%6e%65%75%72%61%6c
-
 The World's Smallest Fully Wireless Neural Implant Achieved
< (From left) Sunwoo Lee, KAIST Joint Professor, Alyosha Molnar, Cornell University Professor >
The human brain contains about 100 billion brain cells, and the chemical and electrical signals they exchange create most mental functions. Neural implant technology for precisely reading these signals is essential for the research and treatment of neurodegenerative diseases. A research team from KAIST and international collaborators has successfully implemented a fully wireless, ultra-small implant, which was previously only a theoretical possibility, going beyond simple miniaturization and weight reduction of neural implants.
KAIST announced on the November 27th that a joint research team led by Professor Sunwoo Lee (Joint Professor in Materials Science and Engineering at KAIST and from the School of Electrical and Electronic Engineering at Nanyang Technological University, NTU) and Professor Alyosha Molnar's team from Cornell University in the US has developed 'MOTE (Micro-Scale Opto-Electronic Tetherless Electrode)', an ultra-small wireless neural implant less than 100 micrometers (µm) — smaller than a grain of salt. The team successfully implanted this device into the brains of laboratory mice and stably measured brain waves for one year.
In the brain, invisible, minute electrical signals constantly move, creating our various mental activities such as memory, judgment, and emotion. The technology to directly measure these signals outside the body without connecting wires has been highlighted as key for brain research and the treatment of neurological disorders like dementia and Parkinson's disease.
However, existing implants have limitations: their thick wired structure causes movement in the brain, leading to inflammation and signal degradation over time, and their size and heat generation restrict long-term use.
To overcome these limitations, the research team created an ultra-small circuit based on the existing semiconductor process (CMOS) and combined it with their self-developed ultra-fine Micro-LEDs (µLEDs) to drastically miniaturize the device. They also applied a special surface coating to significantly enhance durability, allowing it to withstand the biological environment for a long time.
The resulting MOTE is less than 100 µm thick and has a volume of less than 1 nanoliter, making it thinner than a human hair and smaller than a grain of salt, the world's smallest level among currently reported wireless neural implants.
Another key feature of MOTE is that it is a fully wireless system that requires no battery. The device is structured to receive external light to generate power, detect brain waves, and then transmit the information back outside embedded in the light signal using Pulse Position Modulation (PPM).
This method drastically reduces energy consumption, minimizes the risk of heat generation, and eliminates the need for battery replacement, enabling long-term use.
The research team conducted a one-year long-term experiment by implanting the ultra-small MOTE into the brains of mice. The results showed normal brain wave measurement over the extended period, with almost no inflammation observed around the implant and no degradation in device performance.
This is considered the first clear demonstration that an ultra-small wireless implant can maintain normal function for a prolonged time inside a living body.
< MOTE neural implant on a salt crystal (left), MOTE neural implants after 296 days of implantation in a laboratory mouse (right) >
Professor Sunwoo Lee stated, "The greatest significance of the newly developed neural implant lies in its actual implementation of a fully wireless, ultra-small implant that was previously only anticipated as a possibility, going beyond simple miniaturization and weight reduction." He added, "This proves the technological possibility of resolving not only the known unknowns raised during the development and use of wireless neural implants, but also the unknown unknowns that newly emerge during the actual development process."
He further added, "This technology will be broadly applicable not only to brain science research but also to nervous system disease monitoring and the development of long-term recording-based treatment technologies."
The research results were published online in the prestigious journal Nature Electronics on November 3rd. ※ Paper Title: A subnanolitre tetherless optoelectronic microsystem for chronic neural recording in awake mice, DOI: https://doi.org/10.1038/s41928-025-01484-1
This research was supported by the US National Institutes of Health (NIH), Nanyang Technological University (Singapore), the Singapore National Research Foundation, the Singapore Ministry of Education, and the ASPIRE League Partnership Seed Fund 2024. The specialized fabrication processes were conducted at the Cornell NanoScale Facility (part of the US National Nanotechnology Coordinated Infrastructure, NNCI) and NTU's Nanyang NanoFabrication Centre.
2025.11.27 View 2502
The World's Smallest Fully Wireless Neural Implant Achieved
< (From left) Sunwoo Lee, KAIST Joint Professor, Alyosha Molnar, Cornell University Professor >
The human brain contains about 100 billion brain cells, and the chemical and electrical signals they exchange create most mental functions. Neural implant technology for precisely reading these signals is essential for the research and treatment of neurodegenerative diseases. A research team from KAIST and international collaborators has successfully implemented a fully wireless, ultra-small implant, which was previously only a theoretical possibility, going beyond simple miniaturization and weight reduction of neural implants.
KAIST announced on the November 27th that a joint research team led by Professor Sunwoo Lee (Joint Professor in Materials Science and Engineering at KAIST and from the School of Electrical and Electronic Engineering at Nanyang Technological University, NTU) and Professor Alyosha Molnar's team from Cornell University in the US has developed 'MOTE (Micro-Scale Opto-Electronic Tetherless Electrode)', an ultra-small wireless neural implant less than 100 micrometers (µm) — smaller than a grain of salt. The team successfully implanted this device into the brains of laboratory mice and stably measured brain waves for one year.
In the brain, invisible, minute electrical signals constantly move, creating our various mental activities such as memory, judgment, and emotion. The technology to directly measure these signals outside the body without connecting wires has been highlighted as key for brain research and the treatment of neurological disorders like dementia and Parkinson's disease.
However, existing implants have limitations: their thick wired structure causes movement in the brain, leading to inflammation and signal degradation over time, and their size and heat generation restrict long-term use.
To overcome these limitations, the research team created an ultra-small circuit based on the existing semiconductor process (CMOS) and combined it with their self-developed ultra-fine Micro-LEDs (µLEDs) to drastically miniaturize the device. They also applied a special surface coating to significantly enhance durability, allowing it to withstand the biological environment for a long time.
The resulting MOTE is less than 100 µm thick and has a volume of less than 1 nanoliter, making it thinner than a human hair and smaller than a grain of salt, the world's smallest level among currently reported wireless neural implants.
Another key feature of MOTE is that it is a fully wireless system that requires no battery. The device is structured to receive external light to generate power, detect brain waves, and then transmit the information back outside embedded in the light signal using Pulse Position Modulation (PPM).
This method drastically reduces energy consumption, minimizes the risk of heat generation, and eliminates the need for battery replacement, enabling long-term use.
The research team conducted a one-year long-term experiment by implanting the ultra-small MOTE into the brains of mice. The results showed normal brain wave measurement over the extended period, with almost no inflammation observed around the implant and no degradation in device performance.
This is considered the first clear demonstration that an ultra-small wireless implant can maintain normal function for a prolonged time inside a living body.
< MOTE neural implant on a salt crystal (left), MOTE neural implants after 296 days of implantation in a laboratory mouse (right) >
Professor Sunwoo Lee stated, "The greatest significance of the newly developed neural implant lies in its actual implementation of a fully wireless, ultra-small implant that was previously only anticipated as a possibility, going beyond simple miniaturization and weight reduction." He added, "This proves the technological possibility of resolving not only the known unknowns raised during the development and use of wireless neural implants, but also the unknown unknowns that newly emerge during the actual development process."
He further added, "This technology will be broadly applicable not only to brain science research but also to nervous system disease monitoring and the development of long-term recording-based treatment technologies."
The research results were published online in the prestigious journal Nature Electronics on November 3rd. ※ Paper Title: A subnanolitre tetherless optoelectronic microsystem for chronic neural recording in awake mice, DOI: https://doi.org/10.1038/s41928-025-01484-1
This research was supported by the US National Institutes of Health (NIH), Nanyang Technological University (Singapore), the Singapore National Research Foundation, the Singapore Ministry of Education, and the ASPIRE League Partnership Seed Fund 2024. The specialized fabrication processes were conducted at the Cornell NanoScale Facility (part of the US National Nanotechnology Coordinated Infrastructure, NNCI) and NTU's Nanyang NanoFabrication Centre.
2025.11.27 View 2502 -
 Thinking outside the box to Fabricate Customized 3D Neural Chips
<(From Left) Professor Yoonkey Nam, Dr. Dongjo Yoon from the Department of Bio and Brain Engineering>
Cultured neural tissues have been widely used as a simplified experimental model for brain research. However, existing devices for growing and recording neural tissues, which are manufactured using semiconductor processes, have limitations in terms of shape modification and the implementation of three-dimensional (3D) structures.
By "thinking outside the box," a KAIST research team has successfully created a customized 3D neural chip. They first used a 3D printer to fabricate a hollow channel structure, then used capillary action to automatically fill the channels with conductive ink, creating the electrodes and wiring. This achievement is expected to significantly increase the design freedom and versatility of brain science and brain engineering research platforms.
On the 25th, KAIST announced that a research team led by Professor Yoonkey Nam from the Department of Bio and Brain Engineering has successfully developed a platform technology that overcomes the limitations of traditional semiconductor-based manufacturing. This technology allows for the precise fabrication of "3D microelectrode array" (neural interfaces with multiple microelectrodes arranged in a 3D space to measure and stimulate the electrophysiological signal of neurons) in various customized forms for in vitro culture chips.
