma mutations in the SVZ and showed that neural stem cells with mutations migrate from the SVZ lead to the development of glioblastomas in distant brain regions. (See the image below)
Professor Lee conducted this study in collaboration with Professor Seok-Gu Kang of the Brain Tumor Center at Severance Hospital of Yonsei University. He said, “It’s easier to understand when we compare it to fireworks. Every flare flying around sky can be likened to cancer cells even though the fireworks are triggered on the ground. We found the trigger.” The identification of this mutation pathway of glioblastomas will lead to a new paradigm for therapeutic strategies. He added, “Now, we can focus on interrupting the recurrence and evolution of glioblastomas.”
Professor Lee has investigated mutations arising in the brain for a decade. He is developing innovative diagnostics and therapeutics for untreatable brain disorders including intractable epilepsy and glioblastoma at a tech-startup, SoVarGen. “All technologies we used during the research were transferred to the company. This research gave us very good momentum to reach the next phase of our startup,” he remarked.
Figure 2. Genetic analysis of tumor-free SVZ tissue and matching tumor tissue from GBM patients.
Figure 3. Glioma progression in genome edited mice carrying GBM mutations in the SVZ
2018.08.02 View 11526
A Breakthrough for Understanding Glioblastoma: Origin Cells for Deadly Brain Tumors Identified
Figure 1. The pattern of GBM genesis is similar to that of firework. The bottom canon represents the first occurrence of the SVZ mutated cell.
A new study by KAIST researchers identified where the mutation causing glioblastoma starts. According to the study, neural stem cells away from the tumor mass are the cells of origin that contain mutation drivers for glioblastoma, one of the most aggressive brain tumor. This breakthrough research, reported in Nature on August 1, gives insights for understanding why glioblastomas almost always grow back, even after surgery, and suggests novel ways to treat glioblastoma, which was previously thought to be incurable.
Like most cancers, glioblastoma is treated with surgery to remove as much of the tumor as possible, then radiation and chemotherapy. However, it almost always returns in less than a year and its median survival time is only 15 months. Precision therapeutic approaches targeting tumors themselves didn’t lead to any breakthroughs.
Professor Jeong Ho Lee’s team at the Graduate School of Medical Science and Engineering described direct genetic evidence through the deep sequencing of all triple-matched samples: normal SVZ tissue away from the tumor mass, tumor tissue, and normal cortical tissue. The research team studied 28 patients with glioblastomas and other types of brain tumors who underwent supra-total resection or other surgical resections of tumors, providing access to normal subventricular zone (SVZ) tissue (where neural stem cells are located) away from the tumor mass. The researchers used various deep and single cell sequencing technologies to conduct comparative DNA analysis on the samples from the patient’s SVZ tissue and tumors.
They reported that normal SVZ tissue away from the tumor in 56.3% of patients with glioblastoma already contained low-level glioblastoma driver mutations that were observed at high levels in their matching tumors. Furthermore, the research team generated a genome edited mouse carrying glioblastoma mutations in the SVZ and showed that neural stem cells with mutations migrate from the SVZ lead to the development of glioblastomas in distant brain regions. (See the image below)
Professor Lee conducted this study in collaboration with Professor Seok-Gu Kang of the Brain Tumor Center at Severance Hospital of Yonsei University. He said, “It’s easier to understand when we compare it to fireworks. Every flare flying around sky can be likened to cancer cells even though the fireworks are triggered on the ground. We found the trigger.” The identification of this mutation pathway of glioblastomas will lead to a new paradigm for therapeutic strategies. He added, “Now, we can focus on interrupting the recurrence and evolution of glioblastomas.”
Professor Lee has investigated mutations arising in the brain for a decade. He is developing innovative diagnostics and therapeutics for untreatable brain disorders including intractable epilepsy and glioblastoma at a tech-startup, SoVarGen. “All technologies we used during the research were transferred to the company. This research gave us very good momentum to reach the next phase of our startup,” he remarked.
Figure 2. Genetic analysis of tumor-free SVZ tissue and matching tumor tissue from GBM patients.
Figure 3. Glioma progression in genome edited mice carrying GBM mutations in the SVZ
2018.08.02 View 11526 -
 Mechanism Leading to Cortical Malformation from Brain-Only Mutations Identified
Focal malformations of cortical development (FMCDs) are a heterogeneous group of brain cortical abnormalities. These conditions are the most common causes of medically refractory epilepsy in children and are highly associated with intellectual disability, developmental delay, and autism-spectrum disorders. Despite a broad spectrum of cortical abnormalities in FMCDs, the defective migration of neuronal cells is considered a key pathological hallmark.
A research team led by Professor Jeong Ho Lee in the Graduate School of Medical Science and Engineering at KAIST has recently investigated the molecular mechanism of defective neuronal migration in FMCDs. Their research results were published online in Neuron on June 21, 2018.
The research team previously demonstrated that brain-only mutations in the mechanistic target of rapamycin (MTOR) gene causes focal cortical dysplasia, one major form of FMCDs leading to intractable epilepsy in children. However, the molecular mechanisms by which brain-only mutations in MTOR lead to cortical dyslamination and defective neuronal migration in FMCDs remain unclear.
