AI
-
 Taming AI: Engineering, Ethics, and Policy
(Professor Lee, Professor Koene, Professor Walsh, and Professor Ema (from left))
Can AI-powered robotics could be adequate companions for humans? Will the good faith of users and developers work for helping AI-powered robots become the new tribe of the digital future?
AI’s efficiency is creating new socio-economic opportunities in the global market. Despite the opportunities, challenges still remain. It is said that efficiency-enforcing algorithms through deep learning will take an eventual toll on human dignity and safety, bringing out the disastrous fiascos featured in the Terminator movies.
A research group at the Korean Flagship AI Project for Emotional Digital Companionship at KAIST Institute for AI (KI4AI) and the Fourth Industrial Intelligence Center at KAIST Institute co-hosted a seminar, “Taming AI: Engineering, Ethics, and Policy” last week to discuss ways to better employ AI technologies in ways that upholds human values.
The KI4AI has been conducting this flagship project from the end of 2016 with the support of the Ministry of Science and ICT.
The seminar brought together three speakers from Australia, Japan, and the UK to better fathom the implications of the new technology emergence from the ethical perspectives of engineering and discuss policymaking for the responsible usage of technology.
Professor Toby Walsh, an anti-autonomous weapon activist from New South Wales University in Australia continued to argue the possible risk that AI poses to malfunction. He said that an independent ethics committee or group usually monitors academic institutions’ research activities in order to avoid any possible mishaps.
However, he said there is no independent group or committee monitoring the nature of corporations’ engagement of such technologies, while its possible threats against humanity are alleged to be growing. He mentioned that Google’s and Amazon’s information collecting also pose a potent threat. He said that ethical standards similar to academic research integrity should be established to avoid the possible restricting of the dignity of humans and mass destruction. He hoped that KAIST and Google would play a leading role in establishing an international norm toward this compelling issue.
Professor Arisa Ema from the University of Tokyo provided very compelling arguments for thinking about the duplicity of technology and how technology should serve the public interest without any bias against gender, race, and social stratum. She pointed out the information dominated by several Western corporations like Google. She said that such algorithms for deep learning of data provided by several Western corporations will create very biased information, only applicable to limited races and classes.
Meanwhile, Professor Ansgar Koene from the University of Nottingham presented the IEEE’s global initiative on the ethics of autonomous and intelligence systems. He shared the cases of industry standards and ethically-aligned designs made by the IEEE Standards Association. He said more than 250 global cross-disciplinary thought leaders from around the world joined to develop ethical guidelines called Ethically Aligned Design (EAD) V2. EAD V2 includes methodologies to guide ethical research and design, embedding values into autonomous intelligence systems among others. For the next step beyond EAD V2, the association is now working for IEEE P70xx Standards Projects, detailing more technical approaches.
Professor Soo Young Lee at KAIST argued that the eventual goal of complete AI is to have human-like emotions, calling it a new paradigm for the relationship between humans and AI-robots. According to Professor Lee, AI-powered robots will serve as a good companion for humans. “Especially in aging societies affecting the globe, this will be a very viable and practical option,” he said.
He pointed out, “Kids learn from parents’ morality and social behavior. Users should have AI-robots learn morality as well. Their relationships should be based on good faith and trust, no longer that of master and slave. He said that liability issues for any mishap will need to be discussed further, but basically each user and developer should have their own responsibility when dealing with these issues.
2018.06.26 View 10235
Taming AI: Engineering, Ethics, and Policy
(Professor Lee, Professor Koene, Professor Walsh, and Professor Ema (from left))
Can AI-powered robotics could be adequate companions for humans? Will the good faith of users and developers work for helping AI-powered robots become the new tribe of the digital future?
AI’s efficiency is creating new socio-economic opportunities in the global market. Despite the opportunities, challenges still remain. It is said that efficiency-enforcing algorithms through deep learning will take an eventual toll on human dignity and safety, bringing out the disastrous fiascos featured in the Terminator movies.
A research group at the Korean Flagship AI Project for Emotional Digital Companionship at KAIST Institute for AI (KI4AI) and the Fourth Industrial Intelligence Center at KAIST Institute co-hosted a seminar, “Taming AI: Engineering, Ethics, and Policy” last week to discuss ways to better employ AI technologies in ways that upholds human values.
The KI4AI has been conducting this flagship project from the end of 2016 with the support of the Ministry of Science and ICT.
The seminar brought together three speakers from Australia, Japan, and the UK to better fathom the implications of the new technology emergence from the ethical perspectives of engineering and discuss policymaking for the responsible usage of technology.
Professor Toby Walsh, an anti-autonomous weapon activist from New South Wales University in Australia continued to argue the possible risk that AI poses to malfunction. He said that an independent ethics committee or group usually monitors academic institutions’ research activities in order to avoid any possible mishaps.
However, he said there is no independent group or committee monitoring the nature of corporations’ engagement of such technologies, while its possible threats against humanity are alleged to be growing. He mentioned that Google’s and Amazon’s information collecting also pose a potent threat. He said that ethical standards similar to academic research integrity should be established to avoid the possible restricting of the dignity of humans and mass destruction. He hoped that KAIST and Google would play a leading role in establishing an international norm toward this compelling issue.
Professor Arisa Ema from the University of Tokyo provided very compelling arguments for thinking about the duplicity of technology and how technology should serve the public interest without any bias against gender, race, and social stratum. She pointed out the information dominated by several Western corporations like Google. She said that such algorithms for deep learning of data provided by several Western corporations will create very biased information, only applicable to limited races and classes.
Meanwhile, Professor Ansgar Koene from the University of Nottingham presented the IEEE’s global initiative on the ethics of autonomous and intelligence systems. He shared the cases of industry standards and ethically-aligned designs made by the IEEE Standards Association. He said more than 250 global cross-disciplinary thought leaders from around the world joined to develop ethical guidelines called Ethically Aligned Design (EAD) V2. EAD V2 includes methodologies to guide ethical research and design, embedding values into autonomous intelligence systems among others. For the next step beyond EAD V2, the association is now working for IEEE P70xx Standards Projects, detailing more technical approaches.
Professor Soo Young Lee at KAIST argued that the eventual goal of complete AI is to have human-like emotions, calling it a new paradigm for the relationship between humans and AI-robots. According to Professor Lee, AI-powered robots will serve as a good companion for humans. “Especially in aging societies affecting the globe, this will be a very viable and practical option,” he said.
He pointed out, “Kids learn from parents’ morality and social behavior. Users should have AI-robots learn morality as well. Their relationships should be based on good faith and trust, no longer that of master and slave. He said that liability issues for any mishap will need to be discussed further, but basically each user and developer should have their own responsibility when dealing with these issues.
2018.06.26 View 10235 -
 Printed Thermo-Plasmonic Heat Patterns for Neurological Disorder Treatment
(Professor Nam and Dr. Kang, right)
A KAIST team presented a highly customizable neural stimulation method. The research team developed a technology that can print the heat pattern on a micron scale to enable the control of biological activities remotely.
The researchers integrated a precision inkjet printing technology with bio-functional thermo-plasmonic nanoparticles to achieve a ‘selective nano-photothermal neural stimulation method.’
The research team of Professor Yoonkey Nam at the Department of Bio and Brain Engineering expects this will serve as an enabling technology for personalized precision neuromodulation therapy for patients with neurological disorders.
The nano-photothermal neural stimulation method uses the thermo-plasmonic effect of metal nanoparticles to modulate the activities of neuronal networks. With the thermo-plasmonic effect, metal nanoparticles can absorb specific wavelength of illuminated light to efficiently generate localized heat. The research team discovered the inhibitory behavior of spontaneous activities of neurons upon photothermal stimulation four years ago. Since then, they have developed this technology to control hyperactive behaviors of neurons and neural circuits, which is often found in neurological disorders such as epilepsy.
In order to overcome the limitation on the spatial selectivity and resolution of the previously developed nano-photothermal method, the team adopted an inkjet printing technology to micro pattern the plasmonic nanoparticles (a few tens of microns), and successfully demonstrated that the nano-photothermal stimulation can be selectively applied according to the printed patterns.
