ROC
-
 Distinguished Professor Sukbok Chang Donates His Prize Money
The honoree of the 2019 Korea Best Scientist and Technologist Award, Distinguished Professor Sukbok Chang donated his prize money of one hundred million KRW to the Chemistry Department Scholarship Fund and the Lyu Keun-Chul Sports Complex Management Fund during a donation ceremony last week.
Professor Chang won the award last month in recognition of his pioneering achievements and lifetime contributions to the development of carbon-hydrogen activation strategies, especially for carbon-carbon, carbon-nitrogen, and carbon-oxygen formations. Professor Chang, a world renowned chemist, has been recognized for his highly selective catalytic systems, allowing the controlled defunctionalization of bio-derived platform substrates under mild conditions and opening a new avenue for the utilization of biomass-derived platform chemicals.
“All my achievements are the results of my students’ hard work and dedication. I feel very fortunate to have such talented team members. I want to express my sincere gratitude for such a great research environment that we have worked together in so far,” said Professor Chang at the ceremony.
KAIST President Sung-Chul Shin said, “Not only will Professor Chang’s donation make a significant contribution to the Department of Chemistry, but also to the improvement of the Lyu Keun-Chul Sports Complex’s management, which directly links to the health and welfare of the KAIST community.”
Professor Chang currently holds the position of distinguished professor at KAIST and director of the Center for Catalytic Hydrocarbon Functionalizations in the Institute for Basic Science (IBS). He previously received the Kyung-Ahm Academic Award in 2013 and the Korea Toray Science Award in 2018. All these prize money also went to the school.
(END)
2019.08.26 View 9850
Distinguished Professor Sukbok Chang Donates His Prize Money
The honoree of the 2019 Korea Best Scientist and Technologist Award, Distinguished Professor Sukbok Chang donated his prize money of one hundred million KRW to the Chemistry Department Scholarship Fund and the Lyu Keun-Chul Sports Complex Management Fund during a donation ceremony last week.
Professor Chang won the award last month in recognition of his pioneering achievements and lifetime contributions to the development of carbon-hydrogen activation strategies, especially for carbon-carbon, carbon-nitrogen, and carbon-oxygen formations. Professor Chang, a world renowned chemist, has been recognized for his highly selective catalytic systems, allowing the controlled defunctionalization of bio-derived platform substrates under mild conditions and opening a new avenue for the utilization of biomass-derived platform chemicals.
“All my achievements are the results of my students’ hard work and dedication. I feel very fortunate to have such talented team members. I want to express my sincere gratitude for such a great research environment that we have worked together in so far,” said Professor Chang at the ceremony.
KAIST President Sung-Chul Shin said, “Not only will Professor Chang’s donation make a significant contribution to the Department of Chemistry, but also to the improvement of the Lyu Keun-Chul Sports Complex’s management, which directly links to the health and welfare of the KAIST community.”
Professor Chang currently holds the position of distinguished professor at KAIST and director of the Center for Catalytic Hydrocarbon Functionalizations in the Institute for Basic Science (IBS). He previously received the Kyung-Ahm Academic Award in 2013 and the Korea Toray Science Award in 2018. All these prize money also went to the school.
(END)
2019.08.26 View 9850 -
 New Catalyst for Synthesizing Chiral Molecules Selectively
(from left: Dr. Yoonsu Park and Professor Sukbok Chang from the Department of Chemistry)
Molecules in nature often have “twin” molecules that look identical. In particular, the twin molecules that look like mirror images to each other are called enantiomers. However, even though they have the same type and number of elements, these twin molecules exhibit completely different properties.
Professor Sukbok Chang and Dr. Yoonsu Park from the Department of Chemistry developed a new catalyst capable of selectively synthesizing only one of the two enantiomers. Using this catalyst, the have succeeded in manufacturing the chiral lactam, an essential ingredient in pharmaceuticals, from a hydrocarbon compound.
Enantiomerism or chirality is considered very important for drug development. Biomaterials, such as DNAs and proteins also have chiral properties, but they exhibit different physiological activities depending on the types of drugs. One type of the enantiomer could be useful while the other is toxic. Hence, the technology for selective synthesizing (i.e. asymmetric synthesis) is required, but it is still regarded as a great challenge faced by modern chemistry to date.
The researchers solved this problem by developing a new catalyst. Earlier they presented their research on developing an iridium catalyst that converts hydrocarbons into high value γ-lactam compounds, and published it in Science in March 2018. However, the developed catalyst still had a limitation that both types of enantiomers are obtained without selectivity.
In this study, they found that among dozens of other catalyst candidates, iridium catalysts with chiral diamine scaffolds were able to select the correct enantiomer with a selectivity of 99% or more. This novel catalyst can be used to synthesize the various chiral γ-lactam as required. A left-handed γ-lactam and a right-handed γ-lactam can be produced using a left-handed iridium catalyst and a right-handed iridium catalyst, respectively.
They analyzed the reason for the high selectivity through computational chemistry simulations. They identified that temporal hydrogen bonding occurred between the chiral diamine catalysts and the hydrocarbon compound during the reaction. As a result of the hydrogen bonding, the formation of the left-handed lactam was boosted.
With their new catalyst, they also succeeded in synthesizing chiral lactam compounds with different structures. By using inexpensive and readily available feedstock hydrocarbons, the researchers produced a group of chiral lactams in different shapes. As their chirality and diverse structures enable lactams to function as an active compound in the body for antibiotic, anti-inflammatory, or anti-tumoral functions, this study may facilitate the development of potential drugs in a more efficient and cheaper way.
Professor Chang said, “We hope that our research on selectively producing core units of effective drugs will lead to developing new drugs that demonstrate fewer side-effects and higher efficacy. There are also economic advantages of this research because it uses hydrocarbon compounds, which can be abundantly found in nature, to produce high-value raw materials.
This research was published in Nature Catalysis(10.1038/s41929-019-0230-x) on February 19, 2019.
Figure 1. Asymmetric formation of chiral γ-lactam
Figure 2. Outline of research outcome
2019.03.05 View 8211
New Catalyst for Synthesizing Chiral Molecules Selectively
(from left: Dr. Yoonsu Park and Professor Sukbok Chang from the Department of Chemistry)
Molecules in nature often have “twin” molecules that look identical. In particular, the twin molecules that look like mirror images to each other are called enantiomers. However, even though they have the same type and number of elements, these twin molecules exhibit completely different properties.
Professor Sukbok Chang and Dr. Yoonsu Park from the Department of Chemistry developed a new catalyst capable of selectively synthesizing only one of the two enantiomers. Using this catalyst, the have succeeded in manufacturing the chiral lactam, an essential ingredient in pharmaceuticals, from a hydrocarbon compound.
Enantiomerism or chirality is considered very important for drug development. Biomaterials, such as DNAs and proteins also have chiral properties, but they exhibit different physiological activities depending on the types of drugs. One type of the enantiomer could be useful while the other is toxic. Hence, the technology for selective synthesizing (i.e. asymmetric synthesis) is required, but it is still regarded as a great challenge faced by modern chemistry to date.
The researchers solved this problem by developing a new catalyst. Earlier they presented their research on developing an iridium catalyst that converts hydrocarbons into high value γ-lactam compounds, and published it in Science in March 2018. However, the developed catalyst still had a limitation that both types of enantiomers are obtained without selectivity.