To study the molecular mechanism of brain cortical dyslamination, the research team utilized patients’ brain tissues and modeled the MTOR mutation-carrying cell and animal models recapitulating the pathogenesis and symptoms of FMCD patients. By performing comprehensive molecular genetic experiments, they found that the formation of primary cilia, one of cellular organelles, was disrupted in MTOR mutation-carrying neurons and demonstrated that this ciliary disruption was a cause of cortical dyslamination in FMCDs.
MTOR mutations prevented degradation of the OFD1 protein, one of the negative regulators of ciliary formation. As a result, the OFD1 protein was abnormally accumulated in MTOR mutation-carrying neurons, causing focal cortical dyslamination. By suppressing the expression of the OFD1 protein, the research team was able to rescue the defective formation of primary cilia, leading to the restoration of cortical dyslamination and defective neuronal migration considerably.
Based on these results, the research team is carrying out further research to develop novel therapeutics for patients with FMCDs caused by brain-only mutations.
This work was supported by grants from the Suh Kyungbae Foundation and Citizens United for Research in Epilepsy.
The research paper is titled “Brain Somatic Mutations in MTOR Disrupt Neuronal Ciliogenesis, Leading to Focal Cortical Dyslamination.” (Digital Object Identifier #: 10.1016/j.neuron.2018.05.039)
Picture 1: The disrupted formation of primary cilia in brain tissues of FMCD mouse models and patients with FMCDs caused by brain somatic mutations in MTOR.
Picture 2: The rescue of defective ciliary formation in FMCD mouse models leading to the restoration of cortical dyslamination and defective neuronal migration.
2018.07.02 View 7996
Mechanism Leading to Cortical Malformation from Brain-Only Mutations Identified
Focal malformations of cortical development (FMCDs) are a heterogeneous group of brain cortical abnormalities. These conditions are the most common causes of medically refractory epilepsy in children and are highly associated with intellectual disability, developmental delay, and autism-spectrum disorders. Despite a broad spectrum of cortical abnormalities in FMCDs, the defective migration of neuronal cells is considered a key pathological hallmark.
A research team led by Professor Jeong Ho Lee in the Graduate School of Medical Science and Engineering at KAIST has recently investigated the molecular mechanism of defective neuronal migration in FMCDs. Their research results were published online in Neuron on June 21, 2018.
The research team previously demonstrated that brain-only mutations in the mechanistic target of rapamycin (MTOR) gene causes focal cortical dysplasia, one major form of FMCDs leading to intractable epilepsy in children. However, the molecular mechanisms by which brain-only mutations in MTOR lead to cortical dyslamination and defective neuronal migration in FMCDs remain unclear.
To study the molecular mechanism of brain cortical dyslamination, the research team utilized patients’ brain tissues and modeled the MTOR mutation-carrying cell and animal models recapitulating the pathogenesis and symptoms of FMCD patients. By performing comprehensive molecular genetic experiments, they found that the formation of primary cilia, one of cellular organelles, was disrupted in MTOR mutation-carrying neurons and demonstrated that this ciliary disruption was a cause of cortical dyslamination in FMCDs.
MTOR mutations prevented degradation of the OFD1 protein, one of the negative regulators of ciliary formation. As a result, the OFD1 protein was abnormally accumulated in MTOR mutation-carrying neurons, causing focal cortical dyslamination. By suppressing the expression of the OFD1 protein, the research team was able to rescue the defective formation of primary cilia, leading to the restoration of cortical dyslamination and defective neuronal migration considerably.
Based on these results, the research team is carrying out further research to develop novel therapeutics for patients with FMCDs caused by brain-only mutations.
This work was supported by grants from the Suh Kyungbae Foundation and Citizens United for Research in Epilepsy.
The research paper is titled “Brain Somatic Mutations in MTOR Disrupt Neuronal Ciliogenesis, Leading to Focal Cortical Dyslamination.” (Digital Object Identifier #: 10.1016/j.neuron.2018.05.039)
Picture 1: The disrupted formation of primary cilia in brain tissues of FMCD mouse models and patients with FMCDs caused by brain somatic mutations in MTOR.
Picture 2: The rescue of defective ciliary formation in FMCD mouse models leading to the restoration of cortical dyslamination and defective neuronal migration.
2018.07.02 View 7996 -
 Distinguished Professor Koh Donates His Ho-Am Prize Money
(From left: Distinguished Professor Gou Young Koh and KAIST President Sung-Chul Shin)
Distinguished Professor Gou Young Koh from the Graduate School of Medical Science and Engineering donated one hundred million KRW to KAIST that he received for winning the Ho-Am Prize.
Professor Koh, who is widely renowned for angiogenesis, was appointed as the 2018 laureate of the 28th Ho-Am Prize for demonstrating the effective reduction of tumor progression and metastasis via tumor vessel normalization.
He made the donation to the Graduate School of Medical Science and Engineering, where he conducted his research. “As a basic medical scientist, it is my great honor to receive this prize for the recognition of my research outcome. I will give impetus to research for continuous development,” Professor Koh said.