Existing 3D microelectrode array fabrication, based on semiconductor processes, has limited 3D design freedom and is expensive. While 3D printing-based fabrication techniques have recently been proposed to overcome these issues, they still have limitations in terms of 3D design freedom for various in vitro neural network structures because they follow the traditional sequence of "conductive material patterning → insulator coating → electrode opening."
The KAIST research team leveraged the excellent 3D design freedom provided by 3D printing technology and its ability to use printed materials as insulators. By reversing the traditional process, they established an innovative method that allows for more flexible design and functional measurement of 3D neuronal network models for in vitro culture.
<Schematic Diagram of an Integrated Cell Culture Substrate-Microelectrode Array Platform for In Vitro Cultured 3D Neural Network Models>
First, they used a 3D printer to print a hollow 3D insulator with micro-tunnels. This structure was designed to serve as a stable scaffold for conductive materials in 3D space while also supporting the creation of various 3D neuronal networks. They then demonstrated that by using capillary action to fill these internal micro-tunnels with conductive ink, they could create a 3D scaffold-microelectrode array with more freely arranged microelectrodes within a complex 3D culture support structure.
The new platform can be used to create various chip shapes, such as probe-type, cube-type, and modular-type, and supports the fabrication of electrodes using different materials like graphite, conductive polymers, and silver nanoparticles. This allows for the simultaneous measurement of multichannel neural signals from both inside and outside the 3D neuronal network, enabling precise analysis of the dynamic interactions and connectivity between neurons.
Professor Nam stated, "This research, which combines 3D printing and capillary action, is an achievement that significantly expands the freedom of neural chip fabrication." He added that it will contribute to the advancement of fundamental brain science research using neural tissue, as well as applied fields like cell-based biosensors and biocomputing.
Dr. Dongjo Yoon from KAIST's Department of Bio and Brain Engineering participated as the first author of the study. The research findings were published online in the international academic journal Advanced Functional Materials (June 25th issue).
※Paper Title: Highly Customizable Scaffold-Type 3D Microelectrode Array Platform for Design and Analysis of the 3D Neuronal Network In Vitro
This research was supported by the Consolidator Grants Program and the Global Basic Research Laboratory Program of the National Research Foundation of Korea.
2025.09.26 View 2684
Thinking outside the box to Fabricate Customized 3D Neural Chips
<(From Left) Professor Yoonkey Nam, Dr. Dongjo Yoon from the Department of Bio and Brain Engineering>
Cultured neural tissues have been widely used as a simplified experimental model for brain research. However, existing devices for growing and recording neural tissues, which are manufactured using semiconductor processes, have limitations in terms of shape modification and the implementation of three-dimensional (3D) structures.
By "thinking outside the box," a KAIST research team has successfully created a customized 3D neural chip. They first used a 3D printer to fabricate a hollow channel structure, then used capillary action to automatically fill the channels with conductive ink, creating the electrodes and wiring. This achievement is expected to significantly increase the design freedom and versatility of brain science and brain engineering research platforms.
On the 25th, KAIST announced that a research team led by Professor Yoonkey Nam from the Department of Bio and Brain Engineering has successfully developed a platform technology that overcomes the limitations of traditional semiconductor-based manufacturing. This technology allows for the precise fabrication of "3D microelectrode array" (neural interfaces with multiple microelectrodes arranged in a 3D space to measure and stimulate the electrophysiological signal of neurons) in various customized forms for in vitro culture chips.
Existing 3D microelectrode array fabrication, based on semiconductor processes, has limited 3D design freedom and is expensive. While 3D printing-based fabrication techniques have recently been proposed to overcome these issues, they still have limitations in terms of 3D design freedom for various in vitro neural network structures because they follow the traditional sequence of "conductive material patterning → insulator coating → electrode opening."
The KAIST research team leveraged the excellent 3D design freedom provided by 3D printing technology and its ability to use printed materials as insulators. By reversing the traditional process, they established an innovative method that allows for more flexible design and functional measurement of 3D neuronal network models for in vitro culture.
<Schematic Diagram of an Integrated Cell Culture Substrate-Microelectrode Array Platform for In Vitro Cultured 3D Neural Network Models>
First, they used a 3D printer to print a hollow 3D insulator with micro-tunnels. This structure was designed to serve as a stable scaffold for conductive materials in 3D space while also supporting the creation of various 3D neuronal networks. They then demonstrated that by using capillary action to fill these internal micro-tunnels with conductive ink, they could create a 3D scaffold-microelectrode array with more freely arranged microelectrodes within a complex 3D culture support structure.
The new platform can be used to create various chip shapes, such as probe-type, cube-type, and modular-type, and supports the fabrication of electrodes using different materials like graphite, conductive polymers, and silver nanoparticles. This allows for the simultaneous measurement of multichannel neural signals from both inside and outside the 3D neuronal network, enabling precise analysis of the dynamic interactions and connectivity between neurons.
Professor Nam stated, "This research, which combines 3D printing and capillary action, is an achievement that significantly expands the freedom of neural chip fabrication." He added that it will contribute to the advancement of fundamental brain science research using neural tissue, as well as applied fields like cell-based biosensors and biocomputing.
Dr. Dongjo Yoon from KAIST's Department of Bio and Brain Engineering participated as the first author of the study. The research findings were published online in the international academic journal Advanced Functional Materials (June 25th issue).
※Paper Title: Highly Customizable Scaffold-Type 3D Microelectrode Array Platform for Design and Analysis of the 3D Neuronal Network In Vitro
This research was supported by the Consolidator Grants Program and the Global Basic Research Laboratory Program of the National Research Foundation of Korea.
2025.09.26 View 2684 -
 KAIST Successfully Implements 3D Brain-Mimicking Platform with 6x Higher Precision
<(From left) Dr. Dongjo Yoon, Professor Je-Kyun Park from the Department of Bio and Brain Engineering, (upper right) Professor Yoonkey Nam, Dr. Soo Jee Kim>
Existing three-dimensional (3D) neuronal culture technology has limitations in brain research due to the difficulty of precisely replicating the brain's complex multilayered structure and the lack of a platform that can simultaneously analyze both structure and function. A KAIST research team has successfully developed an integrated platform that can implement brain-like layered neuronal structures using 3D printing technology and precisely measure neuronal activity within them.
KAIST (President Kwang Hyung Lee) announced on the 16th of July that a joint research team led by Professors Je-Kyun Park and Yoonkey Nam from the Department of Bio and Brain Engineering has developed an integrated platform capable of fabricating high-resolution 3D multilayer neuronal networks using low-viscosity natural hydrogels with mechanical properties similar to brain tissue, and simultaneously analyzing their structural and functional connectivity.
Conventional bioprinting technology uses high-viscosity bioinks for structural stability, but this limits neuronal proliferation and neurite growth. Conversely, neural cell-friendly low-viscosity hydrogels are difficult to precisely pattern, leading to a fundamental trade-off between structural stability and biological function. The research team completed a sophisticated and stable brain-mimicking platform by combining three key technologies that enable the precise creation of brain structure with dilute gels, accurate alignment between layers, and simultaneous observation of neuronal activity.
The three core technologies are: ▲ 'Capillary Pinning Effect' technology, which enables the dilute gel (hydrogel) to adhere firmly to a stainless steel mesh (micromesh) to prevent it from flowing, thereby reproducing brain structures with six times greater precision (resolution of 500 μm or less) than conventional methods; ▲ the '3D Printing Aligner,' a cylindrical design that ensures the printed layers are precisely stacked without misalignment, guaranteeing the accurate assembly of multilayer structures and stable integration with microelectrode chips; and ▲ 'Dual-mode Analysis System' technology, which simultaneously measures electrical signals from below and observes cell activity with light (calcium imaging) from above, allowing for the simultaneous verification of the functional operation of interlayer connections through multiple methods.
< Figure 1. Platform integrating brain-structure-mimicking neural network model construction and functional measurement technology>
The research team successfully implemented a three-layered mini-brain structure using 3D printing with a fibrin hydrogel, which has elastic properties similar to those of the brain, and experimentally verified the process of actual neural cells transmitting and receiving signals within it.
Cortical neurons were placed in the upper and lower layers, while the middle layer was left empty but designed to allow neurons to penetrate and connect through it. Electrical signals were measured from the lower layer using a microsensor (electrode chip), and cell activity was observed from the upper layer using light (calcium imaging). The results showed that when electrical stimulation was applied, neural cells in both upper and lower layers responded simultaneously. When a synapse-blocking agent (synaptic blocker) was introduced, the response decreased, proving that the neural cells were genuinely connected and transmitting signals.
Professor Je-Kyun Park of KAIST explained, "This research is a joint development achievement of an integrated platform that can simultaneously reproduce the complex multilayered structure and function of brain tissue. Compared to existing technologies where signal measurement was impossible for more than 14 days, this platform maintains a stable microelectrode chip interface for over 27 days, allowing the real-time analysis of structure-function relationships. It can be utilized in various brain research fields such as neurological disease modeling, brain function research, neurotoxicity assessment, and neuroprotective drug screening in the future."
The research, in which Dr. Soo Jee Kim and Dr. Dongjo Yoon from KAIST's Department of Bio and Brain Engineering participated as co-first authors, was published online in the international journal 'Biosensors and Bioelectronics' on June 11, 2025.