In this study, they found that among dozens of other catalyst candidates, iridium catalysts with chiral diamine scaffolds were able to select the correct enantiomer with a selectivity of 99% or more. This novel catalyst can be used to synthesize the various chiral γ-lactam as required. A left-handed γ-lactam and a right-handed γ-lactam can be produced using a left-handed iridium catalyst and a right-handed iridium catalyst, respectively.
They analyzed the reason for the high selectivity through computational chemistry simulations. They identified that temporal hydrogen bonding occurred between the chiral diamine catalysts and the hydrocarbon compound during the reaction. As a result of the hydrogen bonding, the formation of the left-handed lactam was boosted.
With their new catalyst, they also succeeded in synthesizing chiral lactam compounds with different structures. By using inexpensive and readily available feedstock hydrocarbons, the researchers produced a group of chiral lactams in different shapes. As their chirality and diverse structures enable lactams to function as an active compound in the body for antibiotic, anti-inflammatory, or anti-tumoral functions, this study may facilitate the development of potential drugs in a more efficient and cheaper way.
Professor Chang said, “We hope that our research on selectively producing core units of effective drugs will lead to developing new drugs that demonstrate fewer side-effects and higher efficacy. There are also economic advantages of this research because it uses hydrocarbon compounds, which can be abundantly found in nature, to produce high-value raw materials.
This research was published in Nature Catalysis(10.1038/s41929-019-0230-x) on February 19, 2019.
Figure 1. Asymmetric formation of chiral γ-lactam
Figure 2. Outline of research outcome
2019.03.05 View 8211 -
 New LSB with Theoretical Capacity over 90%
(Professor Hee-Tak Kim and Hyunwon Chu)
A KAIST research team has developed a lithium sulfur battery (LSB) that realizes 92% of the theoretical capacity and an areal capacity of 4mAh/cm2.
LSBs are gaining a great deal of attention as an alternative for lithium ion batteries (LIBs) because they have a theoretical energy density up to six to seven times higher than that of LIBs, and can be manufactured in a more cost-effective way.
However, LSBs face the obstacle of having a capacity reaching its theoretical maximum because they are prone to uncontrolled growth of lithium sulfide on the electrodes, which leads to blocking electron transfer.
To address the problem of electrode passivation, researchers introduced additional conductive agent into the electrode; however, it drastically lowered the energy density of LSBs, making it difficult to exceed 70% of the theoretical capacity.
Professor Hee-Tak Kim from the Department of Chemical and Biomolecular Engineering and his team replaced the lithium salt anions used in conventional LSB electrolytes with anions with a high donor number. The team successfully induced the three-dimensional growth of lithium sulfide on electrode surfaces and efficiently delayed the electrode passivation.
Based on this electrolyte design, the research team achieved 92% of the theoretical capacity with their high-capacity sulfur electrode (4mAh/cm2), which is equivalent to that of conventional LIB cathode. Furthermore, they were able to form a stable passivation film on the surface of the lithium anode and exhibited stable operation over 100 cycles.
This technology, which can be flexibly used with various types of sulfur electrodes, can mark a new milestone in the battery industry.
Professor Kim said, “We proposed a new physiochemical principle to overcome the limitations of conventional LSBs. I believe our achievement of obtaining 90% of the LBSs’ theoretical capacity without any capacity loss after 100 cycles will become a new milestone.”
This research, first-authored by Hyunwon Chu and Hyungjun Noh, was published in Nature Communications on January 14, 2019. It was also selected in the editor’s highlight for its outstanding achievements.
Figure 1. Lithium sulfur growth and its deposition mechanism for different sulfide growth behaviors
Figure 2. Capacity and cycle life characteristics of the LSBs
2019.02.11 View 8914
New LSB with Theoretical Capacity over 90%
(Professor Hee-Tak Kim and Hyunwon Chu)
A KAIST research team has developed a lithium sulfur battery (LSB) that realizes 92% of the theoretical capacity and an areal capacity of 4mAh/cm2.
LSBs are gaining a great deal of attention as an alternative for lithium ion batteries (LIBs) because they have a theoretical energy density up to six to seven times higher than that of LIBs, and can be manufactured in a more cost-effective way.
However, LSBs face the obstacle of having a capacity reaching its theoretical maximum because they are prone to uncontrolled growth of lithium sulfide on the electrodes, which leads to blocking electron transfer.
To address the problem of electrode passivation, researchers introduced additional conductive agent into the electrode; however, it drastically lowered the energy density of LSBs, making it difficult to exceed 70% of the theoretical capacity.
Professor Hee-Tak Kim from the Department of Chemical and Biomolecular Engineering and his team replaced the lithium salt anions used in conventional LSB electrolytes with anions with a high donor number. The team successfully induced the three-dimensional growth of lithium sulfide on electrode surfaces and efficiently delayed the electrode passivation.
Based on this electrolyte design, the research team achieved 92% of the theoretical capacity with their high-capacity sulfur electrode (4mAh/cm2), which is equivalent to that of conventional LIB cathode. Furthermore, they were able to form a stable passivation film on the surface of the lithium anode and exhibited stable operation over 100 cycles.
This technology, which can be flexibly used with various types of sulfur electrodes, can mark a new milestone in the battery industry.
Professor Kim said, “We proposed a new physiochemical principle to overcome the limitations of conventional LSBs. I believe our achievement of obtaining 90% of the LBSs’ theoretical capacity without any capacity loss after 100 cycles will become a new milestone.”
This research, first-authored by Hyunwon Chu and Hyungjun Noh, was published in Nature Communications on January 14, 2019. It was also selected in the editor’s highlight for its outstanding achievements.
Figure 1. Lithium sulfur growth and its deposition mechanism for different sulfide growth behaviors
Figure 2. Capacity and cycle life characteristics of the LSBs
2019.02.11 View 8914 -
 Highly Scalable Process to Obtain Stable 2D Nanosheet Dispersion
(Professor Do Hyun Kim and his team)
A KAIST team developed technology that allows the mass production of two-dimensional (2D) nanomaterial dispersion by utilizing the characteristic shearing force of hydraulic power.
The 2D nanosheet dispersion can be directly applied to solution-based processes to manufacture devices for electronics as well as energy storage and conversion. It is expected to be used in these devices with improved performance.
There have been numerous researches on the mass production of various 2D nanomaterial because they show outstanding physical and chemical characteristics when they are truly 2D.
With strong mechanical force or chemical reaction only, each existing exfoliation method has its limitation to make 2D material when the scale of manufacturing increases. They also face the issues of high cost and long process time.
Moreover, 2D nanosheets by the exfoliation have the tendency of agglomeration due to the surface energy. Usually, organic solvent or surfactant is required to obtain high yield and concentration of 2D material by minimizing agglomeration.
After several years of research, Professor Do Hyun Kim in the Department of Chemical and Biomolecular Engineering and his team verified that optimized shearing in their reactor provided the highest efficiency for the exfoliation of nanomaterial. For the increased reactor capacity, they selected a flow and a dispersive agent to develop a high-speed, mass-production process to get 2D nanosheets by physical exfoliation with an aqueous solution.
The team proposed a flow reactor based on Taylor-Couette flow, which has the advantage of high shear rate and mixing efficiency even under large reactor capacity.