Professor Koh also received the 5th Asan Award in Medicine in 2012 and the 7th Kyung-Ahm Award in 2011. He was also the awardee of the 17th Wunsch Medical Award. He has donated cash prizes to the school every time he is awarded.
KAIST President Sung-Chul Shin said, “I would like to express my gratitude to the professor for his generous donation to the school. It will be a great help fostering outstanding medical scientists.
Professor Koh received his MD-PhD from the Medical School of Chonbuk National University. After finishing his post-doctoral program at Cornell University and Indiana State University, he was appointed as a professor at Chonbuk National University and POSTECH. Currently, he holds the position of distinguished professor at KAIST and director of the IBS Center for Vascular Research.
2018.06.20 View 5082
Distinguished Professor Koh Donates His Ho-Am Prize Money
(From left: Distinguished Professor Gou Young Koh and KAIST President Sung-Chul Shin)
Distinguished Professor Gou Young Koh from the Graduate School of Medical Science and Engineering donated one hundred million KRW to KAIST that he received for winning the Ho-Am Prize.
Professor Koh, who is widely renowned for angiogenesis, was appointed as the 2018 laureate of the 28th Ho-Am Prize for demonstrating the effective reduction of tumor progression and metastasis via tumor vessel normalization.
He made the donation to the Graduate School of Medical Science and Engineering, where he conducted his research. “As a basic medical scientist, it is my great honor to receive this prize for the recognition of my research outcome. I will give impetus to research for continuous development,” Professor Koh said.
Professor Koh also received the 5th Asan Award in Medicine in 2012 and the 7th Kyung-Ahm Award in 2011. He was also the awardee of the 17th Wunsch Medical Award. He has donated cash prizes to the school every time he is awarded.
KAIST President Sung-Chul Shin said, “I would like to express my gratitude to the professor for his generous donation to the school. It will be a great help fostering outstanding medical scientists.
Professor Koh received his MD-PhD from the Medical School of Chonbuk National University. After finishing his post-doctoral program at Cornell University and Indiana State University, he was appointed as a professor at Chonbuk National University and POSTECH. Currently, he holds the position of distinguished professor at KAIST and director of the IBS Center for Vascular Research.
2018.06.20 View 5082 -
 Activation of Bystander Immune Cells during Acute Hepatitis A.
A KAIST research team has identified a process of tissue damage caused by bystander immune cells in acute viral infections. This research will pave the way for research to understand the principles of tissue damage in viral infections and immune diseases, and can point toward a possible therapeutic target for the treatment.
Upon viral infection, viral replication itself destroys human cells, but in some cases, viral replication is not the direct cause of the tissue damage. In particular, the destruction of infected cells is the primary cause of tissue damage during non-cytopathic viral infections such as hepatitis A virus, hepatitis B virus and hepatitis C virus. However, the underlying pathological mechanisms involved in the tissue damage during viral infections have not been fully elucidated.
Specificity is one of the most important characteristics of the immune system. In general, infection from a certain virus specifically activates immune cells targeting the virus, while other immune cells specific to different viruses remain inactive.
An immune cell not specific to an infected virus is called a bystander immune cell. A phenomenon that activates irrelevant immune cells not originally targeting the infecting virus, called the activation of bystander immune cells, is already known to the world; however, its clinical significance has not been investigated thoroughly.
Professor Eui-Cheol Shin and Professor Su-Hyung Park from the Graduate School of Medical Science and Engineering analyzed patients with acute hepatitis A, in collaboration with Chung-Ang University Hospital.
The team found not only immune cells specifically targeting the hepatitis A virus were activated, but also bystander immune cells were activated and involved in the damaging of liver tissues during acute hepatitis A.
According to the research, when a person is infected with hepatitis A virus, hepatitis A virus-infected cells produce IL-15, which induces the activation of bystander immune cells. Activated bystander immune cells exert innate-like cytotoxicity, triggered by activating receptors NKG2D and NKp30 and this can lead to liver injury.
Through describing the cause of excessive tissue damage during acute viral hepatitis, the research outcome is expected to provide critical contributions for the development of potential therapeutic intervention that can minimize tissue damage caused by viral hepatitis and immune disorders.
Professor Shin said, “This is a novel research case that discovered the clinical significance of bystander immune cell activation, which was previously unknown. We will continue to work on establishing treatments which could prevent tissue damage in viral and immune diseases in the future.”
This research was published in Immunity on January 2.
Figure 1. Graphical abstract
2018.03.06 View 6334
Activation of Bystander Immune Cells during Acute Hepatitis A.
A KAIST research team has identified a process of tissue damage caused by bystander immune cells in acute viral infections. This research will pave the way for research to understand the principles of tissue damage in viral infections and immune diseases, and can point toward a possible therapeutic target for the treatment.
Upon viral infection, viral replication itself destroys human cells, but in some cases, viral replication is not the direct cause of the tissue damage. In particular, the destruction of infected cells is the primary cause of tissue damage during non-cytopathic viral infections such as hepatitis A virus, hepatitis B virus and hepatitis C virus. However, the underlying pathological mechanisms involved in the tissue damage during viral infections have not been fully elucidated.