※Paper: Hybrid biofabrication of multilayered 3D neuronal networks with structural and functional interlayer connectivity
※DOI: https://doi.org/10.1016/j.bios.2025.117688
2025.07.16 View 4376
KAIST Successfully Implements 3D Brain-Mimicking Platform with 6x Higher Precision
<(From left) Dr. Dongjo Yoon, Professor Je-Kyun Park from the Department of Bio and Brain Engineering, (upper right) Professor Yoonkey Nam, Dr. Soo Jee Kim>
Existing three-dimensional (3D) neuronal culture technology has limitations in brain research due to the difficulty of precisely replicating the brain's complex multilayered structure and the lack of a platform that can simultaneously analyze both structure and function. A KAIST research team has successfully developed an integrated platform that can implement brain-like layered neuronal structures using 3D printing technology and precisely measure neuronal activity within them.
KAIST (President Kwang Hyung Lee) announced on the 16th of July that a joint research team led by Professors Je-Kyun Park and Yoonkey Nam from the Department of Bio and Brain Engineering has developed an integrated platform capable of fabricating high-resolution 3D multilayer neuronal networks using low-viscosity natural hydrogels with mechanical properties similar to brain tissue, and simultaneously analyzing their structural and functional connectivity.
Conventional bioprinting technology uses high-viscosity bioinks for structural stability, but this limits neuronal proliferation and neurite growth. Conversely, neural cell-friendly low-viscosity hydrogels are difficult to precisely pattern, leading to a fundamental trade-off between structural stability and biological function. The research team completed a sophisticated and stable brain-mimicking platform by combining three key technologies that enable the precise creation of brain structure with dilute gels, accurate alignment between layers, and simultaneous observation of neuronal activity.
The three core technologies are: ▲ 'Capillary Pinning Effect' technology, which enables the dilute gel (hydrogel) to adhere firmly to a stainless steel mesh (micromesh) to prevent it from flowing, thereby reproducing brain structures with six times greater precision (resolution of 500 μm or less) than conventional methods; ▲ the '3D Printing Aligner,' a cylindrical design that ensures the printed layers are precisely stacked without misalignment, guaranteeing the accurate assembly of multilayer structures and stable integration with microelectrode chips; and ▲ 'Dual-mode Analysis System' technology, which simultaneously measures electrical signals from below and observes cell activity with light (calcium imaging) from above, allowing for the simultaneous verification of the functional operation of interlayer connections through multiple methods.
< Figure 1. Platform integrating brain-structure-mimicking neural network model construction and functional measurement technology>
The research team successfully implemented a three-layered mini-brain structure using 3D printing with a fibrin hydrogel, which has elastic properties similar to those of the brain, and experimentally verified the process of actual neural cells transmitting and receiving signals within it.
Cortical neurons were placed in the upper and lower layers, while the middle layer was left empty but designed to allow neurons to penetrate and connect through it. Electrical signals were measured from the lower layer using a microsensor (electrode chip), and cell activity was observed from the upper layer using light (calcium imaging). The results showed that when electrical stimulation was applied, neural cells in both upper and lower layers responded simultaneously. When a synapse-blocking agent (synaptic blocker) was introduced, the response decreased, proving that the neural cells were genuinely connected and transmitting signals.
Professor Je-Kyun Park of KAIST explained, "This research is a joint development achievement of an integrated platform that can simultaneously reproduce the complex multilayered structure and function of brain tissue. Compared to existing technologies where signal measurement was impossible for more than 14 days, this platform maintains a stable microelectrode chip interface for over 27 days, allowing the real-time analysis of structure-function relationships. It can be utilized in various brain research fields such as neurological disease modeling, brain function research, neurotoxicity assessment, and neuroprotective drug screening in the future."
The research, in which Dr. Soo Jee Kim and Dr. Dongjo Yoon from KAIST's Department of Bio and Brain Engineering participated as co-first authors, was published online in the international journal 'Biosensors and Bioelectronics' on June 11, 2025.
※Paper: Hybrid biofabrication of multilayered 3D neuronal networks with structural and functional interlayer connectivity
※DOI: https://doi.org/10.1016/j.bios.2025.117688
2025.07.16 View 4376 -
 KAIST Presents Game-Changing Technology for Intractable Brain Disease Treatment Using Micro OLEDs
<(From left)Professor Kyung Cheol Choi, Professor Hyunjoo J. Lee, Dr. Somin Lee from the School of Electrical Engineering>
Optogenetics is a technique that controls neural activity by stimulating neurons expressing light-sensitive proteins with specific wavelengths of light. It has opened new possibilities for identifying causes of brain disorders and developing treatments for intractable neurological diseases. Because this technology requires precise stimulation inside the human brain with minimal damage to soft brain tissue, it must be integrated into a neural probe—a medical device implanted in the brain. KAIST researchers have now proposed a new paradigm for neural probes by integrating micro OLEDs into thin, flexible, implantable medical devices.
KAIST (President Kwang Hyung Lee) announced on the 6th of July that professor Kyung Cheol Choi and professor Hyunjoo J. Lee from the School of Electrical Engineering have jointly succeeded in developing an optogenetic neural probe integrated with flexible micro OLEDs.
Optical fibers have been used for decades in optogenetic research to deliver light to deep brain regions from external light sources. Recently, research has focused on flexible optical fibers and ultra-miniaturized neural probes that integrate light sources for single-neuron stimulation.
The research team focused on micro OLEDs due to their high spatial resolution and flexibility, which allow for precise light delivery to small areas of neurons. This enables detailed brain circuit analysis while minimizing side effects and avoiding restrictions on animal movement. Moreover, micro OLEDs offer precise control of light wavelengths and support multi-site stimulation, making them suitable for studying complex brain functions.
However, the device's electrical properties degrade easily in the presence of moisture or water, which limited their use as implantable bioelectronics. Furthermore, optimizing the high-resolution integration process on thin, flexible probes remained a challenge.
To address this, the team enhanced the operational reliability of OLEDs in moist, oxygen-rich environments and minimized tissue damage during implantation. They patterned an ultrathin, flexible encapsulation layer* composed of aluminum oxide and parylene-C (Al₂O₃/parylene-C) at widths of 260–600 micrometers (μm) to maintain biocompatibility.
*Encapsulation layer: A barrier that completely blocks oxygen and water molecules from the external environment, ensuring the longevity and reliability of the device.
When integrating the high-resolution micro OLEDs, the researchers also used parylene-C, the same biocompatible material as the encapsulation layer, to maintain flexibility and safety. To eliminate electrical interference between adjacent OLED pixels and spatially separate them, they introduced a pixel define layer (PDL), enabling the independent operation of eight micro OLEDs.
Furthermore, they precisely controlled the residual stress and thickness in the multilayer film structure of the device, ensuring its flexibility even in biological environments. This optimization allowed for probe insertion without bending or external shuttles or needles, minimizing mechanical stress during implantation.
2025.07.07 View 5545
KAIST Presents Game-Changing Technology for Intractable Brain Disease Treatment Using Micro OLEDs
<(From left)Professor Kyung Cheol Choi, Professor Hyunjoo J. Lee, Dr. Somin Lee from the School of Electrical Engineering>
Optogenetics is a technique that controls neural activity by stimulating neurons expressing light-sensitive proteins with specific wavelengths of light. It has opened new possibilities for identifying causes of brain disorders and developing treatments for intractable neurological diseases. Because this technology requires precise stimulation inside the human brain with minimal damage to soft brain tissue, it must be integrated into a neural probe—a medical device implanted in the brain. KAIST researchers have now proposed a new paradigm for neural probes by integrating micro OLEDs into thin, flexible, implantable medical devices.
KAIST (President Kwang Hyung Lee) announced on the 6th of July that professor Kyung Cheol Choi and professor Hyunjoo J. Lee from the School of Electrical Engineering have jointly succeeded in developing an optogenetic neural probe integrated with flexible micro OLEDs.
Optical fibers have been used for decades in optogenetic research to deliver light to deep brain regions from external light sources. Recently, research has focused on flexible optical fibers and ultra-miniaturized neural probes that integrate light sources for single-neuron stimulation.
The research team focused on micro OLEDs due to their high spatial resolution and flexibility, which allow for precise light delivery to small areas of neurons. This enables detailed brain circuit analysis while minimizing side effects and avoiding restrictions on animal movement. Moreover, micro OLEDs offer precise control of light wavelengths and support multi-site stimulation, making them suitable for studying complex brain functions.
However, the device's electrical properties degrade easily in the presence of moisture or water, which limited their use as implantable bioelectronics. Furthermore, optimizing the high-resolution integration process on thin, flexible probes remained a challenge.
To address this, the team enhanced the operational reliability of OLEDs in moist, oxygen-rich environments and minimized tissue damage during implantation. They patterned an ultrathin, flexible encapsulation layer* composed of aluminum oxide and parylene-C (Al₂O₃/parylene-C) at widths of 260–600 micrometers (μm) to maintain biocompatibility.
*Encapsulation layer: A barrier that completely blocks oxygen and water molecules from the external environment, ensuring the longevity and reliability of the device.
When integrating the high-resolution micro OLEDs, the researchers also used parylene-C, the same biocompatible material as the encapsulation layer, to maintain flexibility and safety. To eliminate electrical interference between adjacent OLED pixels and spatially separate them, they introduced a pixel define layer (PDL), enabling the independent operation of eight micro OLEDs.