In this research, Professor Young-Kyu Han at Dongguk University-Seoul carried out the Ab initio calculation to select the dispersive agent. According to his calculation, an ionic liquid can stabilize and disperse 2D nanomaterial even in a small concentration. This calculation could maximize the exfoliating efficiency.
Professor Bong Gill Choi at Kangwon National University carried out the evaluation of device made of resulting dispersion. The team used a membrane filtration process to make a flexible and highly conductive film of 2D material. The film was then applied to produce an electrode for the supercapacitor device with very high capacity per volume. They also confirmed its stability in their supercapacitor device.
Additionally, they applied dispersive nanomaterials including graphene, molybdenum disulfide (MoS₂), and boron nitride (BN) to inkjet printer ink and realized micrometer-thick nanomaterial patterns on A4 paper. The graphene ink showed no loss of electrical property after printing without additional heat treatment.
Professor Kim said, “This new technology for the high-speed mass production of nanomaterials can easily be applied to various 2D nanomaterials. It will accelerate the production of highly efficient devices for optoelectronics, biosensors, and energy storage/conversion units with low cost.”
This research, led by Dr. Jae-Min Jeong, was published in Advanced Functional Materials on August 12.
Figure 1. The cover page of Advanced Functional Materials
2018.12.19 View 6520
Highly Scalable Process to Obtain Stable 2D Nanosheet Dispersion
(Professor Do Hyun Kim and his team)
A KAIST team developed technology that allows the mass production of two-dimensional (2D) nanomaterial dispersion by utilizing the characteristic shearing force of hydraulic power.
The 2D nanosheet dispersion can be directly applied to solution-based processes to manufacture devices for electronics as well as energy storage and conversion. It is expected to be used in these devices with improved performance.
There have been numerous researches on the mass production of various 2D nanomaterial because they show outstanding physical and chemical characteristics when they are truly 2D.
With strong mechanical force or chemical reaction only, each existing exfoliation method has its limitation to make 2D material when the scale of manufacturing increases. They also face the issues of high cost and long process time.
Moreover, 2D nanosheets by the exfoliation have the tendency of agglomeration due to the surface energy. Usually, organic solvent or surfactant is required to obtain high yield and concentration of 2D material by minimizing agglomeration.
After several years of research, Professor Do Hyun Kim in the Department of Chemical and Biomolecular Engineering and his team verified that optimized shearing in their reactor provided the highest efficiency for the exfoliation of nanomaterial. For the increased reactor capacity, they selected a flow and a dispersive agent to develop a high-speed, mass-production process to get 2D nanosheets by physical exfoliation with an aqueous solution.
The team proposed a flow reactor based on Taylor-Couette flow, which has the advantage of high shear rate and mixing efficiency even under large reactor capacity.
In this research, Professor Young-Kyu Han at Dongguk University-Seoul carried out the Ab initio calculation to select the dispersive agent. According to his calculation, an ionic liquid can stabilize and disperse 2D nanomaterial even in a small concentration. This calculation could maximize the exfoliating efficiency.
Professor Bong Gill Choi at Kangwon National University carried out the evaluation of device made of resulting dispersion. The team used a membrane filtration process to make a flexible and highly conductive film of 2D material. The film was then applied to produce an electrode for the supercapacitor device with very high capacity per volume. They also confirmed its stability in their supercapacitor device.
Additionally, they applied dispersive nanomaterials including graphene, molybdenum disulfide (MoS₂), and boron nitride (BN) to inkjet printer ink and realized micrometer-thick nanomaterial patterns on A4 paper. The graphene ink showed no loss of electrical property after printing without additional heat treatment.
Professor Kim said, “This new technology for the high-speed mass production of nanomaterials can easily be applied to various 2D nanomaterials. It will accelerate the production of highly efficient devices for optoelectronics, biosensors, and energy storage/conversion units with low cost.”
This research, led by Dr. Jae-Min Jeong, was published in Advanced Functional Materials on August 12.
Figure 1. The cover page of Advanced Functional Materials
2018.12.19 View 6520 -
 KAIST Develops VRFB with Longer Durability
(from left: PhD candidate Soohyun Kim, Professor Hee-Tak Kim and PhD candidate Junghoon Choi)
There has been growing demand for large-scale storage for energy produced from renewable energy sources in an efficient and stable way. To meet this demand, a KAIST research team developed a new vanadium redox-flow battery (VRFB) with 15 times greater capacity retention and five times longer durability. This VRFB battery can be an excellent candidate for a large-scale rechargeable battery with no risk of explosion.
The VRFB has received much attention for its high efficiency and reliability with the absence of cross-contamination. However, it has the limitation of having insufficient charge and discharge efficiency and a low capacity retention rate because its perfluorinated membrane is very permeable to any active materials. To minimize energy loss, it needs a membrane that has low vanadium ion permeability and high ion conductivity. Hence, there was an attempt to incorporate a hydrocarbon membrane that has low cost and high ion selectivity but it turned out that the VO₂+ caused chemical degradation, which led to shortening the battery life drastically.
To develop a membrane with pore sizes smaller than the hydrated size of vanadium ions yet larger than that of the protons, a research team co-led by Professor Hee-Tae Jung and Professor Hee-Tak Kim from the Department of Chemical and Biomolecular Engineering implemented a graphene-oxide framework (GOF) membrane by cross-linking graphene oxide nanosheets. They believed that GOF, having strong ion selectivity, would be a good candidate for the membrane component for the VRFB. The interlayer spacing between the GO sheets limited moisture expansion and provided selective ion permeation.
The GOF membrane increased the capacity retention of the VRFB, which showed a 15 times higher rate than that of perfluorinated membranes. Its cycling stability was also enhanced up to five times, compared to conventional hydrocarbon membranes.
These pore-sized-tuned graphene oxide frameworks will allow pore-sized tuning of membranes and will be applicable to electrochemical systems that utilize ions of various sizes, such as rechargeable batteries and sensors.
Professor Kim said, “Developing a membrane that prevents the mixing of positive and negative active materials has been a chronic issue in the field of redox-flow batteries. Through this research, we showed that nanotechnology can prevent this crossover issue and membrane degradation. I believe that this technology can be applied to various rechargeable batteries requiring large-scale storage.”
This research was published in Nano Letters on May 3.
Figure 1. Electrochemical performances of the VRFBs with Nafion 115, SPAES (sulfonated poly), and GOF/SPAES: discharge capacity
Figure 2. Schematic of the selective ion transfer of hydrated vanadium ions and protons in the GOF membrane and the molecular structure of the GOF membrane, showing that the GO nanosheets are cross-linked with EDA (ethylenediamine)
2018.09.20 View 8271
KAIST Develops VRFB with Longer Durability
(from left: PhD candidate Soohyun Kim, Professor Hee-Tak Kim and PhD candidate Junghoon Choi)
There has been growing demand for large-scale storage for energy produced from renewable energy sources in an efficient and stable way. To meet this demand, a KAIST research team developed a new vanadium redox-flow battery (VRFB) with 15 times greater capacity retention and five times longer durability. This VRFB battery can be an excellent candidate for a large-scale rechargeable battery with no risk of explosion.