Specificity is one of the most important characteristics of the immune system. In general, infection from a certain virus specifically activates immune cells targeting the virus, while other immune cells specific to different viruses remain inactive.
An immune cell not specific to an infected virus is called a bystander immune cell. A phenomenon that activates irrelevant immune cells not originally targeting the infecting virus, called the activation of bystander immune cells, is already known to the world; however, its clinical significance has not been investigated thoroughly.
Professor Eui-Cheol Shin and Professor Su-Hyung Park from the Graduate School of Medical Science and Engineering analyzed patients with acute hepatitis A, in collaboration with Chung-Ang University Hospital.
The team found not only immune cells specifically targeting the hepatitis A virus were activated, but also bystander immune cells were activated and involved in the damaging of liver tissues during acute hepatitis A.
According to the research, when a person is infected with hepatitis A virus, hepatitis A virus-infected cells produce IL-15, which induces the activation of bystander immune cells. Activated bystander immune cells exert innate-like cytotoxicity, triggered by activating receptors NKG2D and NKp30 and this can lead to liver injury.
Through describing the cause of excessive tissue damage during acute viral hepatitis, the research outcome is expected to provide critical contributions for the development of potential therapeutic intervention that can minimize tissue damage caused by viral hepatitis and immune disorders.
Professor Shin said, “This is a novel research case that discovered the clinical significance of bystander immune cell activation, which was previously unknown. We will continue to work on establishing treatments which could prevent tissue damage in viral and immune diseases in the future.”
This research was published in Immunity on January 2.
Figure 1. Graphical abstract
2018.03.06 View 6334 -
 Cellular Mechanism for Severe Viral Hepatitis Identified
(Professor Shin(left) and Professor Jung)
KAIST medical scientists identified a cellular mechanism causing inflammatory changes in regulatory T cells that can lead to severe viral hepatitis. Research on this mechanism will help further understand the nature of various inflammatory diseases and lead to the development of relevant clinical treatments.
It is known that activated immune cells of patients with viral hepatitis destroy hepatocyte, but its regulatory mechanism has not yet been described.
Regulatory T cells inhibit activation of other immune cells and thus are important for homeostasis of the immune system. However, recent studies contradictorily show that immune inhibitory functions of regulatory T cells weaken in inflammatory conditions and the cells secrete inflammatory cytokines in response. Meanwhile, such a phenomenon was not observed in viral hepatitis including types A, B and C.
The team focused on changes in regulatory T cells in patients with viral hepatitis and discovered that regulatory T cells undergo inflammatory changes to secrete inflammatory cytokines (protein secreted by immune cells) called TNF. They also proved regulatory T cells that secrete TNF contribute to the progression of viral hepatitis.
The team confirmed that regulatory T cells of acute hepatitis A patients have reduced immune-inhibitory functions. Instead, their regulatory T cells secrete TNF. Through this research, the team identified a molecular mechanism for changes in regulatory T cells and identified the transcription factor regulating the process. Furthermore, the team found similar changes to be also present in hepatitis B and C patients.
A KAIST immunology research team led by Professors Eui-Cheol Shin and Min Kyung Jung at the Graduate School of Medical Science & Engineering conducted this translational research with teams from Chungnam National University and Yonsei University to identify the mechanism in humans, instead of using animal models. The research was described in Gastroenterology last December.
Professor Shin said, “This is the first research on regulatory T cells that contributes to hepatocyte damage in viral hepatitis.” He continued, “It is significant for identifying the cells and the molecules that can be used as effective treatment targets for viral hepatitis in the future. This research was funded by the Samsung Science and Technology Foundation.
(Figure1: Treg cells from acute hepatitis A (AHA) patients produce tumor necrosis factor (TNF) andhave reduced suppressive activity. These changes are due to a decrease in FoxP3 transcription factor and an increase in RORγt transcription factor. TNF-producing Treg cells are associated with severe liver injury in AHA patients.)
(Figure 2: A higher proportion of Treg cells from patients with acute hepatitis A, compared with healthy controls, produced TNF upon stimulation with anti-CD3 and anti-CD2. This study reports the presence and the significance of TNF-producing Treg cells for the first time in human patients.)
2018.01.18 View 7756
Cellular Mechanism for Severe Viral Hepatitis Identified
(Professor Shin(left) and Professor Jung)
KAIST medical scientists identified a cellular mechanism causing inflammatory changes in regulatory T cells that can lead to severe viral hepatitis. Research on this mechanism will help further understand the nature of various inflammatory diseases and lead to the development of relevant clinical treatments.
It is known that activated immune cells of patients with viral hepatitis destroy hepatocyte, but its regulatory mechanism has not yet been described.
Regulatory T cells inhibit activation of other immune cells and thus are important for homeostasis of the immune system. However, recent studies contradictorily show that immune inhibitory functions of regulatory T cells weaken in inflammatory conditions and the cells secrete inflammatory cytokines in response. Meanwhile, such a phenomenon was not observed in viral hepatitis including types A, B and C.
The team focused on changes in regulatory T cells in patients with viral hepatitis and discovered that regulatory T cells undergo inflammatory changes to secrete inflammatory cytokines (protein secreted by immune cells) called TNF. They also proved regulatory T cells that secrete TNF contribute to the progression of viral hepatitis.