Furthermore, they precisely controlled the residual stress and thickness in the multilayer film structure of the device, ensuring its flexibility even in biological environments. This optimization allowed for probe insertion without bending or external shuttles or needles, minimizing mechanical stress during implantation.
2025.07.07 View 5545 -
 KAIST Accelerates Synthetic Microbe Design by Discovering Novel Enzymes Using AI
< (From left) Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering (top), Hongkeun Ji, PhD candidate of the Department of Chemical and Biomolecular Engineering (top), Ha Rim Kim, PhD candidate of the Department of Chemical and Biomolecular Engineering, and Dr. Gi Bae Kim of the BioProcess Engineering Research Center >
Enzymes are proteins that catalyze biochemical reactions within cells and play a pivotal role in metabolic processes. Accordingly, identifying the functions of novel enzymes is a critical task in the construction of microbial cell factories.
A KAIST research team has leveraged artificial intelligence (AI) to design novel enzymes that do not exist in nature, significantly accelerating microbial cell factory development and boosting the potential for next-generation biotechnological applications such as drug development and biofuel production.
KAIST (represented by President Kwang-Hyung Lee) announced on the 21st of April that Distinguished Professor Sang Yup Lee and his team from the Department of Chemical and Biomolecular Engineering have published a review titled “Enzyme Functional Classification Using Artificial Intelligence,” which outlines the advancement of AI-based enzyme function prediction technologies and analyzes how AI has contributed to the discovery and design of new enzymes.
Professor Lee’s team systematically reviewed the development of enzyme function prediction technologies utilizing machine learning and deep learning, offering a comprehensive analysis.
From sequence similarity-based prediction methods to the integration of convolutional neural networks (CNNs), recurrent neural networks (RNNs), graph neural networks (GNNs), and transformer-based large language models, the paper covers a broad range of AI applications. It analyzes how these technologies extract meaningful information from protein sequences and enhance prediction accuracy.
In particular, enzyme function prediction using deep learning goes beyond simple sequence similarity analysis. By automatically extracting structural and evolutionary features embedded in amino acid sequences, deep learning enables more precise predictions of catalytic functions.
This highlights the unique advantages of AI models compared to traditional bioinformatics approaches.
Moreover, the review suggests that the advancement of generative AI will move future research beyond predicting existing functions to generating entirely new enzymes with functions not found in nature. This shift is expected to profoundly impact the trajectory of biotechnology and synthetic biology.
< Figure 1. Extraction of enzyme characteristics and function prediction using various deep learning structures >
Ha Rim Kim, a Ph.D. candidate and co-first author from the Department of Chemical and Biomolecular Engineering, stated, “AI-based enzyme function prediction and enzyme design are highly important across various fields including metabolic engineering, synthetic biology, and healthcare.”
Distinguished Professor Sang Yup Lee added, “AI-powered enzyme function prediction shows the potential to solve diverse biological problems and will significantly contribute to accelerating research across the entire field.”
The review was published on March 28 in Trends in Biotechnology, a leading biotechnology journal issued by Cell Press.
※ Title: Enzyme Functional Classification Using Artificial Intelligence
※DOI: https://doi.org/10.1016/j.tibtech.2025.03.003
※ Author Information: Ha Rim Kim (KAIST, Co-first author), Hongkeun Ji (KAIST, Co-first author), Gi Bae Kim (KAIST, Third author), Sang Yup Lee (KAIST, Corresponding author)
This research was supported by the Ministry of Science and ICT under the project Development of Core Technologies for Advanced Synthetic Biology to Lead the Bio-Manufacturing Industry (aimed at replacing petroleum-based chemicals), and also by joint support from the Ministry of Science and ICT and the Ministry of Health and Welfare for the project Development of Novel Antibiotic Structures Using Deep Learning-Based Synthetic Biology.
2025.04.07 View 10557
KAIST Accelerates Synthetic Microbe Design by Discovering Novel Enzymes Using AI
< (From left) Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering (top), Hongkeun Ji, PhD candidate of the Department of Chemical and Biomolecular Engineering (top), Ha Rim Kim, PhD candidate of the Department of Chemical and Biomolecular Engineering, and Dr. Gi Bae Kim of the BioProcess Engineering Research Center >
Enzymes are proteins that catalyze biochemical reactions within cells and play a pivotal role in metabolic processes. Accordingly, identifying the functions of novel enzymes is a critical task in the construction of microbial cell factories.
A KAIST research team has leveraged artificial intelligence (AI) to design novel enzymes that do not exist in nature, significantly accelerating microbial cell factory development and boosting the potential for next-generation biotechnological applications such as drug development and biofuel production.
KAIST (represented by President Kwang-Hyung Lee) announced on the 21st of April that Distinguished Professor Sang Yup Lee and his team from the Department of Chemical and Biomolecular Engineering have published a review titled “Enzyme Functional Classification Using Artificial Intelligence,” which outlines the advancement of AI-based enzyme function prediction technologies and analyzes how AI has contributed to the discovery and design of new enzymes.
Professor Lee’s team systematically reviewed the development of enzyme function prediction technologies utilizing machine learning and deep learning, offering a comprehensive analysis.
From sequence similarity-based prediction methods to the integration of convolutional neural networks (CNNs), recurrent neural networks (RNNs), graph neural networks (GNNs), and transformer-based large language models, the paper covers a broad range of AI applications. It analyzes how these technologies extract meaningful information from protein sequences and enhance prediction accuracy.
In particular, enzyme function prediction using deep learning goes beyond simple sequence similarity analysis. By automatically extracting structural and evolutionary features embedded in amino acid sequences, deep learning enables more precise predictions of catalytic functions.
This highlights the unique advantages of AI models compared to traditional bioinformatics approaches.
Moreover, the review suggests that the advancement of generative AI will move future research beyond predicting existing functions to generating entirely new enzymes with functions not found in nature. This shift is expected to profoundly impact the trajectory of biotechnology and synthetic biology.
< Figure 1. Extraction of enzyme characteristics and function prediction using various deep learning structures >
Ha Rim Kim, a Ph.D. candidate and co-first author from the Department of Chemical and Biomolecular Engineering, stated, “AI-based enzyme function prediction and enzyme design are highly important across various fields including metabolic engineering, synthetic biology, and healthcare.”
Distinguished Professor Sang Yup Lee added, “AI-powered enzyme function prediction shows the potential to solve diverse biological problems and will significantly contribute to accelerating research across the entire field.”
The review was published on March 28 in Trends in Biotechnology, a leading biotechnology journal issued by Cell Press.
※ Title: Enzyme Functional Classification Using Artificial Intelligence
※DOI: https://doi.org/10.1016/j.tibtech.2025.03.003
※ Author Information: Ha Rim Kim (KAIST, Co-first author), Hongkeun Ji (KAIST, Co-first author), Gi Bae Kim (KAIST, Third author), Sang Yup Lee (KAIST, Corresponding author)
This research was supported by the Ministry of Science and ICT under the project Development of Core Technologies for Advanced Synthetic Biology to Lead the Bio-Manufacturing Industry (aimed at replacing petroleum-based chemicals), and also by joint support from the Ministry of Science and ICT and the Ministry of Health and Welfare for the project Development of Novel Antibiotic Structures Using Deep Learning-Based Synthetic Biology.
2025.04.07 View 10557 -
 KAIST Proposes a New Way to Circumvent a Long-time Frustration in Neural Computing
The human brain begins learning through spontaneous random activities even before it receives sensory information from the external world. The technology developed by the KAIST research team enables much faster and more accurate learning when exposed to actual data by pre-learning random information in a brain-mimicking artificial neural network, and is expected to be a breakthrough in the development of brain-based artificial intelligence and neuromorphic computing technology in the future.
KAIST (President Kwang-Hyung Lee) announced on the 16th of December that Professor Se-Bum Paik 's research team in the Department of Brain Cognitive Sciences solved the weight transport problem*, a long-standing challenge in neural network learning, and through this, explained the principles that enable resource-efficient learning in biological brain neural networks.
*Weight transport problem: This is the biggest obstacle to the development of artificial intelligence that mimics the biological brain. It is the fundamental reason why large-scale memory and computational work are required in the learning of general artificial neural networks, unlike biological brains.
Over the past several decades, the development of artificial intelligence has been based on error backpropagation learning proposed by Geoffery Hinton, who won the Nobel Prize in Physics this year. However, error backpropagation learning was thought to be impossible in biological brains because it requires the unrealistic assumption that individual neurons must know all the connected information across multiple layers in order to calculate the error signal for learning.
< Figure 1. Illustration depicting the method of random noise training and its effects >
This difficult problem, called the weight transport problem, was raised by Francis Crick, who won the Nobel Prize in Physiology or Medicine for the discovery of the structure of DNA, after the error backpropagation learning was proposed by Hinton in 1986. Since then, it has been considered the reason why the operating principles of natural neural networks and artificial neural networks will forever be fundamentally different.
At the borderline of artificial intelligence and neuroscience, researchers including Hinton have continued to attempt to create biologically plausible models that can implement the learning principles of the brain by solving the weight transport problem.