The researchers applied a polyelectrolyte layer-by-layer coating method to printing substrates in a way to improve the pattern fidelity and achieve the uniform assembly of nanoparticles. The electrostatic attraction between the printed nanoparticles and the coated printing substrate also helped the stability of the attached nanoparticles. Because the polyelectrolyte coating is biocompatible, biological experiments including cell culture are possible with the technology developed in this work.
Using printed gold nanorod particles in a few tens of microns resolution over a several centimeters area, the researchers showed that highly complex heat patterns can be precisely formed upon light illumination according to the printing image.
Lastly, the team confirmed that the printed heat patterns can selectively and instantaneously inhibit the activities of cultured hippocampal neurons upon near-infrared light illumination. Because the printing process is applicable to thin and flexible substrates, the technology can be easily applied to implantable neurological disorder treatment devices and wearable devices. By selectively applying the heat patterns to only the desired cellular areas, customized and personalized photothermal neuromodulation therapy can be applied to patients.
“The fact that any desired heat patterns can be simply ‘printed’ anywhere broadens the applicability of this technology in many engineering fields. In bioengineering, it can be applied to neural interfaces using light and heat to modulate physiological functions. As another engineering application, for example, printed heat patterns can be used as a new concept of anti-counterfeit applications,” said the principal investigator, Yoonkey Nam at KAIST. This work, led mainly by Dr. Hongki Kang, was published in ACS Nano on February 5th 2018.
2018.04.06 View 7559
Printed Thermo-Plasmonic Heat Patterns for Neurological Disorder Treatment
(Professor Nam and Dr. Kang, right)
A KAIST team presented a highly customizable neural stimulation method. The research team developed a technology that can print the heat pattern on a micron scale to enable the control of biological activities remotely.
The researchers integrated a precision inkjet printing technology with bio-functional thermo-plasmonic nanoparticles to achieve a ‘selective nano-photothermal neural stimulation method.’
The research team of Professor Yoonkey Nam at the Department of Bio and Brain Engineering expects this will serve as an enabling technology for personalized precision neuromodulation therapy for patients with neurological disorders.
The nano-photothermal neural stimulation method uses the thermo-plasmonic effect of metal nanoparticles to modulate the activities of neuronal networks. With the thermo-plasmonic effect, metal nanoparticles can absorb specific wavelength of illuminated light to efficiently generate localized heat. The research team discovered the inhibitory behavior of spontaneous activities of neurons upon photothermal stimulation four years ago. Since then, they have developed this technology to control hyperactive behaviors of neurons and neural circuits, which is often found in neurological disorders such as epilepsy.
In order to overcome the limitation on the spatial selectivity and resolution of the previously developed nano-photothermal method, the team adopted an inkjet printing technology to micro pattern the plasmonic nanoparticles (a few tens of microns), and successfully demonstrated that the nano-photothermal stimulation can be selectively applied according to the printed patterns.
The researchers applied a polyelectrolyte layer-by-layer coating method to printing substrates in a way to improve the pattern fidelity and achieve the uniform assembly of nanoparticles. The electrostatic attraction between the printed nanoparticles and the coated printing substrate also helped the stability of the attached nanoparticles. Because the polyelectrolyte coating is biocompatible, biological experiments including cell culture are possible with the technology developed in this work.
Using printed gold nanorod particles in a few tens of microns resolution over a several centimeters area, the researchers showed that highly complex heat patterns can be precisely formed upon light illumination according to the printing image.
Lastly, the team confirmed that the printed heat patterns can selectively and instantaneously inhibit the activities of cultured hippocampal neurons upon near-infrared light illumination. Because the printing process is applicable to thin and flexible substrates, the technology can be easily applied to implantable neurological disorder treatment devices and wearable devices. By selectively applying the heat patterns to only the desired cellular areas, customized and personalized photothermal neuromodulation therapy can be applied to patients.
“The fact that any desired heat patterns can be simply ‘printed’ anywhere broadens the applicability of this technology in many engineering fields. In bioengineering, it can be applied to neural interfaces using light and heat to modulate physiological functions. As another engineering application, for example, printed heat patterns can be used as a new concept of anti-counterfeit applications,” said the principal investigator, Yoonkey Nam at KAIST. This work, led mainly by Dr. Hongki Kang, was published in ACS Nano on February 5th 2018.
2018.04.06 View 7559 -
 Aqueous Storage Device Needs Only 20 Seconds to Go
(from left: PhD candidate Il Woo Ock and Professor Jeung Ku Kang)
A KAIST research team developed a new hybrid energy storage device that can be charged in less than half a minute. It employs aqueous electrolytes instead of flammable organic solvents, so it is both environmentally friendly and safe. It also facilitates a boosting charge with high energy density, which makes it suitable for portable electronic devices.
Professor Jeung Ku Kang and his team from the Graduate School of Energy, Environment, Water, and Sustainability developed this hybrid energy storage with high energy and power densities along over a long cycle life by assembling fibre-like polymer chain anodes and sub-nanoscale metal oxide cathodes on graphene.
Conventional aqueous electrolyte-based energy storage devices have a limitation for boosting charges and high energy density due to low driving voltage and a shortage of anode materials.
Energy storage device capacity is determined by the two electrodes, and the balance between cathode and anode leads to high stability. In general, two electrodes show differences in electrical properties and differ in ion storage mechanism processes, resulting in poor storage and stability from the imbalance.
The research team came up with new structures and materials to facilitate rapid speed in energy exchange on the surfaces of the electrodes and minimize the energy loss between the two electrodes.
The team made anodes with graphene-based polymer chain materials. The web-like structure of graphene leads to a high surface area, thereby allowing higher capacitance.
For cathode materials, the team used metal oxide in sub-nanoscale structures to elevate atom-by-ion redox reactions. This method realized higher energy density and faster energy exchange while minimizing energy loss.
The developed device can be charged within 20 to 30 seconds using a low-power charging system, such as a USB switching charger or a flexible photovoltaic cell. The developed aqueous hybrid energy device shows more than 100-fold higher power density compared to conventional aqueous batteries and can be rapidly recharged. Further, the device showed high stability with its capacity maintained at 100% at a high charge/discharge current.
Professor Kang said, “This eco-friendly technology can be easily manufactured and is highly applicable. In particular, its high capacity and high stability, compared to existing technologies, could contribute to the commercialization of aqueous capacitors. The device can be rapidly charged using a low-power charging system, and thus can be applied to portable electronic device.”
This research, led by a PhD candidate Il Woo Ock, was published in Advanced Energy Materials on January 15.
Figure 1. Switching wearable LED kit with two AHCs in series charged by a flexible photovoltaic cell
Figure 2. Schematic diagram for aqueous hybrid capacitors
Figure 3. TEM images of anode and cathode
2018.02.28 View 13275
Aqueous Storage Device Needs Only 20 Seconds to Go
(from left: PhD candidate Il Woo Ock and Professor Jeung Ku Kang)
A KAIST research team developed a new hybrid energy storage device that can be charged in less than half a minute. It employs aqueous electrolytes instead of flammable organic solvents, so it is both environmentally friendly and safe. It also facilitates a boosting charge with high energy density, which makes it suitable for portable electronic devices.
Professor Jeung Ku Kang and his team from the Graduate School of Energy, Environment, Water, and Sustainability developed this hybrid energy storage with high energy and power densities along over a long cycle life by assembling fibre-like polymer chain anodes and sub-nanoscale metal oxide cathodes on graphene.
Conventional aqueous electrolyte-based energy storage devices have a limitation for boosting charges and high energy density due to low driving voltage and a shortage of anode materials.
Energy storage device capacity is determined by the two electrodes, and the balance between cathode and anode leads to high stability. In general, two electrodes show differences in electrical properties and differ in ion storage mechanism processes, resulting in poor storage and stability from the imbalance.
The research team came up with new structures and materials to facilitate rapid speed in energy exchange on the surfaces of the electrodes and minimize the energy loss between the two electrodes.
The team made anodes with graphene-based polymer chain materials. The web-like structure of graphene leads to a high surface area, thereby allowing higher capacitance.