The team confirmed that regulatory T cells of acute hepatitis A patients have reduced immune-inhibitory functions. Instead, their regulatory T cells secrete TNF. Through this research, the team identified a molecular mechanism for changes in regulatory T cells and identified the transcription factor regulating the process. Furthermore, the team found similar changes to be also present in hepatitis B and C patients.
A KAIST immunology research team led by Professors Eui-Cheol Shin and Min Kyung Jung at the Graduate School of Medical Science & Engineering conducted this translational research with teams from Chungnam National University and Yonsei University to identify the mechanism in humans, instead of using animal models. The research was described in Gastroenterology last December.
Professor Shin said, “This is the first research on regulatory T cells that contributes to hepatocyte damage in viral hepatitis.” He continued, “It is significant for identifying the cells and the molecules that can be used as effective treatment targets for viral hepatitis in the future. This research was funded by the Samsung Science and Technology Foundation.
(Figure1: Treg cells from acute hepatitis A (AHA) patients produce tumor necrosis factor (TNF) andhave reduced suppressive activity. These changes are due to a decrease in FoxP3 transcription factor and an increase in RORγt transcription factor. TNF-producing Treg cells are associated with severe liver injury in AHA patients.)
(Figure 2: A higher proportion of Treg cells from patients with acute hepatitis A, compared with healthy controls, produced TNF upon stimulation with anti-CD3 and anti-CD2. This study reports the presence and the significance of TNF-producing Treg cells for the first time in human patients.)
2018.01.18 View 7756 -
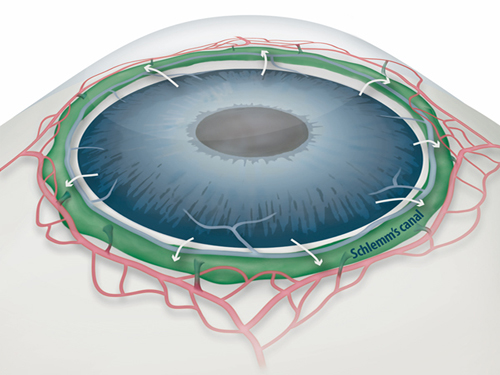 Draining Eyes Clogged with Glaucoma
Professor Gou Young Koh in the Graduate School of Medical Science and Engineering and his team have identified a new mechanism involved in the development and progression of glaucoma, and found a potential therapeutic option to treat it. Glaucoma is the second cause of irreversible blindness, after cataracts. It affects about 3.5% of the world population aged 40 to 80.
Professor Koh also serves as the director of the Center for Vascular Research at the Institute for Basic Science. The IBS said the study, published in the Journal of Clinical Investigation, is expected to help the development of therapies to treat primary open-angle glaucoma (POAG), which counts for three quarters of all glaucoma patients.
One of the most important risk factors for glaucoma is the increased pressure inside the eye. A liquid called aqueous humor is constantly produced and drained out from the eye. It transports nutrients and inflates the eye giving it a roughly spherical shape. However, if this fluid cannot flow out of the eye chambers freely, an increase in intraocular pressure can damage the optic nerve, leading to vision loss. The precise mechanism of elevated resistance to aqueous humor outflow remains unclear, and although the current treatments for glaucoma tackle the production and outflow of aqueous humor, their outcomes are still poor.
A component of the eye that plays a fundamental role in draining out the aqueous humor is Schlemm's canal. It collects the aqueous humor and mediates its transfer from the eye chambers to blood circulation. The cells on the walls of the canal, endothelial cells, ship the liquid from the inner to the outer side in “packages”, called vacuoles. As the shape and number of the vacuoles reflects the outflow performance, several giant vacuoles are expected in the normal outflow process.
The team explained how imbalances in Schlemm's canal significantly increase the risk of glaucoma. They showed that an important regulator for canal functionality is the angiopoietin-Tie2 system. Angiopoietins, such as Ang1 and Ang2, are proteins important for the growth of new blood vessels and Tie2 is the receptor that binds them. It is known that the angiopoietin-Tie2 system plays a role in Schlemm’s canal formation, as Tie2 mutations or angiopoietin absence result in congenital glaucoma. However, this study clarified that it is also critically important during adulthood.
The researchers reported that adult mice deficient in Tie2 suffer from an elevated intraocular pressure, retinal neuronal damage and partial visual impairment. Moreover, they had a markedly decreased number of giant vacuoles inside Schlemm’s canal endothelial cells, which indicate a poor aqueous humor drainage.
The scientists also investigated if and how this process changes in older mice, as aging is a major risk factor for glaucoma, and showed that aged mice experience reduced levels of giant vacuoles, Tie2, Ang1, and Ang2, as well as other proteins connected with the angiopoietin-Tie2 pathway, like Prox1.
To test whether Tie2 activation could shift the situation, the researchers tested the antibody ABTAA (Ang2-binding and Tie2-activating antibody). They injected it in one eye of mice, while the other eye of the same mice functioned as the negative control. After one week, levels of Tie2 and Prox1, number and diameter of giant vacuoles in Schlemm’s canals increased in the ABTAA-treated eyes compared to control eyes. The researchers observed a similar outcome with decreased intraocular pressure when ABTAA was injected to the eyes of mice suffering from POAG with regressed Schlemm’s canals, indicating that this antibody might be considered as a therapeutic option.