In 2016, a joint research team from Oxford University and DeepMind in the UK first proposed the concept of error backpropagation learning being possible without weight transport, drawing attention from the academic world. However, biologically plausible error backpropagation learning without weight transport was inefficient, with slow learning speeds and low accuracy, making it difficult to apply in reality.
KAIST research team noted that the biological brain begins learning through internal spontaneous random neural activity even before experiencing external sensory experiences. To mimic this, the research team pre-trained a biologically plausible neural network without weight transport with meaningless random information (random noise).
As a result, they showed that the symmetry of the forward and backward neural cell connections of the neural network, which is an essential condition for error backpropagation learning, can be created. In other words, learning without weight transport is possible through random pre-training.
< Figure 2. Illustration depicting the meta-learning effect of random noise training >
The research team revealed that learning random information before learning actual data has the property of meta-learning, which is ‘learning how to learn.’ It was shown that neural networks that pre-learned random noise perform much faster and more accurate learning when exposed to actual data, and can achieve high learning efficiency without weight transport.
< Figure 3. Illustration depicting research on understanding the brain's operating principles through artificial neural networks >
Professor Se-Bum Paik said, “It breaks the conventional understanding of existing machine learning that only data learning is important, and provides a new perspective that focuses on the neuroscience principles of creating appropriate conditions before learning,” and added, “It is significant in that it solves important problems in artificial neural network learning through clues from developmental neuroscience, and at the same time provides insight into the brain’s learning principles through artificial neural network models.”
This study, in which Jeonghwan Cheon, a Master’s candidate of KAIST Department of Brain and Cognitive Sciences participated as the first author and Professor Sang Wan Lee of the same department as a co-author, was presented at the 38th Neural Information Processing Systems (NeurIPS), the world's top artificial intelligence conference, on December 14th in Vancouver, Canada. (Paper title: Pretraining with random noise for fast and robust learning without weight transport)
This study was conducted with the support of the National Research Foundation of Korea's Basic Research Program in Science and Engineering, the Information and Communications Technology Planning and Evaluation Institute's Talent Development Program, and the KAIST Singularity Professor Program.
2024.12.16 View 15538
KAIST Proposes a New Way to Circumvent a Long-time Frustration in Neural Computing
The human brain begins learning through spontaneous random activities even before it receives sensory information from the external world. The technology developed by the KAIST research team enables much faster and more accurate learning when exposed to actual data by pre-learning random information in a brain-mimicking artificial neural network, and is expected to be a breakthrough in the development of brain-based artificial intelligence and neuromorphic computing technology in the future.
KAIST (President Kwang-Hyung Lee) announced on the 16th of December that Professor Se-Bum Paik 's research team in the Department of Brain Cognitive Sciences solved the weight transport problem*, a long-standing challenge in neural network learning, and through this, explained the principles that enable resource-efficient learning in biological brain neural networks.
*Weight transport problem: This is the biggest obstacle to the development of artificial intelligence that mimics the biological brain. It is the fundamental reason why large-scale memory and computational work are required in the learning of general artificial neural networks, unlike biological brains.
Over the past several decades, the development of artificial intelligence has been based on error backpropagation learning proposed by Geoffery Hinton, who won the Nobel Prize in Physics this year. However, error backpropagation learning was thought to be impossible in biological brains because it requires the unrealistic assumption that individual neurons must know all the connected information across multiple layers in order to calculate the error signal for learning.
< Figure 1. Illustration depicting the method of random noise training and its effects >
This difficult problem, called the weight transport problem, was raised by Francis Crick, who won the Nobel Prize in Physiology or Medicine for the discovery of the structure of DNA, after the error backpropagation learning was proposed by Hinton in 1986. Since then, it has been considered the reason why the operating principles of natural neural networks and artificial neural networks will forever be fundamentally different.
At the borderline of artificial intelligence and neuroscience, researchers including Hinton have continued to attempt to create biologically plausible models that can implement the learning principles of the brain by solving the weight transport problem.
In 2016, a joint research team from Oxford University and DeepMind in the UK first proposed the concept of error backpropagation learning being possible without weight transport, drawing attention from the academic world. However, biologically plausible error backpropagation learning without weight transport was inefficient, with slow learning speeds and low accuracy, making it difficult to apply in reality.
KAIST research team noted that the biological brain begins learning through internal spontaneous random neural activity even before experiencing external sensory experiences. To mimic this, the research team pre-trained a biologically plausible neural network without weight transport with meaningless random information (random noise).
As a result, they showed that the symmetry of the forward and backward neural cell connections of the neural network, which is an essential condition for error backpropagation learning, can be created. In other words, learning without weight transport is possible through random pre-training.
< Figure 2. Illustration depicting the meta-learning effect of random noise training >
The research team revealed that learning random information before learning actual data has the property of meta-learning, which is ‘learning how to learn.’ It was shown that neural networks that pre-learned random noise perform much faster and more accurate learning when exposed to actual data, and can achieve high learning efficiency without weight transport.
< Figure 3. Illustration depicting research on understanding the brain's operating principles through artificial neural networks >
Professor Se-Bum Paik said, “It breaks the conventional understanding of existing machine learning that only data learning is important, and provides a new perspective that focuses on the neuroscience principles of creating appropriate conditions before learning,” and added, “It is significant in that it solves important problems in artificial neural network learning through clues from developmental neuroscience, and at the same time provides insight into the brain’s learning principles through artificial neural network models.”
This study, in which Jeonghwan Cheon, a Master’s candidate of KAIST Department of Brain and Cognitive Sciences participated as the first author and Professor Sang Wan Lee of the same department as a co-author, was presented at the 38th Neural Information Processing Systems (NeurIPS), the world's top artificial intelligence conference, on December 14th in Vancouver, Canada. (Paper title: Pretraining with random noise for fast and robust learning without weight transport)
This study was conducted with the support of the National Research Foundation of Korea's Basic Research Program in Science and Engineering, the Information and Communications Technology Planning and Evaluation Institute's Talent Development Program, and the KAIST Singularity Professor Program.
2024.12.16 View 15538 -
 KAIST Proposes AI Training Method that will Drastically Shorten Time for Complex Quantum Mechanical Calculations
- Professor Yong-Hoon Kim's team from the School of Electrical Engineering succeeded for the first time in accelerating quantum mechanical electronic structure calculations using a convolutional neural network (CNN) model
- Presenting an AI learning principle of quantum mechanical 3D chemical bonding information, the work is expected to accelerate the computer-assisted designing of next-generation materials and devices
The close relationship between AI and high-performance scientific computing can be seen in the fact that both the 2024 Nobel Prizes in Physics and Chemistry were awarded to scientists for their AI-related research contributions in their respective fields of study. KAIST researchers succeeded in dramatically reducing the computation time for highly sophisticated quantum mechanical computer simulations by predicting atomic-level chemical bonding information distributed in 3D space using a novel AI approach.
KAIST (President Kwang-Hyung Lee) announced on the 30th of October that Professor Yong-Hoon Kim's team from the School of Electrical Engineering developed a 3D computer vision artificial neural network-based computation methodology that bypasses the complex algorithms required for atomic-level quantum mechanical calculations traditionally performed using supercomputers to derive the properties of materials.
< Figure 1. Various methodologies are utilized in the simulation of materials and materials, such as quantum mechanical calculations at the nanometer (nm) level, classical mechanical force fields at the scale of tens to hundreds of nanometers, continuum dynamics calculations at the macroscopic scale, and calculations that mix simulations at different scales. These simulations are already playing a key role in a wide range of basic research and application development fields in combination with informatics techniques. Recently, there have been active efforts to introduce machine learning techniques to radically accelerate simulations, but research on introducing machine learning techniques to quantum mechanical electronic structure calculations, which form the basis of high-scale simulations, is still insufficient. >
The quantum mechanical density functional theory (DFT) calculations using supercomputers have become an essential and standard tool in a wide range of research and development fields, including advanced materials and drug design, as they allow fast and accurate prediction of material properties.
*Density functional theory (DFT): A representative theory of ab initio (first principles) calculations that calculate quantum mechanical properties from the atomic level.
However, practical DFT calculations require generating 3D electron density and solving quantum mechanical equations through a complex, iterative self-consistent field (SCF)* process that must be repeated tens to hundreds of times. This restricts its application to systems with only a few hundred to a few thousand atoms.
*Self-consistent field (SCF): A scientific computing method widely used to solve complex many-body problems that must be described by a number of interconnected simultaneous differential equations.
Professor Yong-Hoon Kim’s research team questioned whether recent advancements in AI techniques could be used to bypass the SCF process. As a result, they developed the DeepSCF model, which accelerates calculations by learning chemical bonding information distributed in a 3D space using neural network algorithms from the field of computer vision.
< Figure 2. The deepSCF methodology developed in this study provides a way to rapidly accelerate DFT calculations by avoiding the self-consistent field process (orange box) that had to be performed repeatedly in traditional quantum mechanical electronic structure calculations through artificial neural network techniques (green box). The self-consistent field process is a process of predicting the 3D electron density, constructing the corresponding potential, and then solving the quantum mechanical Cohn-Sham equations, repeating tens to hundreds of times. The core idea of the deepSCF methodology is that the residual electron density (δρ), which is the difference between the electron density (ρ) and the sum of the electron densities of the constituent atoms (ρ0), corresponds to chemical bonding information, so the self-consistent field process is replaced with a 3D convolutional neural network model. >
The research team focused on the fact that, according to density functional theory, electron density contains all quantum mechanical information of electrons, and that the residual electron density — the difference between the total electron density and the sum of the electron densities of the constituent atoms — contains chemical bonding information. They used this as the target for machine learning.