For cathode materials, the team used metal oxide in sub-nanoscale structures to elevate atom-by-ion redox reactions. This method realized higher energy density and faster energy exchange while minimizing energy loss.
The developed device can be charged within 20 to 30 seconds using a low-power charging system, such as a USB switching charger or a flexible photovoltaic cell. The developed aqueous hybrid energy device shows more than 100-fold higher power density compared to conventional aqueous batteries and can be rapidly recharged. Further, the device showed high stability with its capacity maintained at 100% at a high charge/discharge current.
Professor Kang said, “This eco-friendly technology can be easily manufactured and is highly applicable. In particular, its high capacity and high stability, compared to existing technologies, could contribute to the commercialization of aqueous capacitors. The device can be rapidly charged using a low-power charging system, and thus can be applied to portable electronic device.”
This research, led by a PhD candidate Il Woo Ock, was published in Advanced Energy Materials on January 15.
Figure 1. Switching wearable LED kit with two AHCs in series charged by a flexible photovoltaic cell
Figure 2. Schematic diagram for aqueous hybrid capacitors
Figure 3. TEM images of anode and cathode
2018.02.28 View 13275 -
 Finding Human Thermal Comfort with a Watch-type Sweat Rate Sensor
(from left: Professor Young-Ho Cho and Researcher SungHyun Yoon)
KAIST developed a watch-type sweat rate sensor. This subminiature device can detect human thermal comfort accurately and steadily by measuring an individual’s sweat rate.
It is natural to sweat more in the summer and less in the winter; however, an individual’s sweat rate may vary in a given environment. Therefore, sweat can be an excellent proxy for sensing core body temperature.
Conventional sweat rate sensors using natural ventilation require bulky external devices, such as pumps and ice condensers. They are usually for physiological experiments, hence they need a manual ventilation process or high power, bulky thermos-pneumatic actuators to lift sweat rate detection chambers above skin for continuous measurement. There is also a small sweat rate sensor, but it needs a long recovery period.
To overcome these problems, Professor Young-Ho Cho and his team from the Department of Bio and Brain Engineering developed a lightweight, watch-type sweat sensor. The team integrated miniaturized thermos-pneumatic actuators for automatic natural ventilation, which allows sweat to be measured continuously.
This watch-type sensor measures sweat rate with the humidity rising rate when the chamber is closed during skin contact. Since the team integrated thermos-pneumatic actuators, the chamber no longer needs to be separated manually from skin after each measurement in order for the chamber to ventilate the collected humidity.
Moreover, this sensor is wind-resistant enough to be used for portable and wearable devices. The team identified that the sensor operates steadily with air velocity ranging up to 1.5m/s, equivalent to the average human walking speed.
Although this subminiature sensor (35mm x 25mm) only weighs 30 grams, it operates continuously for more than four hours using the conventional wrist watch batteries.
The team plans to utilize this technology for developing a new concept of cognitive air-conditioning systems recognizing Human thermal status directly; while the conventional air-conditioning systems measuring air temperature and humidity.
Professor Cho said, “Our sensor for human thermal comfort monitoring can be applied to customized or smart air conditioners. Furthermore, there will be more demands for both physical and mental healthcare, hence this technology will serve as a new platform for personalized emotional communion between humans and devices.”
This research, led by researchers Jai Kyoung Sim and SungHyun Yoon, was published in Scientific Reports on January 19, 2018.
Figure1. The fabricated watch-type sweat rate sensor for human thermal comfort monitoring
Figure 2. Views of the watch-type sweat rate sensor
Figure 3. Operation of the watch-type sweat rate sensor
2018.02.08 View 9370
Finding Human Thermal Comfort with a Watch-type Sweat Rate Sensor
(from left: Professor Young-Ho Cho and Researcher SungHyun Yoon)
KAIST developed a watch-type sweat rate sensor. This subminiature device can detect human thermal comfort accurately and steadily by measuring an individual’s sweat rate.
It is natural to sweat more in the summer and less in the winter; however, an individual’s sweat rate may vary in a given environment. Therefore, sweat can be an excellent proxy for sensing core body temperature.
Conventional sweat rate sensors using natural ventilation require bulky external devices, such as pumps and ice condensers. They are usually for physiological experiments, hence they need a manual ventilation process or high power, bulky thermos-pneumatic actuators to lift sweat rate detection chambers above skin for continuous measurement. There is also a small sweat rate sensor, but it needs a long recovery period.
To overcome these problems, Professor Young-Ho Cho and his team from the Department of Bio and Brain Engineering developed a lightweight, watch-type sweat sensor. The team integrated miniaturized thermos-pneumatic actuators for automatic natural ventilation, which allows sweat to be measured continuously.
This watch-type sensor measures sweat rate with the humidity rising rate when the chamber is closed during skin contact. Since the team integrated thermos-pneumatic actuators, the chamber no longer needs to be separated manually from skin after each measurement in order for the chamber to ventilate the collected humidity.
Moreover, this sensor is wind-resistant enough to be used for portable and wearable devices. The team identified that the sensor operates steadily with air velocity ranging up to 1.5m/s, equivalent to the average human walking speed.
Although this subminiature sensor (35mm x 25mm) only weighs 30 grams, it operates continuously for more than four hours using the conventional wrist watch batteries.
The team plans to utilize this technology for developing a new concept of cognitive air-conditioning systems recognizing Human thermal status directly; while the conventional air-conditioning systems measuring air temperature and humidity.
Professor Cho said, “Our sensor for human thermal comfort monitoring can be applied to customized or smart air conditioners. Furthermore, there will be more demands for both physical and mental healthcare, hence this technology will serve as a new platform for personalized emotional communion between humans and devices.”
This research, led by researchers Jai Kyoung Sim and SungHyun Yoon, was published in Scientific Reports on January 19, 2018.
Figure1. The fabricated watch-type sweat rate sensor for human thermal comfort monitoring
Figure 2. Views of the watch-type sweat rate sensor
Figure 3. Operation of the watch-type sweat rate sensor
2018.02.08 View 9370 -
 Technology to Find Optimum Drug Target for Cancer Developed
(Professor Kwang-Hyun Cho (right) and lead author Dr. Minsoo Choi)
A KAIST research team led by Professor Kwang-Hyun Cho of the Department of Bio and Brain Engineering developed technology to find the optimum drug target according to the type of cancer cell. The team used systems biology to analyze molecular network dynamics that reflect genetic mutations in cancer cells and to predict drug response. The technology could contribute greatly to future anti-cancer drug development.
There are many types of genetic variations found in cancer cells, including gene mutations and copy number variations. These variations differ in cancer cells even within the same type of cancer, and thus the drug response varies cell by cell. Cancer researchers worked towards identifying frequently occurring genetic variations in cancer patients and, in particular, the mutations that can be used as an index for specific drugs. Previous studies focused on identifying a single genetic mutation or creating an analysis of the structural characteristics of a gene network. However, this approach was limited in its inability to explain the biological properties of cancer which are induced by various gene and protein interactions in cancer cells, which result in differences in drug response.
Gene mutations in cancer cells not only affect the function of the affected gene, but also other genes that interact with the mutated gene and proteins. As a consequence, one mutation could lead to changes in the dynamical properties of the molecular network. Therefore, the responses to anti-cancer drugs by cancer cells differ. The current treatment approach that ignores molecular network dynamics and targets a few cancer-related genes is only effective on a fraction of patients, while many other patients exhibit resistance to the drug.
Professor Cho’s team integrated a large-scale computer simulation using super-computing and cellular experiments to analyze changes in molecular network dynamics in cancer cells.
This led to development of technology to find the optimum drug target according to the type of cancer cells by predicting drug response. This technology was applied to the molecular network of known tumor suppressor p53. The team used large-scale cancer cell genomic data available from The Cancer Cell Line Encyclopedia (CCLE) to construct different molecular networks specific to the characteristics of genetic variations.
Perturbation analysis on drug response in each molecular network was used to quantify changes in cancer cells from drug response and similar networks were clustered. Then, computer simulations were used to analyze the synergetic effects in terms of efficacy and combination to predict the level of drug response. Based on the simulation results from various cancer cell lines including lung, breast, bone, skin, kidney, and ovary cancers were used in drug response experiments for compare analysis.