The VRFB has received much attention for its high efficiency and reliability with the absence of cross-contamination. However, it has the limitation of having insufficient charge and discharge efficiency and a low capacity retention rate because its perfluorinated membrane is very permeable to any active materials. To minimize energy loss, it needs a membrane that has low vanadium ion permeability and high ion conductivity. Hence, there was an attempt to incorporate a hydrocarbon membrane that has low cost and high ion selectivity but it turned out that the VO₂+ caused chemical degradation, which led to shortening the battery life drastically.
To develop a membrane with pore sizes smaller than the hydrated size of vanadium ions yet larger than that of the protons, a research team co-led by Professor Hee-Tae Jung and Professor Hee-Tak Kim from the Department of Chemical and Biomolecular Engineering implemented a graphene-oxide framework (GOF) membrane by cross-linking graphene oxide nanosheets. They believed that GOF, having strong ion selectivity, would be a good candidate for the membrane component for the VRFB. The interlayer spacing between the GO sheets limited moisture expansion and provided selective ion permeation.
The GOF membrane increased the capacity retention of the VRFB, which showed a 15 times higher rate than that of perfluorinated membranes. Its cycling stability was also enhanced up to five times, compared to conventional hydrocarbon membranes.
These pore-sized-tuned graphene oxide frameworks will allow pore-sized tuning of membranes and will be applicable to electrochemical systems that utilize ions of various sizes, such as rechargeable batteries and sensors.
Professor Kim said, “Developing a membrane that prevents the mixing of positive and negative active materials has been a chronic issue in the field of redox-flow batteries. Through this research, we showed that nanotechnology can prevent this crossover issue and membrane degradation. I believe that this technology can be applied to various rechargeable batteries requiring large-scale storage.”
This research was published in Nano Letters on May 3.
Figure 1. Electrochemical performances of the VRFBs with Nafion 115, SPAES (sulfonated poly), and GOF/SPAES: discharge capacity
Figure 2. Schematic of the selective ion transfer of hydrated vanadium ions and protons in the GOF membrane and the molecular structure of the GOF membrane, showing that the GO nanosheets are cross-linked with EDA (ethylenediamine)
2018.09.20 View 8271 -
 Using Donut-shaped Lithium Sulfide for Higher Performing Batteries
(from left: Research Professor Fangmin Ye and Professor Hee-Tak Kim)
A KAIST research team developed a lithium-sulfur battery with a doughnut-shaped active material structure showing a record lifecycle of over 600 cycles. Having higher energy density and lower production cost than a lithium-ion battery (LIB), it can be used in electric vehicles that require a longer battery life.
There has been an intense research conducted for developing lithium-sulfur batteries with high energy density because LIBs only allow for a very short travel distance per charge. However, Li-S batteries are still unable to provide a longer lifecycle due to the poor reversibility of the lithium metal cathode.
To tackle this issue, Professor Hee-Tak Kim from the Department of Chemical and Biomolecular Engineering and his team used lithium sulfide (Li₂S) cathodes and combine them with graphite anodes to enhance energy density and lifecycles for the batteries.
Yet, lithium sulfide is costly and, so far, there has not been an electrode architecture and electrolyte design that enables a longer lifecycle between the graphite anodes and lithium sulfide cathodes.
Hence, the team produced a doughnut-shaped lithium sulfide cathode active material from low-cost lithium sulfide developed from raw materials. They have also developed a lithium sulfide ion battery with a graphite anode and lithium sulfide cathode using a high concentration salt electrolyte.
This doughnut-shaped lithium sulfide showed outstanding charge and discharge reversibility through improving the transfer of lithium ions. Its highly concentrated salt electrolyte formed a stable film on the surface of the graphite electrode, which showed strong durability.
Through this technology, the team achieved 30% higher energy density than that of conventional LIBs and secured a lifecycle of more than 600 cycles. This doughnut-shaped lithium sulfide-based electrode can be manufactured using low-cost raw materials and a single heat treatment process. The electrode can also be applied to existing LIBs.
Professor Kim said, “We have demonstrated that applying low-cost sulfur compounds to LIBs can improve both energy density and the lifecycle simultaneously.”
This research, led by Research Professor Fangmin Ye, was published in Advanced Science on May 7.
Figure 1. Structural characterization of Li₂SO₄/CNT and Li₂S/CNT electrodes and suggested mechanism for the formation of the holey-Li₂S nanoarchitecture
2018.09.19 View 6390
Using Donut-shaped Lithium Sulfide for Higher Performing Batteries
(from left: Research Professor Fangmin Ye and Professor Hee-Tak Kim)
A KAIST research team developed a lithium-sulfur battery with a doughnut-shaped active material structure showing a record lifecycle of over 600 cycles. Having higher energy density and lower production cost than a lithium-ion battery (LIB), it can be used in electric vehicles that require a longer battery life.
There has been an intense research conducted for developing lithium-sulfur batteries with high energy density because LIBs only allow for a very short travel distance per charge. However, Li-S batteries are still unable to provide a longer lifecycle due to the poor reversibility of the lithium metal cathode.
To tackle this issue, Professor Hee-Tak Kim from the Department of Chemical and Biomolecular Engineering and his team used lithium sulfide (Li₂S) cathodes and combine them with graphite anodes to enhance energy density and lifecycles for the batteries.
Yet, lithium sulfide is costly and, so far, there has not been an electrode architecture and electrolyte design that enables a longer lifecycle between the graphite anodes and lithium sulfide cathodes.
Hence, the team produced a doughnut-shaped lithium sulfide cathode active material from low-cost lithium sulfide developed from raw materials. They have also developed a lithium sulfide ion battery with a graphite anode and lithium sulfide cathode using a high concentration salt electrolyte.
This doughnut-shaped lithium sulfide showed outstanding charge and discharge reversibility through improving the transfer of lithium ions. Its highly concentrated salt electrolyte formed a stable film on the surface of the graphite electrode, which showed strong durability.
Through this technology, the team achieved 30% higher energy density than that of conventional LIBs and secured a lifecycle of more than 600 cycles. This doughnut-shaped lithium sulfide-based electrode can be manufactured using low-cost raw materials and a single heat treatment process. The electrode can also be applied to existing LIBs.
Professor Kim said, “We have demonstrated that applying low-cost sulfur compounds to LIBs can improve both energy density and the lifecycle simultaneously.”
This research, led by Research Professor Fangmin Ye, was published in Advanced Science on May 7.
Figure 1. Structural characterization of Li₂SO₄/CNT and Li₂S/CNT electrodes and suggested mechanism for the formation of the holey-Li₂S nanoarchitecture
2018.09.19 View 6390 -
 Mathematical Principle behind AI's 'Black Box'
(from left: Professor Jong Chul Ye, PhD candidates Yoseob Han and Eunju Cha)
A KAIST research team identified the geometrical structure of artificial intelligence (AI) and discovered the mathematical principles of highly performing artificial neural networks, which can be applicable in fields such as medical imaging.
Deep neural networks are an exemplary method of implementing deep learning, which is at the core of the AI technology, and have shown explosive growth in recent years. This technique has been used in various fields, such as image and speech recognition as well as image processing.
Despite its excellent performance and usefulness, the exact working principles of deep neural networks has not been well understood, and they often suffer from unexpected results or errors. Hence, there is an increasing social and technical demand for interpretable deep neural network models.