They then adopted a dataset of organic molecules with various chemical bonding characteristics, and applied random rotations and deformations to the atomic structures of these molecules to further enhance the model’s accuracy and generalization capabilities. Ultimately, the research team demonstrated the validity and efficiency of the DeepSCF methodology on large, complex systems.
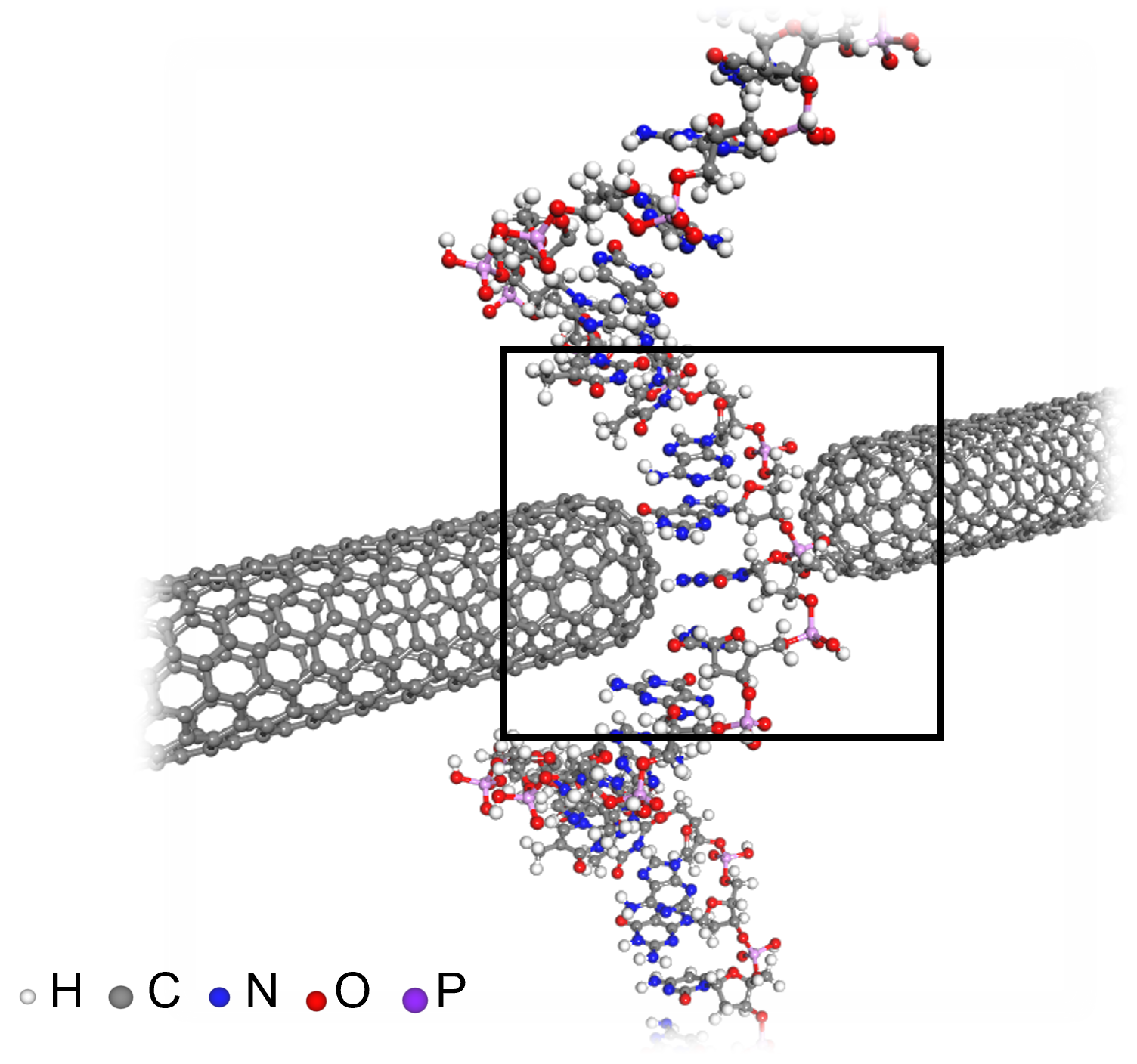
< Figure 3. An example of applying the deepSCF methodology to a carbon nanotube-based DNA sequence analysis device model (top left). In addition to classical mechanical interatomic forces (bottom right), the residual electron density (top right) and quantum mechanical electronic structure properties such as the electronic density of states (DOS) (bottom left) containing information on chemical bonding are rapidly predicted with an accuracy corresponding to the standard DFT calculation results that perform the SCF process. >
Professor Yong-Hoon Kim, who supervised the research, explained that his team had found a way to map quantum mechanical chemical bonding information in a 3D space onto artificial neural networks. He noted, “Since quantum mechanical electron structure calculations underpin materials simulations across all scales, this research establishes a foundational principle for accelerating material calculations using artificial intelligence.”
Ryong-Gyu Lee, a PhD candidate in the School of Electrical Engineering, served as the first author of this research, which was published online on October 24 in Npj Computational Materials, a prestigious journal in the field of material computation. (Paper title: “Convolutional network learning of self-consistent electron density via grid-projected atomic fingerprints”)
This research was conducted with support from the KAIST High-Risk Research Program for Graduate Students and the National Research Foundation of Korea’s Mid-career Researcher Support Program.
2024.10.30 View 12438
KAIST Proposes AI Training Method that will Drastically Shorten Time for Complex Quantum Mechanical Calculations
- Professor Yong-Hoon Kim's team from the School of Electrical Engineering succeeded for the first time in accelerating quantum mechanical electronic structure calculations using a convolutional neural network (CNN) model
- Presenting an AI learning principle of quantum mechanical 3D chemical bonding information, the work is expected to accelerate the computer-assisted designing of next-generation materials and devices
The close relationship between AI and high-performance scientific computing can be seen in the fact that both the 2024 Nobel Prizes in Physics and Chemistry were awarded to scientists for their AI-related research contributions in their respective fields of study. KAIST researchers succeeded in dramatically reducing the computation time for highly sophisticated quantum mechanical computer simulations by predicting atomic-level chemical bonding information distributed in 3D space using a novel AI approach.
KAIST (President Kwang-Hyung Lee) announced on the 30th of October that Professor Yong-Hoon Kim's team from the School of Electrical Engineering developed a 3D computer vision artificial neural network-based computation methodology that bypasses the complex algorithms required for atomic-level quantum mechanical calculations traditionally performed using supercomputers to derive the properties of materials.
< Figure 1. Various methodologies are utilized in the simulation of materials and materials, such as quantum mechanical calculations at the nanometer (nm) level, classical mechanical force fields at the scale of tens to hundreds of nanometers, continuum dynamics calculations at the macroscopic scale, and calculations that mix simulations at different scales. These simulations are already playing a key role in a wide range of basic research and application development fields in combination with informatics techniques. Recently, there have been active efforts to introduce machine learning techniques to radically accelerate simulations, but research on introducing machine learning techniques to quantum mechanical electronic structure calculations, which form the basis of high-scale simulations, is still insufficient. >
The quantum mechanical density functional theory (DFT) calculations using supercomputers have become an essential and standard tool in a wide range of research and development fields, including advanced materials and drug design, as they allow fast and accurate prediction of material properties.
*Density functional theory (DFT): A representative theory of ab initio (first principles) calculations that calculate quantum mechanical properties from the atomic level.
However, practical DFT calculations require generating 3D electron density and solving quantum mechanical equations through a complex, iterative self-consistent field (SCF)* process that must be repeated tens to hundreds of times. This restricts its application to systems with only a few hundred to a few thousand atoms.
*Self-consistent field (SCF): A scientific computing method widely used to solve complex many-body problems that must be described by a number of interconnected simultaneous differential equations.
Professor Yong-Hoon Kim’s research team questioned whether recent advancements in AI techniques could be used to bypass the SCF process. As a result, they developed the DeepSCF model, which accelerates calculations by learning chemical bonding information distributed in a 3D space using neural network algorithms from the field of computer vision.
< Figure 2. The deepSCF methodology developed in this study provides a way to rapidly accelerate DFT calculations by avoiding the self-consistent field process (orange box) that had to be performed repeatedly in traditional quantum mechanical electronic structure calculations through artificial neural network techniques (green box). The self-consistent field process is a process of predicting the 3D electron density, constructing the corresponding potential, and then solving the quantum mechanical Cohn-Sham equations, repeating tens to hundreds of times. The core idea of the deepSCF methodology is that the residual electron density (δρ), which is the difference between the electron density (ρ) and the sum of the electron densities of the constituent atoms (ρ0), corresponds to chemical bonding information, so the self-consistent field process is replaced with a 3D convolutional neural network model. >
The research team focused on the fact that, according to density functional theory, electron density contains all quantum mechanical information of electrons, and that the residual electron density — the difference between the total electron density and the sum of the electron densities of the constituent atoms — contains chemical bonding information. They used this as the target for machine learning.