"Slow development of glaucoma treatments is partly due to the poor understanding of the underlying pathogenesis," said Professor Koh, the corresponding author of the study. "We hope that identifying the critical role of the angiopoietin-Tie2 system in adult Schlemm’s canals will bring a significant boost in the development of therapeutics."
Figure 1: Schlemm's canal position inside the eye.
Schlemm's canal (green) plays a fundamental role in draining the aqueous humor (white arrows) from the anterior chamber of the eye to blood circulation. If the aqueous humor is not able to flow out freely, elevated intraocular pressure damages the optical nerve causing glaucoma and eventually blindness.
Figure 2: Electron microscope images reveal how the aqueous humor is packaged in vacuoles (arrowheads) inside the cells forming the walls of Schlemm's canal.
Aging and glaucoma cause the number and size of giant vacuoles to decrease, meaning that the aqueous humor outflow is compromised. The images compare the giant vacuoles in Schlemm's canals of a healthy mouse (top) and a mouse lacking Tie2 (bottom)
Figure 3: The Ang2-binding and Tie2-activating antibody (ABTAA) rejuvenates the eye of aged mice and rescues them from glaucoma.
Aging causes a reduction of the protein Tie2, a risk factor for increased intraocular pressure and glaucoma. In this experiment, one eye of mice lacking Ang1 and Ang2 was injected with the premixed ABTAA and Ang2, while the other eye was used as negative control. The researchers observed an increase in the area of Schlemm’s canal, together with higher levels of Tie2 (red) and lower intraocular pressure, suggesting that ABTAA restores the canal's functionality. The image includes the transcription factor Prox1 (green) and CD144 (blue), a protein present at the junctions between cells that form the wall of the canal. The angiopoietin-Tie2 system and Prox1 are linked by a vicious circle: the less Tie2 and Ang2, the less Prox1, leading to Schlemm's canal damage, increase in intraocular pressure, and acceleration of glaucoma progression.
2017.09.19 View 6689
Draining Eyes Clogged with Glaucoma
Professor Gou Young Koh in the Graduate School of Medical Science and Engineering and his team have identified a new mechanism involved in the development and progression of glaucoma, and found a potential therapeutic option to treat it. Glaucoma is the second cause of irreversible blindness, after cataracts. It affects about 3.5% of the world population aged 40 to 80.
Professor Koh also serves as the director of the Center for Vascular Research at the Institute for Basic Science. The IBS said the study, published in the Journal of Clinical Investigation, is expected to help the development of therapies to treat primary open-angle glaucoma (POAG), which counts for three quarters of all glaucoma patients.
One of the most important risk factors for glaucoma is the increased pressure inside the eye. A liquid called aqueous humor is constantly produced and drained out from the eye. It transports nutrients and inflates the eye giving it a roughly spherical shape. However, if this fluid cannot flow out of the eye chambers freely, an increase in intraocular pressure can damage the optic nerve, leading to vision loss. The precise mechanism of elevated resistance to aqueous humor outflow remains unclear, and although the current treatments for glaucoma tackle the production and outflow of aqueous humor, their outcomes are still poor.
A component of the eye that plays a fundamental role in draining out the aqueous humor is Schlemm's canal. It collects the aqueous humor and mediates its transfer from the eye chambers to blood circulation. The cells on the walls of the canal, endothelial cells, ship the liquid from the inner to the outer side in “packages”, called vacuoles. As the shape and number of the vacuoles reflects the outflow performance, several giant vacuoles are expected in the normal outflow process.
The team explained how imbalances in Schlemm's canal significantly increase the risk of glaucoma. They showed that an important regulator for canal functionality is the angiopoietin-Tie2 system. Angiopoietins, such as Ang1 and Ang2, are proteins important for the growth of new blood vessels and Tie2 is the receptor that binds them. It is known that the angiopoietin-Tie2 system plays a role in Schlemm’s canal formation, as Tie2 mutations or angiopoietin absence result in congenital glaucoma. However, this study clarified that it is also critically important during adulthood.
The researchers reported that adult mice deficient in Tie2 suffer from an elevated intraocular pressure, retinal neuronal damage and partial visual impairment. Moreover, they had a markedly decreased number of giant vacuoles inside Schlemm’s canal endothelial cells, which indicate a poor aqueous humor drainage.
The scientists also investigated if and how this process changes in older mice, as aging is a major risk factor for glaucoma, and showed that aged mice experience reduced levels of giant vacuoles, Tie2, Ang1, and Ang2, as well as other proteins connected with the angiopoietin-Tie2 pathway, like Prox1.
To test whether Tie2 activation could shift the situation, the researchers tested the antibody ABTAA (Ang2-binding and Tie2-activating antibody). They injected it in one eye of mice, while the other eye of the same mice functioned as the negative control. After one week, levels of Tie2 and Prox1, number and diameter of giant vacuoles in Schlemm’s canals increased in the ABTAA-treated eyes compared to control eyes. The researchers observed a similar outcome with decreased intraocular pressure when ABTAA was injected to the eyes of mice suffering from POAG with regressed Schlemm’s canals, indicating that this antibody might be considered as a therapeutic option.