This technique can be applied in any molecular network to identify the optimum drug target for personalized medicine.
The research team suggests that the technology can analyze varying drug response due to the heterogeneity of cancer cells by considering the overall modulatory interactions rather than focusing only on a specific gene or protein. Further, the technology aids the prediction of causes of drug resistance and thus the identification of the optimum drug target to inhibit the resistance. This could be core source technology that can be used in drug repositioning, a process of applying existing drugs to new disease targets.
Professor Cho said, “Genetic variations in cancer cells are the cause of diverse drug response, but a complete analysis had not yet been made.” He continued, “Systems biology allowed the simulation of drug responses by cancer cell molecular networks to identify fundamental principles of drug response and optimum drug targets using a new conceptual approach.”
This research was published in Nature Communications on December 5 and was funded by Ministry of Science and ICT and National Research Foundation of Korea.
(Figure 1. Drug response prediction for each cancer cell type from computer simulation and cellular experiment verification for comparison)
(Figure 2. Drug response prediction based on cancer cell molecular network dynamics and clustering of cancer cells by their molecular networks)
(Figure 3. Identification of drug target for each cancer cell type by cellular molecular network analysis and establishment for personalized medicine strategy for each cancer patient)
2017.12.15 View 7624
Technology to Find Optimum Drug Target for Cancer Developed
(Professor Kwang-Hyun Cho (right) and lead author Dr. Minsoo Choi)
A KAIST research team led by Professor Kwang-Hyun Cho of the Department of Bio and Brain Engineering developed technology to find the optimum drug target according to the type of cancer cell. The team used systems biology to analyze molecular network dynamics that reflect genetic mutations in cancer cells and to predict drug response. The technology could contribute greatly to future anti-cancer drug development.
There are many types of genetic variations found in cancer cells, including gene mutations and copy number variations. These variations differ in cancer cells even within the same type of cancer, and thus the drug response varies cell by cell. Cancer researchers worked towards identifying frequently occurring genetic variations in cancer patients and, in particular, the mutations that can be used as an index for specific drugs. Previous studies focused on identifying a single genetic mutation or creating an analysis of the structural characteristics of a gene network. However, this approach was limited in its inability to explain the biological properties of cancer which are induced by various gene and protein interactions in cancer cells, which result in differences in drug response.
Gene mutations in cancer cells not only affect the function of the affected gene, but also other genes that interact with the mutated gene and proteins. As a consequence, one mutation could lead to changes in the dynamical properties of the molecular network. Therefore, the responses to anti-cancer drugs by cancer cells differ. The current treatment approach that ignores molecular network dynamics and targets a few cancer-related genes is only effective on a fraction of patients, while many other patients exhibit resistance to the drug.
Professor Cho’s team integrated a large-scale computer simulation using super-computing and cellular experiments to analyze changes in molecular network dynamics in cancer cells.
This led to development of technology to find the optimum drug target according to the type of cancer cells by predicting drug response. This technology was applied to the molecular network of known tumor suppressor p53. The team used large-scale cancer cell genomic data available from The Cancer Cell Line Encyclopedia (CCLE) to construct different molecular networks specific to the characteristics of genetic variations.
Perturbation analysis on drug response in each molecular network was used to quantify changes in cancer cells from drug response and similar networks were clustered. Then, computer simulations were used to analyze the synergetic effects in terms of efficacy and combination to predict the level of drug response. Based on the simulation results from various cancer cell lines including lung, breast, bone, skin, kidney, and ovary cancers were used in drug response experiments for compare analysis.
This technique can be applied in any molecular network to identify the optimum drug target for personalized medicine.
The research team suggests that the technology can analyze varying drug response due to the heterogeneity of cancer cells by considering the overall modulatory interactions rather than focusing only on a specific gene or protein. Further, the technology aids the prediction of causes of drug resistance and thus the identification of the optimum drug target to inhibit the resistance. This could be core source technology that can be used in drug repositioning, a process of applying existing drugs to new disease targets.
Professor Cho said, “Genetic variations in cancer cells are the cause of diverse drug response, but a complete analysis had not yet been made.” He continued, “Systems biology allowed the simulation of drug responses by cancer cell molecular networks to identify fundamental principles of drug response and optimum drug targets using a new conceptual approach.”
This research was published in Nature Communications on December 5 and was funded by Ministry of Science and ICT and National Research Foundation of Korea.
(Figure 1. Drug response prediction for each cancer cell type from computer simulation and cellular experiment verification for comparison)
(Figure 2. Drug response prediction based on cancer cell molecular network dynamics and clustering of cancer cells by their molecular networks)
(Figure 3. Identification of drug target for each cancer cell type by cellular molecular network analysis and establishment for personalized medicine strategy for each cancer patient)
2017.12.15 View 7624 -
 CBNU Wins the First AI World Cup 2017
KAIST hosted the first AI World Cup 2017. A total of 26 teams, comprised of Korean undergraduates or graduates, participated in AI Soccer, AI Commentator, and AI Reporter competitions.
On the first of December, the final tournament for AI Soccer was held in the KI Building. More than 200 people came to watch the first AI soccer match based on the Deep-Q Network Learning. The games were commentated by two KAIST students who humorously interpreted the games, catching people’s attention.
In AI Soccer, 18 teams participated, and KAIST BML (KAIST), AIM (KAIST), WISRL (KAIST), and AR Lab (Chonbuk National University) made it to the final four. Each team had its own unique style and strategy.
After some fierce contests, the win went to AR Lab, which showed fearless attacks throughout the game. Having no goalkeeper, AR Lab focused on attacking. According to the team, instead of training individual players, they concentrated on training a manager that directed the players, giving them a choice when to attack and defend.
(AR Lab from Chonbuk National University, the winning team of AI Soccer tournament)
Different from humans playing soccer, it was interesting to see how AI robots self-improved their movements while playing the games.
For AI Commentators, judges looked for a team with the most accuracy, forecasting ability, and fluency. Four teams competed, and the Yally team won the title.
For AI Reporters, which required informativeness and reliability, four teams participated, and the prize went to a team, named Deep CMT (KAIST).
Although this year’s games only included domestic universities, KAIST plans to extend participation to university students from around the world in the future, and hopes that the AI World Cup 2017 will become the foundation for launching the next games on an international level.
2017.12.04 View 6221
CBNU Wins the First AI World Cup 2017
KAIST hosted the first AI World Cup 2017. A total of 26 teams, comprised of Korean undergraduates or graduates, participated in AI Soccer, AI Commentator, and AI Reporter competitions.
On the first of December, the final tournament for AI Soccer was held in the KI Building. More than 200 people came to watch the first AI soccer match based on the Deep-Q Network Learning. The games were commentated by two KAIST students who humorously interpreted the games, catching people’s attention.
In AI Soccer, 18 teams participated, and KAIST BML (KAIST), AIM (KAIST), WISRL (KAIST), and AR Lab (Chonbuk National University) made it to the final four. Each team had its own unique style and strategy.
After some fierce contests, the win went to AR Lab, which showed fearless attacks throughout the game. Having no goalkeeper, AR Lab focused on attacking. According to the team, instead of training individual players, they concentrated on training a manager that directed the players, giving them a choice when to attack and defend.
(AR Lab from Chonbuk National University, the winning team of AI Soccer tournament)
Different from humans playing soccer, it was interesting to see how AI robots self-improved their movements while playing the games.
For AI Commentators, judges looked for a team with the most accuracy, forecasting ability, and fluency. Four teams competed, and the Yally team won the title.
For AI Reporters, which required informativeness and reliability, four teams participated, and the prize went to a team, named Deep CMT (KAIST).
Although this year’s games only included domestic universities, KAIST plans to extend participation to university students from around the world in the future, and hopes that the AI World Cup 2017 will become the foundation for launching the next games on an international level.