They then adopted a dataset of organic molecules with various chemical bonding characteristics, and applied random rotations and deformations to the atomic structures of these molecules to further enhance the model’s accuracy and generalization capabilities. Ultimately, the research team demonstrated the validity and efficiency of the DeepSCF methodology on large, complex systems.
< Figure 3. An example of applying the deepSCF methodology to a carbon nanotube-based DNA sequence analysis device model (top left). In addition to classical mechanical interatomic forces (bottom right), the residual electron density (top right) and quantum mechanical electronic structure properties such as the electronic density of states (DOS) (bottom left) containing information on chemical bonding are rapidly predicted with an accuracy corresponding to the standard DFT calculation results that perform the SCF process. >
Professor Yong-Hoon Kim, who supervised the research, explained that his team had found a way to map quantum mechanical chemical bonding information in a 3D space onto artificial neural networks. He noted, “Since quantum mechanical electron structure calculations underpin materials simulations across all scales, this research establishes a foundational principle for accelerating material calculations using artificial intelligence.”
Ryong-Gyu Lee, a PhD candidate in the School of Electrical Engineering, served as the first author of this research, which was published online on October 24 in Npj Computational Materials, a prestigious journal in the field of material computation. (Paper title: “Convolutional network learning of self-consistent electron density via grid-projected atomic fingerprints”)
This research was conducted with support from the KAIST High-Risk Research Program for Graduate Students and the National Research Foundation of Korea’s Mid-career Researcher Support Program.
2024.10.30 View 12438 -
 KAIST Employs Image-recognition AI to Determine Battery Composition and Conditions
An international collaborative research team has developed an image recognition technology that can accurately determine the elemental composition and the number of charge and discharge cycles of a battery by examining only its surface morphology using AI learning.
KAIST (President Kwang-Hyung Lee) announced on July 2nd that Professor Seungbum Hong from the Department of Materials Science and Engineering, in collaboration with the Electronics and Telecommunications Research Institute (ETRI) and Drexel University in the United States, has developed a method to predict the major elemental composition and charge-discharge state of NCM cathode materials with 99.6% accuracy using convolutional neural networks (CNN)*.
*Convolutional Neural Network (CNN): A type of multi-layer, feed-forward, artificial neural network used for analyzing visual images.
The research team noted that while scanning electron microscopy (SEM) is used in semiconductor manufacturing to inspect wafer defects, it is rarely used in battery inspections. SEM is used for batteries to analyze the size of particles only at research sites, and reliability is predicted from the broken particles and the shape of the breakage in the case of deteriorated battery materials.
The research team decided that it would be groundbreaking if an automated SEM can be used in the process of battery production, just like in the semiconductor manufacturing, to inspect the surface of the cathode material to determine whether it was synthesized according to the desired composition and that the lifespan would be reliable, thereby reducing the defect rate.
< Figure 1. Example images of true cases and their grad-CAM overlays from the best trained network. >
The researchers trained a CNN-based AI applicable to autonomous vehicles to learn the surface images of battery materials, enabling it to predict the major elemental composition and charge-discharge cycle states of the cathode materials. They found that while the method could accurately predict the composition of materials with additives, it had lower accuracy for predicting charge-discharge states. The team plans to further train the AI with various battery material morphologies produced through different processes and ultimately use it for inspecting the compositional uniformity and predicting the lifespan of next-generation batteries.
Professor Joshua C. Agar, one of the collaborating researchers of the project from the Department of Mechanical Engineering and Mechanics of Drexel University, said, "In the future, artificial intelligence is expected to be applied not only to battery materials but also to various dynamic processes in functional materials synthesis, clean energy generation in fusion, and understanding foundations of particles and the universe."
Professor Seungbum Hong from KAIST, who led the research, stated, "This research is significant as it is the first in the world to develop an AI-based methodology that can quickly and accurately predict the major elemental composition and the state of the battery from the structural data of micron-scale SEM images. The methodology developed in this study for identifying the composition and state of battery materials based on microscopic images is expected to play a crucial role in improving the performance and quality of battery materials in the future."
< Figure 2. Accuracies of CNN Model predictions on SEM images of NCM cathode materials with additives under various conditions. >
This research was conducted by KAIST’s Materials Science and Engineering Department graduates Dr. Jimin Oh and Dr. Jiwon Yeom, the co-first authors, in collaboration with Professor Josh Agar and Dr. Kwang Man Kim from ETRI. It was supported by the National Research Foundation of Korea, the KAIST Global Singularity project, and international collaboration with the US research team. The results were published in the international journal npj Computational Materials on May 4. (Paper Title: “Composition and state prediction of lithium-ion cathode via convolutional neural network trained on scanning electron microscopy images”)
2024.07.02 View 13025
KAIST Employs Image-recognition AI to Determine Battery Composition and Conditions
An international collaborative research team has developed an image recognition technology that can accurately determine the elemental composition and the number of charge and discharge cycles of a battery by examining only its surface morphology using AI learning.
KAIST (President Kwang-Hyung Lee) announced on July 2nd that Professor Seungbum Hong from the Department of Materials Science and Engineering, in collaboration with the Electronics and Telecommunications Research Institute (ETRI) and Drexel University in the United States, has developed a method to predict the major elemental composition and charge-discharge state of NCM cathode materials with 99.6% accuracy using convolutional neural networks (CNN)*.
*Convolutional Neural Network (CNN): A type of multi-layer, feed-forward, artificial neural network used for analyzing visual images.
The research team noted that while scanning electron microscopy (SEM) is used in semiconductor manufacturing to inspect wafer defects, it is rarely used in battery inspections. SEM is used for batteries to analyze the size of particles only at research sites, and reliability is predicted from the broken particles and the shape of the breakage in the case of deteriorated battery materials.
The research team decided that it would be groundbreaking if an automated SEM can be used in the process of battery production, just like in the semiconductor manufacturing, to inspect the surface of the cathode material to determine whether it was synthesized according to the desired composition and that the lifespan would be reliable, thereby reducing the defect rate.
< Figure 1. Example images of true cases and their grad-CAM overlays from the best trained network. >
The researchers trained a CNN-based AI applicable to autonomous vehicles to learn the surface images of battery materials, enabling it to predict the major elemental composition and charge-discharge cycle states of the cathode materials. They found that while the method could accurately predict the composition of materials with additives, it had lower accuracy for predicting charge-discharge states. The team plans to further train the AI with various battery material morphologies produced through different processes and ultimately use it for inspecting the compositional uniformity and predicting the lifespan of next-generation batteries.
Professor Joshua C. Agar, one of the collaborating researchers of the project from the Department of Mechanical Engineering and Mechanics of Drexel University, said, "In the future, artificial intelligence is expected to be applied not only to battery materials but also to various dynamic processes in functional materials synthesis, clean energy generation in fusion, and understanding foundations of particles and the universe."
Professor Seungbum Hong from KAIST, who led the research, stated, "This research is significant as it is the first in the world to develop an AI-based methodology that can quickly and accurately predict the major elemental composition and the state of the battery from the structural data of micron-scale SEM images. The methodology developed in this study for identifying the composition and state of battery materials based on microscopic images is expected to play a crucial role in improving the performance and quality of battery materials in the future."
< Figure 2. Accuracies of CNN Model predictions on SEM images of NCM cathode materials with additives under various conditions. >
This research was conducted by KAIST’s Materials Science and Engineering Department graduates Dr. Jimin Oh and Dr. Jiwon Yeom, the co-first authors, in collaboration with Professor Josh Agar and Dr. Kwang Man Kim from ETRI. It was supported by the National Research Foundation of Korea, the KAIST Global Singularity project, and international collaboration with the US research team. The results were published in the international journal npj Computational Materials on May 4. (Paper Title: “Composition and state prediction of lithium-ion cathode via convolutional neural network trained on scanning electron microscopy images”)
2024.07.02 View 13025 -
 KAIST Research Team Breaks Down Musical Instincts with AI
Music, often referred to as the universal language, is known to be a common component in all cultures. Then, could ‘musical instinct’ be something that is shared to some degree despite the extensive environmental differences amongst cultures?
On January 16, a KAIST research team led by Professor Hawoong Jung from the Department of Physics announced to have identified the principle by which musical instincts emerge from the human brain without special learning using an artificial neural network model.
Previously, many researchers have attempted to identify the similarities and differences between the music that exist in various different cultures, and tried to understand the origin of the universality. A paper published in Science in 2019 had revealed that music is produced in all ethnographically distinct cultures, and that similar forms of beats and tunes are used. Neuroscientist have also previously found out that a specific part of the human brain, namely the auditory cortex, is responsible for processing musical information.
Professor Jung’s team used an artificial neural network model to show that cognitive functions for music forms spontaneously as a result of processing auditory information received from nature, without being taught music. The research team utilized AudioSet, a large-scale collection of sound data provided by Google, and taught the artificial neural network to learn the various sounds. Interestingly, the research team discovered that certain neurons within the network model would respond selectively to music. In other words, they observed the spontaneous generation of neurons that reacted minimally to various other sounds like those of animals, nature, or machines, but showed high levels of response to various forms of music including both instrumental and vocal.