"Slow development of glaucoma treatments is partly due to the poor understanding of the underlying pathogenesis," said Professor Koh, the corresponding author of the study. "We hope that identifying the critical role of the angiopoietin-Tie2 system in adult Schlemm’s canals will bring a significant boost in the development of therapeutics."
Figure 1: Schlemm's canal position inside the eye.
Schlemm's canal (green) plays a fundamental role in draining the aqueous humor (white arrows) from the anterior chamber of the eye to blood circulation. If the aqueous humor is not able to flow out freely, elevated intraocular pressure damages the optical nerve causing glaucoma and eventually blindness.
Figure 2: Electron microscope images reveal how the aqueous humor is packaged in vacuoles (arrowheads) inside the cells forming the walls of Schlemm's canal.
Aging and glaucoma cause the number and size of giant vacuoles to decrease, meaning that the aqueous humor outflow is compromised. The images compare the giant vacuoles in Schlemm's canals of a healthy mouse (top) and a mouse lacking Tie2 (bottom)
Figure 3: The Ang2-binding and Tie2-activating antibody (ABTAA) rejuvenates the eye of aged mice and rescues them from glaucoma.
Aging causes a reduction of the protein Tie2, a risk factor for increased intraocular pressure and glaucoma. In this experiment, one eye of mice lacking Ang1 and Ang2 was injected with the premixed ABTAA and Ang2, while the other eye was used as negative control. The researchers observed an increase in the area of Schlemm’s canal, together with higher levels of Tie2 (red) and lower intraocular pressure, suggesting that ABTAA restores the canal's functionality. The image includes the transcription factor Prox1 (green) and CD144 (blue), a protein present at the junctions between cells that form the wall of the canal. The angiopoietin-Tie2 system and Prox1 are linked by a vicious circle: the less Tie2 and Ang2, the less Prox1, leading to Schlemm's canal damage, increase in intraocular pressure, and acceleration of glaucoma progression.
2017.09.19 View 6689 -
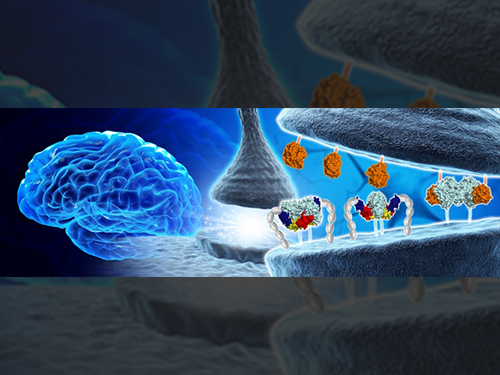 Structural Insights into the Modulation of Synaptic Adhesion by MDGA for Synaptogenesis
Synapses connected by various synaptic adhesion molecules are communication spaces between neurons for transmitting information. Among various synaptic adhesion molecules, neuroligins are arguably the most widely studied class of postsynaptic adhesion molecules, which mainly interact with presynaptic neurexins to induce excitatory or inhibitory synapse development. Recently, the membrane-associated mucin (MAM) domain-containing GPI anchor protein 1 (MDGA1) has been characterized as a key suppressor of Neuroligin-2/Neurexin-1β-mediated inhibitory synapse development, but how it acts remains a mystery.
In a recent issue of Neuron, published on June 21, 2017, a research team led by Professor Ho Min Kim at the Graduate School of Medical Science and Engineering of KAIST reported the three-dimensional structure of MDGA1/Neuroligin-2 complex and mechanistic insights into how MDGAs negatively modulate synapse development governed by Neurexins/Neuroligins trans -synaptic adhesion complex.
MDGA1 consists of six Ig-like domains, fibronectin type III repeat domain, and MAM domain . The crystal structure of MDGA1/Neuroligin-2 complex reveals that they form the 2:2 hetero-tetrameric complex and only the Ig1-Ig2 domains of MDGA1 are involved in interactions with Neuroligin-2. The structural comparison between the MDGA1/Neuroligin-2 and Neurexin-1β/Neuroligin-1 complexes intriguingly indicates that the Neuroligin-2 region binding to MDGA1 largely overlaps with that of Neurexin-1β, but the interaction interface of the MDGA1/Neuroligin-2 complex is much larger than that of the Neurexin-1β/Neuroligin-1 complex. This explains why Neuroligin-2 binds stronger to MDGA1 than Neurexin-1β, and how the favored MDGA1 binding to Neuroligin-2 sterically blocks the interaction between Neuroligin-2 and Neurexin-1β, which is critical for the suppression of inhibitory synapse development.
“Although we found that MDGA Ig domains (Ig 1 and Ig 2) are sufficient to form a complex with NL2, other extracellular domains, including Ig 3–6, FN III, and MAM domains, may also contribute to stable cis-interactions between MDGA1 and Neuroligin-2 by providing conformational flexibility. Therefore, further structural analysis of full-length MDGA will be required,” Professor Kim said.