To address these issues, Professor Jong Chul Ye from the Department of Bio & Brain Engineering and his team attempted to find the geometric structure in a higher dimensional space where the structure of the deep neural network can be easily understood. They proposed a general deep learning framework, called deep convolutional framelets, to understand the mathematical principle of a deep neural network in terms of the mathematical tools in Harmonic analysis.
As a result, it was found that deep neural networks’ structure appears during the process of decomposition of high dimensionally lifted signal via Hankel matrix, which is a high-dimensional structure formerly studied intensively in the field of signal processing.
In the process of decomposing the lifted signal, two bases categorized as local and non-local basis emerge. The researchers found that non-local and local basis functions play a role in pooling and filtering operation in convolutional neural network, respectively.
Previously, when implementing AI, deep neural networks were usually constructed through empirical trial and errors. The significance of the research lies in the fact that it provides a mathematical understanding on the neural network structure in high dimensional space, which guides users to design an optimized neural network.
They demonstrated improved performance of the deep convolutional framelets’ neural networks in the applications of image denoising, image pixel in painting, and medical image restoration.
Professor Ye said, “Unlike conventional neural networks designed through trial-and-error, our theory shows that neural network structure can be optimized to each desired application and are easily predictable in their effects by exploiting the high dimensional geometry. This technology can be applied to a variety of fields requiring interpretation of the architecture, such as medical imaging.”
This research, led by PhD candidates Yoseob Han and Eunju Cha, was published in the April 26th issue of the SIAM Journal on Imaging Sciences.
Figure 1. The design of deep neural network using mathematical principles
Figure 2. The results of image noise cancelling
Figure 3. The artificial neural network restoration results in the case where 80% of the pixels are lost
2018.09.12 View 6983
Mathematical Principle behind AI's 'Black Box'
(from left: Professor Jong Chul Ye, PhD candidates Yoseob Han and Eunju Cha)
A KAIST research team identified the geometrical structure of artificial intelligence (AI) and discovered the mathematical principles of highly performing artificial neural networks, which can be applicable in fields such as medical imaging.
Deep neural networks are an exemplary method of implementing deep learning, which is at the core of the AI technology, and have shown explosive growth in recent years. This technique has been used in various fields, such as image and speech recognition as well as image processing.
Despite its excellent performance and usefulness, the exact working principles of deep neural networks has not been well understood, and they often suffer from unexpected results or errors. Hence, there is an increasing social and technical demand for interpretable deep neural network models.
To address these issues, Professor Jong Chul Ye from the Department of Bio & Brain Engineering and his team attempted to find the geometric structure in a higher dimensional space where the structure of the deep neural network can be easily understood. They proposed a general deep learning framework, called deep convolutional framelets, to understand the mathematical principle of a deep neural network in terms of the mathematical tools in Harmonic analysis.
As a result, it was found that deep neural networks’ structure appears during the process of decomposition of high dimensionally lifted signal via Hankel matrix, which is a high-dimensional structure formerly studied intensively in the field of signal processing.
In the process of decomposing the lifted signal, two bases categorized as local and non-local basis emerge. The researchers found that non-local and local basis functions play a role in pooling and filtering operation in convolutional neural network, respectively.
Previously, when implementing AI, deep neural networks were usually constructed through empirical trial and errors. The significance of the research lies in the fact that it provides a mathematical understanding on the neural network structure in high dimensional space, which guides users to design an optimized neural network.
They demonstrated improved performance of the deep convolutional framelets’ neural networks in the applications of image denoising, image pixel in painting, and medical image restoration.
Professor Ye said, “Unlike conventional neural networks designed through trial-and-error, our theory shows that neural network structure can be optimized to each desired application and are easily predictable in their effects by exploiting the high dimensional geometry. This technology can be applied to a variety of fields requiring interpretation of the architecture, such as medical imaging.”
This research, led by PhD candidates Yoseob Han and Eunju Cha, was published in the April 26th issue of the SIAM Journal on Imaging Sciences.
Figure 1. The design of deep neural network using mathematical principles
Figure 2. The results of image noise cancelling
Figure 3. The artificial neural network restoration results in the case where 80% of the pixels are lost
2018.09.12 View 6983 -
 Rh Ensemble Catalyst for Effective Automobile Exhaust Treatment
(from left: Professor Hyunjoo Lee and PhD candidate Hojin Jeong)
A KAIST research team has developed a fully dispersed Rh ensemble catalyst (ENS) that shows better performance than commercial diesel oxidation catalyst (DOC). This newly developed ENSs could improve low-temperature automobile exhaust treatment.
Precious metals have been used for various heterogeneous reactions, but it is crucial to maximize efficiency of catalysts due to their high cost. Single-atom catalysts (SACs) have received much attention because it is possible for all of the metal atoms to be used for reactions, yet they do not show catalytic activity for reactions that require ensemble sites.
Meanwhile, hydrocarbons, such as propylene (C3H6) and propane (C3H8) are typical automobile exhaust gas pollutants and must be converted to carbon dioxide (CO2) and water (H2O) before they are released as exhaust. Since the hydrocarbon oxidation reaction proceeds only during carbon-carbon (C-C) or carbon-hydrogen (C-H) bond cleavage, it is essential to secure the metal ensemble site for the catalytic reaction. Therefore, precious metal catalysts with high dispersion and ensemble sites are greatly needed.
To solve this issue, Professor Hyunjoo Lee from the Department of Chemical and Biomolecular Engineering and Professor Jeong Woo Han from POSTECH developed an Rh ensemble catalyst with 100% dispersion, and applied it to automobile after-treatment. Having a 100% dispersion means that every metal atom is used for the reaction since it is exposed on the surface.
SACs also have 100% dispersion, but the difference is that ENSs have the unique advantage of having an ensemble site with two or more atoms.
As a result of the experiment, the ENSs showed excellent catalytic performance in CO, NO, propylene, and propane oxidation at low temperatures. This complements the disadvantage of nanoparticle catalyst (NPs) that perform catalysis poorly at low temperatures due to low metal dispersion, or SACs without hydrocarbon oxidation.
In particular, the ENSs have superior low-temperature activity even better than commercial DOC, hence they are expected to be applied to automobile exhaust treatment.
Professor Lee said, “I believe that the ENSs have given academic contribution for proposing a new concept of metal catalysts, differentiating from conventional SACs and NPs. At the same time, they are of great value in the industry of exhaust treatment catalysts.”
This research, led by PhD candidate Hojin Jeong, was published in the Journal of the American Chemical Society on July 5.
Figure 1. Concept of Rh ensemble catalyst for automobile exhaust treatment
Figure 2. Structure and performance comparison of single-atom catalyst and ensemble catalyst
Figure 3. Energy-dispersive X-ray spectroscopy (EDS) mapping images for SAC, ENS, and NP, respectively (green, Eh; red, Ce)
2018.08.29 View 8100
Rh Ensemble Catalyst for Effective Automobile Exhaust Treatment
(from left: Professor Hyunjoo Lee and PhD candidate Hojin Jeong)
A KAIST research team has developed a fully dispersed Rh ensemble catalyst (ENS) that shows better performance than commercial diesel oxidation catalyst (DOC). This newly developed ENSs could improve low-temperature automobile exhaust treatment.
Precious metals have been used for various heterogeneous reactions, but it is crucial to maximize efficiency of catalysts due to their high cost. Single-atom catalysts (SACs) have received much attention because it is possible for all of the metal atoms to be used for reactions, yet they do not show catalytic activity for reactions that require ensemble sites.