2017.12.04 View 6221 -
 Seoul Climate-Energy Conference Seeks Global Sustainability
(President Shin and Former UN Secretary General at the Seoul Climate Change-Energy Conference)
Global leaders from both the private and public sectors discussed creative ways to seek inclusive green growth and sustainable development at the Seoul Climate-Energy Conference on November 24 in Seoul. The annual conference was co-hosted by KAIST and the Coalition for Our Common Future under the theme “Creating New Momentum for the Paris Agreement and a Sustainable Future.”
More than 100 global leaders participated in the forum including the Director General Frank Rijsbermanof the Global Green Growth Institute and Executive Director Howard Bamsey of the Green Climate Fund. Former UN Secretary-General Ban Ki-Moon, who played a significant role in the signing of the Paris Agreement, was the keynote speaker.
This year’s conference focused on Korea’s low carbon-energy transition and the Fourth Industrial Revolution to be aligned with green growth. At the conference, speakers and participants reviewed the progress of the decisions made by the UN Framework Convention on Climate Change (UNFCCC) COP23 in Bonn, Germany. The conference discussed topics of global collaboration for new climate regimes, green energy infrastructure, the Asia super grid, financing green energy, smart green cities, and new mobility.
President Sung-Chul Shin emphasized global action and greater resilience toward climate change in his opening remarks. He said, “Today’s climate change can be attributed directly to the past three industrial revolutions. As industrialization continues, we must not make future generations pay the cost of this Fourth Industrial Revolution.” He explained that it is increasingly complicated to address climate change and energy issues because even though the use of energy consumption will continue to increase, energy policies are interwoven with global politics.
He stressed three keywords to better address this global problem: innovation, collaboration, and speed. First he emphasized innovation as a priority for future success as it is hard to retain confidence without innovation.
He noted KAIST has made sustainability initiatives in the fields of EEWS (energy, environment, water, sustainability) and green mobility. He also noted the importance of collaboration as industries are moving beyond a single discipline. KAIST is making collaborations in R&D and sustainability sectors, such as Saudi Aramco’s CO2 management center in KAIST. Finally, he explained that the speed of new transformation will be beyond our imagination, and governments should work efficiently to address issues in a fast manner.
Meanwhile, Secretary-General Ban called for global unity in addressing climate change. He strongly emphasized that countries should make agreements not of willingness but of action, and that politicians should realize that this global agenda should be given top priority above domestic politics. He addressed how the world is experiencing the most powerful and destructive effects of climate change which makes active participation in the Paris Agreement increasingly important.
He expressed his concern that the richest and most powerful countries are backing off, emphasizing the role of these countries as both global leaders and top producers of CO2. He also shared his hopes that the OECD will continue to work to fill the absence of the United States, and stressed the importance of acquiring 10 billion USD by 2020 to fund mitigation and adaptation technologies for developing countries’ CO2 emissions.
Click for President Shin's opening remarks
2017.11.29 View 9958
Seoul Climate-Energy Conference Seeks Global Sustainability
(President Shin and Former UN Secretary General at the Seoul Climate Change-Energy Conference)
Global leaders from both the private and public sectors discussed creative ways to seek inclusive green growth and sustainable development at the Seoul Climate-Energy Conference on November 24 in Seoul. The annual conference was co-hosted by KAIST and the Coalition for Our Common Future under the theme “Creating New Momentum for the Paris Agreement and a Sustainable Future.”
More than 100 global leaders participated in the forum including the Director General Frank Rijsbermanof the Global Green Growth Institute and Executive Director Howard Bamsey of the Green Climate Fund. Former UN Secretary-General Ban Ki-Moon, who played a significant role in the signing of the Paris Agreement, was the keynote speaker.
This year’s conference focused on Korea’s low carbon-energy transition and the Fourth Industrial Revolution to be aligned with green growth. At the conference, speakers and participants reviewed the progress of the decisions made by the UN Framework Convention on Climate Change (UNFCCC) COP23 in Bonn, Germany. The conference discussed topics of global collaboration for new climate regimes, green energy infrastructure, the Asia super grid, financing green energy, smart green cities, and new mobility.
President Sung-Chul Shin emphasized global action and greater resilience toward climate change in his opening remarks. He said, “Today’s climate change can be attributed directly to the past three industrial revolutions. As industrialization continues, we must not make future generations pay the cost of this Fourth Industrial Revolution.” He explained that it is increasingly complicated to address climate change and energy issues because even though the use of energy consumption will continue to increase, energy policies are interwoven with global politics.
He stressed three keywords to better address this global problem: innovation, collaboration, and speed. First he emphasized innovation as a priority for future success as it is hard to retain confidence without innovation.
He noted KAIST has made sustainability initiatives in the fields of EEWS (energy, environment, water, sustainability) and green mobility. He also noted the importance of collaboration as industries are moving beyond a single discipline. KAIST is making collaborations in R&D and sustainability sectors, such as Saudi Aramco’s CO2 management center in KAIST. Finally, he explained that the speed of new transformation will be beyond our imagination, and governments should work efficiently to address issues in a fast manner.
Meanwhile, Secretary-General Ban called for global unity in addressing climate change. He strongly emphasized that countries should make agreements not of willingness but of action, and that politicians should realize that this global agenda should be given top priority above domestic politics. He addressed how the world is experiencing the most powerful and destructive effects of climate change which makes active participation in the Paris Agreement increasingly important.
He expressed his concern that the richest and most powerful countries are backing off, emphasizing the role of these countries as both global leaders and top producers of CO2. He also shared his hopes that the OECD will continue to work to fill the absence of the United States, and stressed the importance of acquiring 10 billion USD by 2020 to fund mitigation and adaptation technologies for developing countries’ CO2 emissions.
Click for President Shin's opening remarks
2017.11.29 View 9958 -
 Mutant Gene Network in Colon Cancer Identified
The principles of the gene network for colon tumorigenesis have been identified by a KAIST research team. The principles will be used to find the molecular target for effective anti-cancer drugs in the future. Further, this research gained attention for using a systems biology approach, which is an integrated research area of IT and BT.
The KAIST research team led by Professor Kwang-Hyun Cho for the Department of Bio and Brain Engineering succeeded in the identification. Conducted by Dr. Dongkwan Shin and student researchers Jonghoon Lee and Jeong-Ryeol Gong, the research was published in Nature Communications online on November 2.
Human cancer is caused by genetic mutations. The frequency of the mutations differs by the type of cancer; for example, only around 10 mutations are found in leukemia and childhood cancer, but an average of 50 mutations are found in adult solid cancers and even hundreds of mutations are found in cancers due to external factors, such as with lung cancer.
Cancer researchers around the world are working to identify frequently found genetic mutations in patients, and in turn identify important cancer-inducing genes (called ‘driver genes’) to develop targets for anti-cancer drugs. However, gene mutations not only affect their own functions but also affect other genes through interactions. Therefore, there are limitations in current treatments targeting a few cancer-inducing genes without further knowledge on gene networks, hence current drugs are only effective in a few patients and often induce drug resistance.
Professor Cho’s team used large-scale genomic data from cancer patients to construct a mathematical model on the cooperative effects of multiple genetic mutations found in gene interaction networks. The basis of the model construction was The Cancer Genome Atlas (TCGA) presented at the International Cancer Genome Consortium. The team successfully quantified the effects of mutations in gene networks to group colon cancer patients by clinical characteristics.
Further, the critical transition phenomenon that occurs in tumorigenesis was identified using large-scale computer simulation analysis, which was the first hidden gene network principle to be identified. Critical transition is the phenomenon in which the state of matter is suddenly changed through phase transition. It was not possible to identify the presence of transition phenomenon in the past, as it was difficult to track the sequence of gene mutations during tumorigenesis.
The research team used a systems biology-based research method to find that colon cancer tumorigenesis shows a critical transition phenomenon if the known driver gene mutations follow sequentially. Using the developed mathematical model, it can be possible to develop a new anti-cancer targeting drug that most effectively inhibits the effects of many gene mutations found in cancer patients. In particular, not only driver genes, but also other passenger genes affected by the gene mutations, could be evaluated to find the most effective drug targets.
Professor Cho said, “Little was known about the contribution of many gene mutations during tumorigenesis.” He continued, “In this research, a systems biology approach identified the principle of gene networks for the first time to suggest the possibility of anti-cancer drug target identification from a new perspective.”