Neuroligin-2 specifically promotes the development of inhibitory synapses, whereas neuroligin-1 promotes the development of excitatory synapses. Recently, not only MDGA1, but also MDGA2 have emerged as synaptic regulators for the development of excitatory or inhibitory synapses. In vitro biochemical analysis in this research clearly demonstrates that Neuroligin-1 and Neuroligin-2 bind to both MDGA1 and MDGA2 with comparable affinity. However, pull-down assays using detergent-solubilized mouse brain membrane fractions show the specific interaction of MDGA1 with Neuroligin-2, but not with Neuroligin-1. “This suggests that unidentified processes may dictate the selective association of MDGA1 with Neuroligin-2 in vivo , ” explained Professor Jaewon Ko at the Daegu Gyeongbuk Institute of Science and Technology (DGIST).
A balance between excitatory and inhibitory synapses is crucial to healthy cognition and behavior. Mutations in neuroligins, neurexins, and MDGAs, which can disrupt the excitatory/inhibitory balance, are associated with neuropsychiatric diseases such as autism and schizophrenia. Jung A Kim at KAIST, first author in this study, said, “Our discovery from integrative investigations are an important first step both for a better understanding of Neuroligin/Neurexin synaptic adhesion pathways and MDGA-mediated regulation of synapse development as well as the development of potential new therapies for autism, schizophrenia, and epilepsy.”
2017.07.10 View 7946
Structural Insights into the Modulation of Synaptic Adhesion by MDGA for Synaptogenesis
Synapses connected by various synaptic adhesion molecules are communication spaces between neurons for transmitting information. Among various synaptic adhesion molecules, neuroligins are arguably the most widely studied class of postsynaptic adhesion molecules, which mainly interact with presynaptic neurexins to induce excitatory or inhibitory synapse development. Recently, the membrane-associated mucin (MAM) domain-containing GPI anchor protein 1 (MDGA1) has been characterized as a key suppressor of Neuroligin-2/Neurexin-1β-mediated inhibitory synapse development, but how it acts remains a mystery.
In a recent issue of Neuron, published on June 21, 2017, a research team led by Professor Ho Min Kim at the Graduate School of Medical Science and Engineering of KAIST reported the three-dimensional structure of MDGA1/Neuroligin-2 complex and mechanistic insights into how MDGAs negatively modulate synapse development governed by Neurexins/Neuroligins trans -synaptic adhesion complex.
MDGA1 consists of six Ig-like domains, fibronectin type III repeat domain, and MAM domain . The crystal structure of MDGA1/Neuroligin-2 complex reveals that they form the 2:2 hetero-tetrameric complex and only the Ig1-Ig2 domains of MDGA1 are involved in interactions with Neuroligin-2. The structural comparison between the MDGA1/Neuroligin-2 and Neurexin-1β/Neuroligin-1 complexes intriguingly indicates that the Neuroligin-2 region binding to MDGA1 largely overlaps with that of Neurexin-1β, but the interaction interface of the MDGA1/Neuroligin-2 complex is much larger than that of the Neurexin-1β/Neuroligin-1 complex. This explains why Neuroligin-2 binds stronger to MDGA1 than Neurexin-1β, and how the favored MDGA1 binding to Neuroligin-2 sterically blocks the interaction between Neuroligin-2 and Neurexin-1β, which is critical for the suppression of inhibitory synapse development.
“Although we found that MDGA Ig domains (Ig 1 and Ig 2) are sufficient to form a complex with NL2, other extracellular domains, including Ig 3–6, FN III, and MAM domains, may also contribute to stable cis-interactions between MDGA1 and Neuroligin-2 by providing conformational flexibility. Therefore, further structural analysis of full-length MDGA will be required,” Professor Kim said.
Neuroligin-2 specifically promotes the development of inhibitory synapses, whereas neuroligin-1 promotes the development of excitatory synapses. Recently, not only MDGA1, but also MDGA2 have emerged as synaptic regulators for the development of excitatory or inhibitory synapses. In vitro biochemical analysis in this research clearly demonstrates that Neuroligin-1 and Neuroligin-2 bind to both MDGA1 and MDGA2 with comparable affinity. However, pull-down assays using detergent-solubilized mouse brain membrane fractions show the specific interaction of MDGA1 with Neuroligin-2, but not with Neuroligin-1. “This suggests that unidentified processes may dictate the selective association of MDGA1 with Neuroligin-2 in vivo , ” explained Professor Jaewon Ko at the Daegu Gyeongbuk Institute of Science and Technology (DGIST).
A balance between excitatory and inhibitory synapses is crucial to healthy cognition and behavior. Mutations in neuroligins, neurexins, and MDGAs, which can disrupt the excitatory/inhibitory balance, are associated with neuropsychiatric diseases such as autism and schizophrenia. Jung A Kim at KAIST, first author in this study, said, “Our discovery from integrative investigations are an important first step both for a better understanding of Neuroligin/Neurexin synaptic adhesion pathways and MDGA-mediated regulation of synapse development as well as the development of potential new therapies for autism, schizophrenia, and epilepsy.”
2017.07.10 View 7946