Meanwhile, hydrocarbons, such as propylene (C3H6) and propane (C3H8) are typical automobile exhaust gas pollutants and must be converted to carbon dioxide (CO2) and water (H2O) before they are released as exhaust. Since the hydrocarbon oxidation reaction proceeds only during carbon-carbon (C-C) or carbon-hydrogen (C-H) bond cleavage, it is essential to secure the metal ensemble site for the catalytic reaction. Therefore, precious metal catalysts with high dispersion and ensemble sites are greatly needed.
To solve this issue, Professor Hyunjoo Lee from the Department of Chemical and Biomolecular Engineering and Professor Jeong Woo Han from POSTECH developed an Rh ensemble catalyst with 100% dispersion, and applied it to automobile after-treatment. Having a 100% dispersion means that every metal atom is used for the reaction since it is exposed on the surface.
SACs also have 100% dispersion, but the difference is that ENSs have the unique advantage of having an ensemble site with two or more atoms.
As a result of the experiment, the ENSs showed excellent catalytic performance in CO, NO, propylene, and propane oxidation at low temperatures. This complements the disadvantage of nanoparticle catalyst (NPs) that perform catalysis poorly at low temperatures due to low metal dispersion, or SACs without hydrocarbon oxidation.
In particular, the ENSs have superior low-temperature activity even better than commercial DOC, hence they are expected to be applied to automobile exhaust treatment.
Professor Lee said, “I believe that the ENSs have given academic contribution for proposing a new concept of metal catalysts, differentiating from conventional SACs and NPs. At the same time, they are of great value in the industry of exhaust treatment catalysts.”
This research, led by PhD candidate Hojin Jeong, was published in the Journal of the American Chemical Society on July 5.
Figure 1. Concept of Rh ensemble catalyst for automobile exhaust treatment
Figure 2. Structure and performance comparison of single-atom catalyst and ensemble catalyst
Figure 3. Energy-dispersive X-ray spectroscopy (EDS) mapping images for SAC, ENS, and NP, respectively (green, Eh; red, Ce)
2018.08.29 View 8100 -
 Successful Synthesis of Gamma-Lanctam Rings from Hydrocarbons
(The team of Professor Chang, far right, at the Department of Chemistry)
KAIST chemists have designed a novel strategy to synthesize ring-shaped cyclic molecules, highly sought-after by pharmaceutical and chemical industries, and known as gamma-lactams. This study describes how these five-membered rings can be prepared from inexpensive and readily available feedstock hydrocarbons, as well as from complex organic molecules, such as amino acids and steroids.
Gamma-lactams find several applications in medicinal, synthetic, and material chemistry. For example, they are included in a large number of pharmaceutically active compounds with antibiotic, anti-inflammatory, and anti-tumoral functions. This research was published in Science on March 2.
Conversion of hydrocarbons into nitrogen-containing compounds is an important area of research, where the challenge lies in breaking strong carbon-hydrogen (C−H) bonds, and converting them into carbon-nitrogen (C–N) bonds in a controlled fashion. For this reason, hydrocarbons are difficult to use as starting materials, albeit the fact that they exist in large quantities in nature.
Over the last 35 years, chemists have found ways of converting simple hydrocarbons into nitrogen-containing rings, such as indoles or pyrrolidines, but gamma-lactams proved impossible to prepare using the same approaches. Researchers hypothesized that such failure was due to alternative chemical pathways that steer the reaction away from the wanted rings: The reaction intermediate (carbonylnitrene) quickly breaks down into unsought products. Using computer models of the desired and undesired reaction pathways, the team found a strategy to completely shut down the latter in order to obtain the longed-for gamma-lactams. For the first time, these four carbons and one nitrogen cyclic molecules were obtained directly from simple feedstock chemicals.
Led by Professor Chang Sukbok at the Department of Chemistry, the team designed the winning reaction with the help of computer simulations that analyze the reaction mechanisms and calculate the energy required for the reaction to take place. According to such computer predictions, the reaction could follow three pathways, leading to the formation of either the desired gamma-lactam, an unwanted product (isocyanate), or the degradation of the catalyst caused by the substrate reacting with the catalyst backbone. Combining experimental observations and detailed computer simulations, the team designed an iridium-based catalyst, highly selective for the gamma-lactam formation. In this way, the two undesired pathways were systematically shut down, leaving the formation of the nitrogen-containing ring as the only possible outcome. Professor Chang is also in charge of the Center for Catalytic Hydrocarbon Functionalizations at the Institute for Basic Science (IBS).
“With this work we offer a brand new solution to a long-standing challenge and demonstrate the power of what we call mechanism-based reaction development,” explains Professor Baik Mu-Hyun, a corresponding author of the study.
Beyond using cheap feedstock hydrocarbons as substrates, the team was also successful in converting amino acids, steroids, and other bio-relevant molecules into gamma-lactams, which might find a variety of applications as plant insecticide, drugs against parasitic worms, or anti-aging agents. This new synthetic technology gives much easier access to these complicated molecules and will enable the development of potential drugs in a much shorter amount of time at a lower cost.
Figure 1: Selective amidation reaction using newly designed iridium (Ir) catalysts. Abundant in nature Hydrocarbons are used as substrates to synthesize nitrogen-containing ring, called gamma-lactams.
Figure 2: Three possible reaction pathways and energy barriers predicted by computational chemistry. The scientists developed new iridium-based catalysts that are highly selective for the C–H insertion pathway which leads to the desired gamma-lactam molecules.
Figure 3: Interesting gamma-lactams derived from natural and unnatural amino acids, steroids, etc., which may be used to protect plants against insects, fight parasitic worms, or as anti-aging agents.
2018.03.02 View 9568
Successful Synthesis of Gamma-Lanctam Rings from Hydrocarbons
(The team of Professor Chang, far right, at the Department of Chemistry)
KAIST chemists have designed a novel strategy to synthesize ring-shaped cyclic molecules, highly sought-after by pharmaceutical and chemical industries, and known as gamma-lactams. This study describes how these five-membered rings can be prepared from inexpensive and readily available feedstock hydrocarbons, as well as from complex organic molecules, such as amino acids and steroids.
Gamma-lactams find several applications in medicinal, synthetic, and material chemistry. For example, they are included in a large number of pharmaceutically active compounds with antibiotic, anti-inflammatory, and anti-tumoral functions. This research was published in Science on March 2.
Conversion of hydrocarbons into nitrogen-containing compounds is an important area of research, where the challenge lies in breaking strong carbon-hydrogen (C−H) bonds, and converting them into carbon-nitrogen (C–N) bonds in a controlled fashion. For this reason, hydrocarbons are difficult to use as starting materials, albeit the fact that they exist in large quantities in nature.
Over the last 35 years, chemists have found ways of converting simple hydrocarbons into nitrogen-containing rings, such as indoles or pyrrolidines, but gamma-lactams proved impossible to prepare using the same approaches. Researchers hypothesized that such failure was due to alternative chemical pathways that steer the reaction away from the wanted rings: The reaction intermediate (carbonylnitrene) quickly breaks down into unsought products. Using computer models of the desired and undesired reaction pathways, the team found a strategy to completely shut down the latter in order to obtain the longed-for gamma-lactams. For the first time, these four carbons and one nitrogen cyclic molecules were obtained directly from simple feedstock chemicals.