This research was funded by the Ministry of Science and ICT and the National Research Foundation of Korea.
Figure1. Formation of giant clusters via mutation propagation
Figure2. Critical transition phenomenon by cooperative effect of mutations in tumorigenesis
2017.11.10 View 8693
Mutant Gene Network in Colon Cancer Identified
The principles of the gene network for colon tumorigenesis have been identified by a KAIST research team. The principles will be used to find the molecular target for effective anti-cancer drugs in the future. Further, this research gained attention for using a systems biology approach, which is an integrated research area of IT and BT.
The KAIST research team led by Professor Kwang-Hyun Cho for the Department of Bio and Brain Engineering succeeded in the identification. Conducted by Dr. Dongkwan Shin and student researchers Jonghoon Lee and Jeong-Ryeol Gong, the research was published in Nature Communications online on November 2.
Human cancer is caused by genetic mutations. The frequency of the mutations differs by the type of cancer; for example, only around 10 mutations are found in leukemia and childhood cancer, but an average of 50 mutations are found in adult solid cancers and even hundreds of mutations are found in cancers due to external factors, such as with lung cancer.
Cancer researchers around the world are working to identify frequently found genetic mutations in patients, and in turn identify important cancer-inducing genes (called ‘driver genes’) to develop targets for anti-cancer drugs. However, gene mutations not only affect their own functions but also affect other genes through interactions. Therefore, there are limitations in current treatments targeting a few cancer-inducing genes without further knowledge on gene networks, hence current drugs are only effective in a few patients and often induce drug resistance.
Professor Cho’s team used large-scale genomic data from cancer patients to construct a mathematical model on the cooperative effects of multiple genetic mutations found in gene interaction networks. The basis of the model construction was The Cancer Genome Atlas (TCGA) presented at the International Cancer Genome Consortium. The team successfully quantified the effects of mutations in gene networks to group colon cancer patients by clinical characteristics.
Further, the critical transition phenomenon that occurs in tumorigenesis was identified using large-scale computer simulation analysis, which was the first hidden gene network principle to be identified. Critical transition is the phenomenon in which the state of matter is suddenly changed through phase transition. It was not possible to identify the presence of transition phenomenon in the past, as it was difficult to track the sequence of gene mutations during tumorigenesis.
The research team used a systems biology-based research method to find that colon cancer tumorigenesis shows a critical transition phenomenon if the known driver gene mutations follow sequentially. Using the developed mathematical model, it can be possible to develop a new anti-cancer targeting drug that most effectively inhibits the effects of many gene mutations found in cancer patients. In particular, not only driver genes, but also other passenger genes affected by the gene mutations, could be evaluated to find the most effective drug targets.
Professor Cho said, “Little was known about the contribution of many gene mutations during tumorigenesis.” He continued, “In this research, a systems biology approach identified the principle of gene networks for the first time to suggest the possibility of anti-cancer drug target identification from a new perspective.”
This research was funded by the Ministry of Science and ICT and the National Research Foundation of Korea.
Figure1. Formation of giant clusters via mutation propagation
Figure2. Critical transition phenomenon by cooperative effect of mutations in tumorigenesis
2017.11.10 View 8693 -
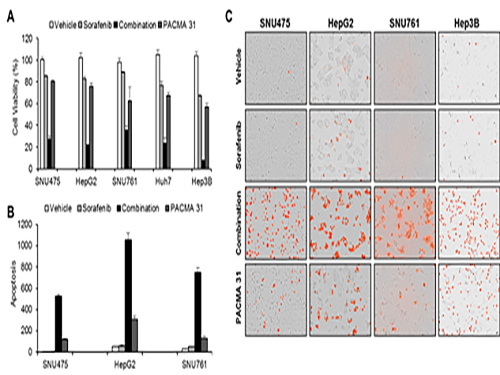 Discovery of an Optimal Drug Combination: Overcoming Resistance to Targeted Drugs for Liver Cancer
A KAIST research team presented a novel method for improving medication treatment for liver cancer using Systems Biology, combining research from information technology and the life sciences. Professor Kwang-Hyun Cho in the Department of Bio and Brain Engineering at KAIST conducted the research in collaboration with Professor Jung-Hwan Yoon in the Department of Internal Medicine at Seoul National University Hospital. This research was published in Hepatology in September 2017 (available online from August 24, 2017).
Liver cancer is the fifth and seventh most common cancer found in men and women throughout the world, which places it second in the cause of cancer deaths. In particular, Korea has 28.4 deaths from liver cancer per 100,000 persons, the highest death rate among OECD countries and twice that of Japan.
Each year in Korea, 16,000 people get liver cancer on average, yet the five-year survival rate stands below 12%. According to the National Cancer Information Center, lung cancer (17,399) took the highest portion of cancer-related deaths, followed by liver cancer (11,311) based on last year data.
Liver cancer is known to carry the highest social cost in comparison to other cancers and it causes the highest fatality in earlier age groups (40s-50s). In that sense, it is necessary to develop a new treatment that mitigates side effects yet elevates the survival rate.
There are ways in which liver cancer can be cured, such as surgery, embolization, and medication treatments; however, the options become limited for curing progressive cancer, a stage in which surgical methods cannot be executed.
Among anticancer medications, Sorafenib, a drug known for enhancing the survival rate of cancer patients, is a unique drug allowed for use as a targeted anticancer medication for progressive liver cancer patients. Its sales reached more than ten billion KRW annually in Korea, but its efficacy works on only about 20% of the treated patients. Also, acquired resistance to Sorafenib is emerging. Additionally, the action mechanism and resistance mechanism of Sorafenib is only vaguely identified.Although Sorafenib only extends the survival rate of terminal cancer patients less than three months on average, it is widely being used because drugs developed by global pharmaceutical companies failed to outperform its effectiveness.
Professor Cho’s research team analyzed the expression changes of genes in cell lines in response to Sorafenib in order to identify the effect and the resistance mechanism of Sorafenib.
As a result, the team discovered the resistance mechanism of Sorafenib using Systems Biology analysis. By combining computer simulations and biological experiments, it was revealed that protein disulfide isomerase (PDI) plays a crucial role in the resistance mechanism of Sorafenib and that its efficacy can be improved significantly by blocking PDI.
The research team used mice in the experiment and discovered the synergic effect of PDI inhibition with Sorafenib for reducing liver cancer cells, known as hepatocellular carcinoma. Also, more PDIs are shown in tissue from patients who possess a resistance to Sorafenib. From these findings, the team could identify the possibility of its clinical applications. The team also confirmed these findings from clinical data through a retrospective cohort study.
“Molecules that play an important role in cell lines are mostly put under complex regulation. For this reason, the existing biological research has a fundamental limitations for discovering its underlying principles,” Professor Cho said. “This research is a representative case of overcoming this limitation of traditional life science research by using a Systems Biology approach, combining IT and life science. It suggests the possibility of developing a new method that overcomes drug resistance with a network analysis of the targeted drug action mechanism of cancer.”
The research was supported by the National Research Foundation of Korea (NRF) and funded by the Ministry of Science and ICT.
(Figure 1. Simulation results from cellular experiments using hepatocellular carcinoma)
(Figure 2. Network analysis and computer simulation by using the endoplasmic reticulum (ER) stress network)
(Figure 3. ER stress network model)
2017.08.30 View 13191
Discovery of an Optimal Drug Combination: Overcoming Resistance to Targeted Drugs for Liver Cancer
A KAIST research team presented a novel method for improving medication treatment for liver cancer using Systems Biology, combining research from information technology and the life sciences. Professor Kwang-Hyun Cho in the Department of Bio and Brain Engineering at KAIST conducted the research in collaboration with Professor Jung-Hwan Yoon in the Department of Internal Medicine at Seoul National University Hospital. This research was published in Hepatology in September 2017 (available online from August 24, 2017).
Liver cancer is the fifth and seventh most common cancer found in men and women throughout the world, which places it second in the cause of cancer deaths. In particular, Korea has 28.4 deaths from liver cancer per 100,000 persons, the highest death rate among OECD countries and twice that of Japan.