Led by Professor Chang Sukbok at the Department of Chemistry, the team designed the winning reaction with the help of computer simulations that analyze the reaction mechanisms and calculate the energy required for the reaction to take place. According to such computer predictions, the reaction could follow three pathways, leading to the formation of either the desired gamma-lactam, an unwanted product (isocyanate), or the degradation of the catalyst caused by the substrate reacting with the catalyst backbone. Combining experimental observations and detailed computer simulations, the team designed an iridium-based catalyst, highly selective for the gamma-lactam formation. In this way, the two undesired pathways were systematically shut down, leaving the formation of the nitrogen-containing ring as the only possible outcome. Professor Chang is also in charge of the Center for Catalytic Hydrocarbon Functionalizations at the Institute for Basic Science (IBS).
“With this work we offer a brand new solution to a long-standing challenge and demonstrate the power of what we call mechanism-based reaction development,” explains Professor Baik Mu-Hyun, a corresponding author of the study.
Beyond using cheap feedstock hydrocarbons as substrates, the team was also successful in converting amino acids, steroids, and other bio-relevant molecules into gamma-lactams, which might find a variety of applications as plant insecticide, drugs against parasitic worms, or anti-aging agents. This new synthetic technology gives much easier access to these complicated molecules and will enable the development of potential drugs in a much shorter amount of time at a lower cost.
Figure 1: Selective amidation reaction using newly designed iridium (Ir) catalysts. Abundant in nature Hydrocarbons are used as substrates to synthesize nitrogen-containing ring, called gamma-lactams.
Figure 2: Three possible reaction pathways and energy barriers predicted by computational chemistry. The scientists developed new iridium-based catalysts that are highly selective for the C–H insertion pathway which leads to the desired gamma-lactam molecules.
Figure 3: Interesting gamma-lactams derived from natural and unnatural amino acids, steroids, etc., which may be used to protect plants against insects, fight parasitic worms, or as anti-aging agents.
2018.03.02 View 9568 -
 Platinum Single Atom Catalysts for 'Direct Formic Acid Fuel Cells'
(Professor Hyunjoo Lee (left) and Ph.D. candidate Jiwhan Kim)
A research team co-led by Professor Hyunjoo Lee at the Department of Chemical and Biomolecular Engineering at KAIST and Professor Jeong Woo Han from the University of Seoul synthesized highly stable high-Pt-content single atom catalysts for direct formic acid fuel cells. The amount of platinum can be reduced to 1/10 of that of conventional platinum nanoparticle catalysts.
Platinum (Pt) catalysts have been used in various catalytic reactions due to their high activity and stability. However, because Pt is rare and expensive, it is important to reduce the amount of Pt used. Pt single atom catalysts can reduce the size of the Pt particles to the size of an atom. Thus, the cost of Pt catalysts can be minimized because all of the Pt atoms can participate in the catalytic reactions. Additionally, single atom catalysts have no ensemble site in which two or more atoms are attached, and thus, the reaction selectivity is different from that of nanoparticle catalysts.
Despite these advantages, single atom catalysts are easily aggregated and less stable due to their low coordination number and high surface free energy. It is difficult to develop a single atom catalyst with high content and high stability, and thus, its application in practical devices is limited.
Direct formic acid fuel cells can be an energy source for next-generation portable devices because liquid formic acid as a fuel is safer and easier to store and transport than high-pressure hydrogen gas.
To improve the stability of Pt single atom catalysts, Professor Lee’s group developed a Pt-Sn single atom alloy structure on an antimony-doped tin oxide (ATO) support. This structure has been proven by computational calculations which show that Pt single atoms substitute antimony sites in the antimony-tin alloy structure and are thermodynamically stable. This catalyst has been shown to have a higher activity up to 50 times per weight of Pt than that of the commercial catalyst, Pt/C, in the oxidation of formic acid, and the stability of the catalyst was also remarkably high.
Professor Lee’s group also used a single atomic catalyst in a 'direct formic acid fuel cell’ consisting of membranes and electrodes. It is the first attempt to apply a single atomic catalyst to a full cell. In this case, an output similar to that of the commercial catalyst could be obtained by using 1/10 of the platinum compared to the commercial Pt/C catalyst.
Ph.D. candidate Jiwhan Kim from KAIST was the first author of the research. This research was published online on September 11 in Advanced Energy Materials.
This research was carried out with the support of the Samsung Electronics Future Technology Development Center.
(Figure 1. Concept photograph for Pt single atom catalysts.)
(Figure 2. Pt single atom catalysts by HAADF-STEM analysis (bright white circles))
2017.10.31 View 8263
Platinum Single Atom Catalysts for 'Direct Formic Acid Fuel Cells'
(Professor Hyunjoo Lee (left) and Ph.D. candidate Jiwhan Kim)
A research team co-led by Professor Hyunjoo Lee at the Department of Chemical and Biomolecular Engineering at KAIST and Professor Jeong Woo Han from the University of Seoul synthesized highly stable high-Pt-content single atom catalysts for direct formic acid fuel cells. The amount of platinum can be reduced to 1/10 of that of conventional platinum nanoparticle catalysts.
Platinum (Pt) catalysts have been used in various catalytic reactions due to their high activity and stability. However, because Pt is rare and expensive, it is important to reduce the amount of Pt used. Pt single atom catalysts can reduce the size of the Pt particles to the size of an atom. Thus, the cost of Pt catalysts can be minimized because all of the Pt atoms can participate in the catalytic reactions. Additionally, single atom catalysts have no ensemble site in which two or more atoms are attached, and thus, the reaction selectivity is different from that of nanoparticle catalysts.
Despite these advantages, single atom catalysts are easily aggregated and less stable due to their low coordination number and high surface free energy. It is difficult to develop a single atom catalyst with high content and high stability, and thus, its application in practical devices is limited.
Direct formic acid fuel cells can be an energy source for next-generation portable devices because liquid formic acid as a fuel is safer and easier to store and transport than high-pressure hydrogen gas.
To improve the stability of Pt single atom catalysts, Professor Lee’s group developed a Pt-Sn single atom alloy structure on an antimony-doped tin oxide (ATO) support. This structure has been proven by computational calculations which show that Pt single atoms substitute antimony sites in the antimony-tin alloy structure and are thermodynamically stable. This catalyst has been shown to have a higher activity up to 50 times per weight of Pt than that of the commercial catalyst, Pt/C, in the oxidation of formic acid, and the stability of the catalyst was also remarkably high.
Professor Lee’s group also used a single atomic catalyst in a 'direct formic acid fuel cell’ consisting of membranes and electrodes. It is the first attempt to apply a single atomic catalyst to a full cell. In this case, an output similar to that of the commercial catalyst could be obtained by using 1/10 of the platinum compared to the commercial Pt/C catalyst.
Ph.D. candidate Jiwhan Kim from KAIST was the first author of the research. This research was published online on September 11 in Advanced Energy Materials.
This research was carried out with the support of the Samsung Electronics Future Technology Development Center.
(Figure 1. Concept photograph for Pt single atom catalysts.)