Each year in Korea, 16,000 people get liver cancer on average, yet the five-year survival rate stands below 12%. According to the National Cancer Information Center, lung cancer (17,399) took the highest portion of cancer-related deaths, followed by liver cancer (11,311) based on last year data.
Liver cancer is known to carry the highest social cost in comparison to other cancers and it causes the highest fatality in earlier age groups (40s-50s). In that sense, it is necessary to develop a new treatment that mitigates side effects yet elevates the survival rate.
There are ways in which liver cancer can be cured, such as surgery, embolization, and medication treatments; however, the options become limited for curing progressive cancer, a stage in which surgical methods cannot be executed.
Among anticancer medications, Sorafenib, a drug known for enhancing the survival rate of cancer patients, is a unique drug allowed for use as a targeted anticancer medication for progressive liver cancer patients. Its sales reached more than ten billion KRW annually in Korea, but its efficacy works on only about 20% of the treated patients. Also, acquired resistance to Sorafenib is emerging. Additionally, the action mechanism and resistance mechanism of Sorafenib is only vaguely identified.Although Sorafenib only extends the survival rate of terminal cancer patients less than three months on average, it is widely being used because drugs developed by global pharmaceutical companies failed to outperform its effectiveness.
Professor Cho’s research team analyzed the expression changes of genes in cell lines in response to Sorafenib in order to identify the effect and the resistance mechanism of Sorafenib.
As a result, the team discovered the resistance mechanism of Sorafenib using Systems Biology analysis. By combining computer simulations and biological experiments, it was revealed that protein disulfide isomerase (PDI) plays a crucial role in the resistance mechanism of Sorafenib and that its efficacy can be improved significantly by blocking PDI.
The research team used mice in the experiment and discovered the synergic effect of PDI inhibition with Sorafenib for reducing liver cancer cells, known as hepatocellular carcinoma. Also, more PDIs are shown in tissue from patients who possess a resistance to Sorafenib. From these findings, the team could identify the possibility of its clinical applications. The team also confirmed these findings from clinical data through a retrospective cohort study.
“Molecules that play an important role in cell lines are mostly put under complex regulation. For this reason, the existing biological research has a fundamental limitations for discovering its underlying principles,” Professor Cho said. “This research is a representative case of overcoming this limitation of traditional life science research by using a Systems Biology approach, combining IT and life science. It suggests the possibility of developing a new method that overcomes drug resistance with a network analysis of the targeted drug action mechanism of cancer.”
The research was supported by the National Research Foundation of Korea (NRF) and funded by the Ministry of Science and ICT.
(Figure 1. Simulation results from cellular experiments using hepatocellular carcinoma)
(Figure 2. Network analysis and computer simulation by using the endoplasmic reticulum (ER) stress network)
(Figure 3. ER stress network model)
2017.08.30 View 13191 -
 Cooperative Tumor Cell Membrane-Targeted Phototherapy
A KAIST research team led by Professor Ji-Ho Park in the Bio and Brain Engineering Department at KAIST developed a technology for the effective treatment of cancer by delivering synthetic receptors throughout tumor tissue. The study, led by Ph.D. candidate Heegon Kim, was published online in Nature Communications on June 19.
Cancer targeted therapy generally refers to therapy targeting specific molecules that are involved in the growth and generation of cancer. The targeted delivery of therapeutics using targeting agents such as antibodies or nanomaterials has improved the precision and safety of cancer therapy.
However, the paucity and heterogeneity of identified molecular targets within tumors have resulted in poor and uneven distribution of targeted agents, thus compromising treatment outcomes.
To solve this problem, the team constructed a cooperative targeting system in which synthetic and biological nanocomponents participate together in the tumor cell membrane-selective localization of synthetic receptors to amplify the subsequent targeting of therapeutics. Here, synthetic and biological nanocomponents refer to liposomes and extracellular vesicles, respectively.
The synthetic receptors are first delivered selectively to tumor cell membranes in the perivascular region using liposomes. By hitchhiking with extracellular vesicles secreted by the cells, the synthetic receptors are transferred to neighboring cells and further spread throughout the tumor tissues where the molecular targets are limited.
Hitchhiking extracellular vesicles for delivery of synthetic receptors was possible since extracellular vesicles, such as exosomes, mediate intercellular communications by transferring various biological components such as lipids, cytosolic proteins, and RNA through a membrane fusion process. They also play a supportive role in promoting tumor progression in that tumor-derived extracellular vesicles deliver oncogenic signals to normal host cells.
The team showed that this tumor cell membrane-targeted delivery of synthetic receptors led to a uniform distribution of synthetic receptors throughout a tumor and subsequently led to enhanced phototherapeutic efficacy of the targeted photosensitizer.
Professor Park said, “The cooperative tumor targeting system is expected to be applied in treating various diseases that are hard to target.”
The research was funded by the Basic Science Research Program through the National Research Foundation funded by the Ministry of Science, ICT & Future Planning, and the National R&D Program for Cancer Control funded by the Ministry for Health and Welfare.
(Ph.D. candidates Hee Gon Kim (left) and Chanhee Oh)
Figure 1. A schematic of a cooperative tumor targeting system via delivery of synthetic receptors.
Figure 2. A confocal microscopic image of a tumor section after cooperative targeting by synthetic receptor delivery. Green and magenta represent vessels and therapeutic agents inside a tumor respectively.
2017.07.07 View 12216
Cooperative Tumor Cell Membrane-Targeted Phototherapy
A KAIST research team led by Professor Ji-Ho Park in the Bio and Brain Engineering Department at KAIST developed a technology for the effective treatment of cancer by delivering synthetic receptors throughout tumor tissue. The study, led by Ph.D. candidate Heegon Kim, was published online in Nature Communications on June 19.
Cancer targeted therapy generally refers to therapy targeting specific molecules that are involved in the growth and generation of cancer. The targeted delivery of therapeutics using targeting agents such as antibodies or nanomaterials has improved the precision and safety of cancer therapy.
However, the paucity and heterogeneity of identified molecular targets within tumors have resulted in poor and uneven distribution of targeted agents, thus compromising treatment outcomes.
To solve this problem, the team constructed a cooperative targeting system in which synthetic and biological nanocomponents participate together in the tumor cell membrane-selective localization of synthetic receptors to amplify the subsequent targeting of therapeutics. Here, synthetic and biological nanocomponents refer to liposomes and extracellular vesicles, respectively.
The synthetic receptors are first delivered selectively to tumor cell membranes in the perivascular region using liposomes. By hitchhiking with extracellular vesicles secreted by the cells, the synthetic receptors are transferred to neighboring cells and further spread throughout the tumor tissues where the molecular targets are limited.
Hitchhiking extracellular vesicles for delivery of synthetic receptors was possible since extracellular vesicles, such as exosomes, mediate intercellular communications by transferring various biological components such as lipids, cytosolic proteins, and RNA through a membrane fusion process. They also play a supportive role in promoting tumor progression in that tumor-derived extracellular vesicles deliver oncogenic signals to normal host cells.
The team showed that this tumor cell membrane-targeted delivery of synthetic receptors led to a uniform distribution of synthetic receptors throughout a tumor and subsequently led to enhanced phototherapeutic efficacy of the targeted photosensitizer.
Professor Park said, “The cooperative tumor targeting system is expected to be applied in treating various diseases that are hard to target.”
The research was funded by the Basic Science Research Program through the National Research Foundation funded by the Ministry of Science, ICT & Future Planning, and the National R&D Program for Cancer Control funded by the Ministry for Health and Welfare.
(Ph.D. candidates Hee Gon Kim (left) and Chanhee Oh)
Figure 1. A schematic of a cooperative tumor targeting system via delivery of synthetic receptors.
Figure 2. A confocal microscopic image of a tumor section after cooperative targeting by synthetic receptor delivery. Green and magenta represent vessels and therapeutic agents inside a tumor respectively.
2017.07.07 View 12216 -
 The 2017 International Food Festival
The savory smell of exotic dishes filled the main plaza of the KAIST campus on May 26. Exotic music reverberated throughout the campus. The KAIST community took a break together on a breezy early summer Friday afternoon sharing food with their friends and family. KAIST international student body, KISA (KAIST International Students Association), installed white food tents and prepared their country’s favorite dishes at their 13th annual International Food Festival.
The festival was co-organized with Chungnam National University and the University of Science & Technology (UST). At the festival, students from 18 nations cooked about 60 dishes and sold them to the public. Foreign students’ performances of traditional dance and music on the stage livened the atmosphere.
KISA President Sanzhar Kerimbek of the Department of Chemical and Biomolecular Engineering said, “We are so glad to show the diversity of KAIST and its rich culture. This is a big opportunity to get together with neighboring universities, CNU and UST and say thank you for their participation and support." Valentin Porcellini, an exchange students from France in the School of Computing, said, “We are so excited to have people taste our crepes, ratatouille, and other dishes.”
Associate Vice President of the International Office Jay Hyung Lee also said he was glad to see so many people joining this festival. While congratulating the students on the success of the festival, he said the festival will serve as an opportunity to better understand each other by sharing the food and culture.
(Photo caption: Paricipants stop by the Indonesian booth to purchase the food at the International Food Festival on May 26.)
2017.05.29 View 6771
The 2017 International Food Festival
The savory smell of exotic dishes filled the main plaza of the KAIST campus on May 26. Exotic music reverberated throughout the campus. The KAIST community took a break together on a breezy early summer Friday afternoon sharing food with their friends and family. KAIST international student body, KISA (KAIST International Students Association), installed white food tents and prepared their country’s favorite dishes at their 13th annual International Food Festival.
The festival was co-organized with Chungnam National University and the University of Science & Technology (UST). At the festival, students from 18 nations cooked about 60 dishes and sold them to the public. Foreign students’ performances of traditional dance and music on the stage livened the atmosphere.
KISA President Sanzhar Kerimbek of the Department of Chemical and Biomolecular Engineering said, “We are so glad to show the diversity of KAIST and its rich culture. This is a big opportunity to get together with neighboring universities, CNU and UST and say thank you for their participation and support." Valentin Porcellini, an exchange students from France in the School of Computing, said, “We are so excited to have people taste our crepes, ratatouille, and other dishes.”
Associate Vice President of the International Office Jay Hyung Lee also said he was glad to see so many people joining this festival. While congratulating the students on the success of the festival, he said the festival will serve as an opportunity to better understand each other by sharing the food and culture.
(Photo caption: Paricipants stop by the Indonesian booth to purchase the food at the International Food Festival on May 26.)
2017.05.29 View 6771 -
 Gout Diagnostic Strip Using a Single Teardrop
A novel diagnostic strip for gout patients using a single teardrop has been announced by KAIST research team. This technology analyzes biological molecules in tears for a non-invasive diagnosis, significantly reducing the time and expense previously required for a diagnosis.
The research team under Professor Ki-Hun Jeong of the Department of Bio and Brain Engineering succeeded in developing an affordable and elaborate gout diagnostic strip by depositing metal nanoparticles on paper. This technology can not only be used in diagnostic medicine and drug testing, but also in various other areas such as field diagnoses that require prompt and accurate detection of a certain substance.
Gout induces pain in joints due to needle-shaped uric acid crystal build up. In general, therapeutic treatments exist to administer pain relief, stimulate uric acid discharge, and uric acid depressant. Such treatments work for temporary relief, but there have significant limitations. Thus, patients are required to regularly check uric acid concentrations, as well as control their diets. Therefore, simpler ways to measure uric acid would greatly benefit gout control and its prevention in a more affordable and convenient manner.
Existing gout diagnostic techniques include measuring uric acid concentrations from blood samples or observing uric acid crystals from joint synovial fluid under a microscope. These existing methods are invasive and time consuming. To overcome their limitations, the research team uniformly deposited gold nanoislands with nanoplasnomics properties on the surface of paper that can easily collect tears.
Nanoplasnomics techniques collect light on the surface of a metal nanostructure, and can be applied to disease and health diagnostic indicators as well as for genetic material detection. Further, metals such as gold absorb stronger light when it is irradiated, and thus can maximize light concentration on board surfaces while maintaining the properties of paper. The developed metal nanostructure production technology allows the flexible manufacturing of nanostructures on a large surface, which in turn allows flexible control of light concentrations.
The research team grafted surface-enhanced Raman spectroscopy on paper diagnostic strips to allow uric acid concentration measurements in teardrops without additional indicators. The measured concentration in teardrops can be compared to blood uric acid concentrations for diagnosing gout.
Professor Jeong explained, “Based on these research results, our strip will make it possible to conduct low-cost, no indicator, supersensitive biological molecule analysis and fast field diagnosis using tears.” He continued, “Tears, as well as various other bodily fluids, can be used to contribute to disease diagnosis and physiological functional research.”
Ph.D. candidate Moonseong Park participated in the research as the first author of the paper that was published in the online edition of ACS Nano on December 14, 2016. Park said, “The strip will allow fast and simple field diagnosis, and can be produced on a large scale using the existing semiconductor process.”
(Figure 1. Optical image of paper gout diagnostic strip covered with gold)
(Figure 2. Scanning delectron microscopic image of paper gout diagnostic strip)
(Figure 3. Scanning electron microscope image of cellulos fiber coated with gold nanoislands)
(Figure 4. Gout diagnosis using tears)
2017.04.27 View 9710
Gout Diagnostic Strip Using a Single Teardrop
A novel diagnostic strip for gout patients using a single teardrop has been announced by KAIST research team. This technology analyzes biological molecules in tears for a non-invasive diagnosis, significantly reducing the time and expense previously required for a diagnosis.
The research team under Professor Ki-Hun Jeong of the Department of Bio and Brain Engineering succeeded in developing an affordable and elaborate gout diagnostic strip by depositing metal nanoparticles on paper. This technology can not only be used in diagnostic medicine and drug testing, but also in various other areas such as field diagnoses that require prompt and accurate detection of a certain substance.
Gout induces pain in joints due to needle-shaped uric acid crystal build up. In general, therapeutic treatments exist to administer pain relief, stimulate uric acid discharge, and uric acid depressant. Such treatments work for temporary relief, but there have significant limitations. Thus, patients are required to regularly check uric acid concentrations, as well as control their diets. Therefore, simpler ways to measure uric acid would greatly benefit gout control and its prevention in a more affordable and convenient manner.
Existing gout diagnostic techniques include measuring uric acid concentrations from blood samples or observing uric acid crystals from joint synovial fluid under a microscope. These existing methods are invasive and time consuming. To overcome their limitations, the research team uniformly deposited gold nanoislands with nanoplasnomics properties on the surface of paper that can easily collect tears.
Nanoplasnomics techniques collect light on the surface of a metal nanostructure, and can be applied to disease and health diagnostic indicators as well as for genetic material detection. Further, metals such as gold absorb stronger light when it is irradiated, and thus can maximize light concentration on board surfaces while maintaining the properties of paper. The developed metal nanostructure production technology allows the flexible manufacturing of nanostructures on a large surface, which in turn allows flexible control of light concentrations.
The research team grafted surface-enhanced Raman spectroscopy on paper diagnostic strips to allow uric acid concentration measurements in teardrops without additional indicators. The measured concentration in teardrops can be compared to blood uric acid concentrations for diagnosing gout.
Professor Jeong explained, “Based on these research results, our strip will make it possible to conduct low-cost, no indicator, supersensitive biological molecule analysis and fast field diagnosis using tears.” He continued, “Tears, as well as various other bodily fluids, can be used to contribute to disease diagnosis and physiological functional research.”
Ph.D. candidate Moonseong Park participated in the research as the first author of the paper that was published in the online edition of ACS Nano on December 14, 2016. Park said, “The strip will allow fast and simple field diagnosis, and can be produced on a large scale using the existing semiconductor process.”
(Figure 1. Optical image of paper gout diagnostic strip covered with gold)
(Figure 2. Scanning delectron microscopic image of paper gout diagnostic strip)
(Figure 3. Scanning electron microscope image of cellulos fiber coated with gold nanoislands)
(Figure 4. Gout diagnosis using tears)
2017.04.27 View 9710