(Figure 2. Pt single atom catalysts by HAADF-STEM analysis (bright white circles))
2017.10.31 View 8263 -
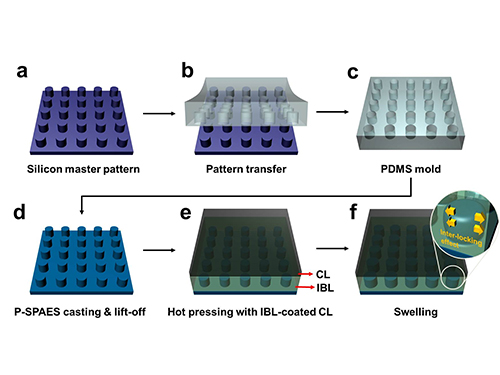 Membrane
Scientists at KAIST have developed a new way of making fuel cell membranes using nanoscale fasteners, paving the way for lower-cost, higher-efficiency and more easily manufactured fuel cells.
The internal workings of fuel cells vary, but basically all types mix hydrogen and oxygen to produce a chemical reaction that delivers usable electricity and exhausts ordinary water as a by-product. One of the most efficient types is the proton exchange membrane (PEM) fuel cell, which operates at low enough temperatures to be used in homes and vehicles.
To generate electricity, PEM fuel cells rely on two chemical compartments separated by a permeable catalyst membrane. This membrane acts as an electrolyte; a negative electrode is bonded to one side of the membrane and a positive electrode is bonded to the other. The electrolyte membrane is often based on a polymer of perfluorosulfonic acid. Due to its high cost, however, a less expensive hydrocarbon-based electrolyte membrane has attracted interest in this technology sector.
Until now, the challenge in adopting such a hydrocarbon membrane has been that the interface between the electrode and hydrocarbon membrane is weak. This causes the membrane to delaminate relatively easily, falling apart and losing efficiency with use.
Professor Hee-Tak Kim of the Department of Chemical and Biomolecular Engineering at the Korea Advanced Institute of Science and Technology (KAIST) and his research team have developed a new fastening system that bonds the two materials mechanically rather than chemically. This opens the way to the development of fuel cell membranes that are less expensive, easier to manufacture, stronger and more efficient.
The researchers achieved this by moulding a pattern of tiny cylindrical pillars on the face of the hydrocarbon membrane. The pillars protrude into a softened skin of the electrode with heat. The mechanical bond sets and strengthens as the material cools and absorbs water. The pillar-patterned hydrocarbon membrane is cast using silicone moulds.
Professor Kim said, “This physically fastened bond is almost five times stronger and harder to separate than current bonds between the same layers.”
The new interlocking method also appears to offer a way to bond many types of hydrocarbon membranes that, until now, have been rejected because they couldn’t be fastened robustly. This would make hydrocarbon membranes practical for a number of applications beyond fuel cells such as rechargeable “redox flow” batteries.
The research team is now developing a stronger and more scalable interlocking interface for their nanoscale fasteners.
Picture: Schematic Diagram of the Fabrication of the Pillar P-SPAES Membrane and Its Working Principle of Interlocking Effects
2015.11.06 View 10840
Membrane
Scientists at KAIST have developed a new way of making fuel cell membranes using nanoscale fasteners, paving the way for lower-cost, higher-efficiency and more easily manufactured fuel cells.
The internal workings of fuel cells vary, but basically all types mix hydrogen and oxygen to produce a chemical reaction that delivers usable electricity and exhausts ordinary water as a by-product. One of the most efficient types is the proton exchange membrane (PEM) fuel cell, which operates at low enough temperatures to be used in homes and vehicles.
To generate electricity, PEM fuel cells rely on two chemical compartments separated by a permeable catalyst membrane. This membrane acts as an electrolyte; a negative electrode is bonded to one side of the membrane and a positive electrode is bonded to the other. The electrolyte membrane is often based on a polymer of perfluorosulfonic acid. Due to its high cost, however, a less expensive hydrocarbon-based electrolyte membrane has attracted interest in this technology sector.
Until now, the challenge in adopting such a hydrocarbon membrane has been that the interface between the electrode and hydrocarbon membrane is weak. This causes the membrane to delaminate relatively easily, falling apart and losing efficiency with use.
Professor Hee-Tak Kim of the Department of Chemical and Biomolecular Engineering at the Korea Advanced Institute of Science and Technology (KAIST) and his research team have developed a new fastening system that bonds the two materials mechanically rather than chemically. This opens the way to the development of fuel cell membranes that are less expensive, easier to manufacture, stronger and more efficient.
The researchers achieved this by moulding a pattern of tiny cylindrical pillars on the face of the hydrocarbon membrane. The pillars protrude into a softened skin of the electrode with heat. The mechanical bond sets and strengthens as the material cools and absorbs water. The pillar-patterned hydrocarbon membrane is cast using silicone moulds.
Professor Kim said, “This physically fastened bond is almost five times stronger and harder to separate than current bonds between the same layers.”
The new interlocking method also appears to offer a way to bond many types of hydrocarbon membranes that, until now, have been rejected because they couldn’t be fastened robustly. This would make hydrocarbon membranes practical for a number of applications beyond fuel cells such as rechargeable “redox flow” batteries.
The research team is now developing a stronger and more scalable interlocking interface for their nanoscale fasteners.
Picture: Schematic Diagram of the Fabrication of the Pillar P-SPAES Membrane and Its Working Principle of Interlocking Effects
2015.11.06 View 10840 -
 KAIST Agrees to Personnel Exchange with the Ministry of Economic Affairs of the Republic of China
A delegation from the Ministry of Economic Affairs (MOEA) of the Republic of China (ROC) visited KAIST on October 16, 2015.
President Steve Kang of KAIST and Director General Yu-Ping Lien of the Department of Investment Services, MOEA, signed a memorandum of understanding (MOU) on exchanging personnel, recruiting and attracting top talents, and sharing job information.
With the MOU, KAIST and MOEA will develop a mobility program for students in Korea and ROC to help them seek employment opportunities in both nations. Director General Lien hoped that the MOU would bring more of KAIST students in the information technology field to work in ROC.
President Kang responded,
“KAIST has fostered highly talented engineers and researchers across all fields of science and engineering. If these students can have a place in ROC to realize their potential, this certainly could benefit the two nations.”
In the picture from left to right is Director General Yu-Ping Lien of the Department of Investment Services, the Ministry of Economic Affairs of the Republic of China and President Steve Kang of KAIST.
2015.10.17 View 6396
KAIST Agrees to Personnel Exchange with the Ministry of Economic Affairs of the Republic of China
A delegation from the Ministry of Economic Affairs (MOEA) of the Republic of China (ROC) visited KAIST on October 16, 2015.
President Steve Kang of KAIST and Director General Yu-Ping Lien of the Department of Investment Services, MOEA, signed a memorandum of understanding (MOU) on exchanging personnel, recruiting and attracting top talents, and sharing job information.
With the MOU, KAIST and MOEA will develop a mobility program for students in Korea and ROC to help them seek employment opportunities in both nations. Director General Lien hoped that the MOU would bring more of KAIST students in the information technology field to work in ROC.
President Kang responded,
“KAIST has fostered highly talented engineers and researchers across all fields of science and engineering. If these students can have a place in ROC to realize their potential, this certainly could benefit the two nations.”
In the picture from left to right is Director General Yu-Ping Lien of the Department of Investment Services, the Ministry of Economic Affairs of the Republic of China and President Steve Kang of KAIST.
2015.10.17 View 6396