The neurons in the artificial neural network model showed similar reactive behaviours to those in the auditory cortex of a real brain. For example, artificial neurons responded less to the sound of music that was cropped into short intervals and were rearranged. This indicates that the spontaneously-generated music-selective neurons encode the temporal structure of music. This property was not limited to a specific genre of music, but emerged across 25 different genres including classic, pop, rock, jazz, and electronic.
< Figure 1. Illustration of the musicality of the brain and artificial neural network (created with DALL·E3 AI based on the paper content) >
Furthermore, suppressing the activity of the music-selective neurons was found to greatly impede the cognitive accuracy for other natural sounds. That is to say, the neural function that processes musical information helps process other sounds, and that ‘musical ability’ may be an instinct formed as a result of an evolutionary adaptation acquired to better process sounds from nature.
Professor Hawoong Jung, who advised the research, said, “The results of our study imply that evolutionary pressure has contributed to forming the universal basis for processing musical information in various cultures.” As for the significance of the research, he explained, “We look forward for this artificially built model with human-like musicality to become an original model for various applications including AI music generation, musical therapy, and for research in musical cognition.” He also commented on its limitations, adding, “This research however does not take into consideration the developmental process that follows the learning of music, and it must be noted that this is a study on the foundation of processing musical information in early development.”
< Figure 2. The artificial neural network that learned to recognize non-musical natural sounds in the cyber space distinguishes between music and non-music. >
This research, conducted by first author Dr. Gwangsu Kim of the KAIST Department of Physics (current affiliation: MIT Department of Brain and Cognitive Sciences) and Dr. Dong-Kyum Kim (current affiliation: IBS) was published in Nature Communications under the title, “Spontaneous emergence of rudimentary music detectors in deep neural networks”.
This research was supported by the National Research Foundation of Korea.
2024.01.23 View 13354
KAIST Research Team Breaks Down Musical Instincts with AI
Music, often referred to as the universal language, is known to be a common component in all cultures. Then, could ‘musical instinct’ be something that is shared to some degree despite the extensive environmental differences amongst cultures?
On January 16, a KAIST research team led by Professor Hawoong Jung from the Department of Physics announced to have identified the principle by which musical instincts emerge from the human brain without special learning using an artificial neural network model.
Previously, many researchers have attempted to identify the similarities and differences between the music that exist in various different cultures, and tried to understand the origin of the universality. A paper published in Science in 2019 had revealed that music is produced in all ethnographically distinct cultures, and that similar forms of beats and tunes are used. Neuroscientist have also previously found out that a specific part of the human brain, namely the auditory cortex, is responsible for processing musical information.
Professor Jung’s team used an artificial neural network model to show that cognitive functions for music forms spontaneously as a result of processing auditory information received from nature, without being taught music. The research team utilized AudioSet, a large-scale collection of sound data provided by Google, and taught the artificial neural network to learn the various sounds. Interestingly, the research team discovered that certain neurons within the network model would respond selectively to music. In other words, they observed the spontaneous generation of neurons that reacted minimally to various other sounds like those of animals, nature, or machines, but showed high levels of response to various forms of music including both instrumental and vocal.
The neurons in the artificial neural network model showed similar reactive behaviours to those in the auditory cortex of a real brain. For example, artificial neurons responded less to the sound of music that was cropped into short intervals and were rearranged. This indicates that the spontaneously-generated music-selective neurons encode the temporal structure of music. This property was not limited to a specific genre of music, but emerged across 25 different genres including classic, pop, rock, jazz, and electronic.
< Figure 1. Illustration of the musicality of the brain and artificial neural network (created with DALL·E3 AI based on the paper content) >
Furthermore, suppressing the activity of the music-selective neurons was found to greatly impede the cognitive accuracy for other natural sounds. That is to say, the neural function that processes musical information helps process other sounds, and that ‘musical ability’ may be an instinct formed as a result of an evolutionary adaptation acquired to better process sounds from nature.
Professor Hawoong Jung, who advised the research, said, “The results of our study imply that evolutionary pressure has contributed to forming the universal basis for processing musical information in various cultures.” As for the significance of the research, he explained, “We look forward for this artificially built model with human-like musicality to become an original model for various applications including AI music generation, musical therapy, and for research in musical cognition.” He also commented on its limitations, adding, “This research however does not take into consideration the developmental process that follows the learning of music, and it must be noted that this is a study on the foundation of processing musical information in early development.”
< Figure 2. The artificial neural network that learned to recognize non-musical natural sounds in the cyber space distinguishes between music and non-music. >
This research, conducted by first author Dr. Gwangsu Kim of the KAIST Department of Physics (current affiliation: MIT Department of Brain and Cognitive Sciences) and Dr. Dong-Kyum Kim (current affiliation: IBS) was published in Nature Communications under the title, “Spontaneous emergence of rudimentary music detectors in deep neural networks”.
This research was supported by the National Research Foundation of Korea.
2024.01.23 View 13354 -
 KAIST team develops smart immune system that can pin down on malignant tumors
A joint research team led by Professor Jung Kyoon Choi of the KAIST Department of Bio and Brain Engineering and Professor Jong-Eun Park of the KAIST Graduate School of Medical Science and Engineering (GSMSE) announced the development of the key technologies to treat cancers using smart immune cells designed based on AI and big data analysis. This technology is expected to be a next-generation immunotherapy that allows precision targeting of tumor cells by having the chimeric antigen receptors (CARs) operate through a logical circuit. Professor Hee Jung An of CHA Bundang Medical Center and Professor Hae-Ock Lee of the Catholic University of Korea also participated in this research to contribute joint effort.
Professor Jung Kyoon Choi’s team built a gene expression database from millions of cells, and used this to successfully develop and verify a deep-learning algorithm that could detect the differences in gene expression patterns between tumor cells and normal cells through a logical circuit. CAR immune cells that were fitted with the logic circuits discovered through this methodology could distinguish between tumorous and normal cells as a computer would, and therefore showed potentials to strike only on tumor cells accurately without causing unwanted side effects.
This research, conducted by co-first authors Dr. Joonha Kwon of the KAIST Department of Bio and Brain Engineering and Ph.D. candidate Junho Kang of KAIST GSMSE, was published by Nature Biotechnology on February 16, under the title Single-cell mapping of combinatorial target antigens for CAR switches using logic gates.
An area in cancer research where the most attempts and advances have been made in recent years is immunotherapy. This field of treatment, which utilizes the patient’s own immune system in order to overcome cancer, has several methods including immune checkpoint inhibitors, cancer vaccines and cellular treatments. Immune cells like CAR-T or CAR-NK equipped with chimera antigen receptors, in particular, can recognize cancer antigens and directly destroy cancer cells.
Starting with its success in blood cancer treatment, scientists have been trying to expand the application of CAR cell therapy to treat solid cancer. But there have been difficulties to develop CAR cells with effective killing abilities against solid cancer cells with minimized side effects. Accordingly, in recent years, the development of smarter CAR engineering technologies, i.e., computational logic gates such as AND, OR, and NOT, to effectively target cancer cells has been underway.
At this point in time, the research team built a large-scale database for cancer and normal cells to discover the exact genes that are expressed only from cancer cells at a single-cell level. The team followed this up by developing an AI algorithm that could search for a combination of genes that best distinguishes cancer cells from normal cells. This algorithm, in particular, has been used to find a logic circuit that can specifically target cancer cells through cell-level simulations of all gene combinations. CAR-T cells equipped with logic circuits discovered through this methodology are expected to distinguish cancerous cells from normal cells like computers, thereby minimizing side effects and maximizing the effects of chemotherapy.
Dr. Joonha Kwon, who is the first author of this paper, said, “this research suggests a new method that hasn’t been tried before. What’s particularly noteworthy is the process in which we found the optimal CAR cell circuit through simulations of millions of individual tumors and normal cells.” He added, “This is an innovative technology that can apply AI and computer logic circuits to immune cell engineering. It would contribute greatly to expanding CAR therapy, which is being successfully used for blood cancer, to solid cancers as well.”
This research was funded by the Original Technology Development Project and Research Program for Next Generation Applied Omic of the Korea Research Foundation.
Figure 1. A schematic diagram of manufacturing and administration process of CAR therapy and of cancer cell-specific dual targeting using CAR.
Figure 2. Deep learning (convolutional neural networks, CNNs) algorithm for selection of dual targets based on gene combination (left) and algorithm for calculating expressing cell fractions by gene combination according to logical circuit (right).
2023.03.09 View 18686
KAIST team develops smart immune system that can pin down on malignant tumors
A joint research team led by Professor Jung Kyoon Choi of the KAIST Department of Bio and Brain Engineering and Professor Jong-Eun Park of the KAIST Graduate School of Medical Science and Engineering (GSMSE) announced the development of the key technologies to treat cancers using smart immune cells designed based on AI and big data analysis. This technology is expected to be a next-generation immunotherapy that allows precision targeting of tumor cells by having the chimeric antigen receptors (CARs) operate through a logical circuit. Professor Hee Jung An of CHA Bundang Medical Center and Professor Hae-Ock Lee of the Catholic University of Korea also participated in this research to contribute joint effort.
Professor Jung Kyoon Choi’s team built a gene expression database from millions of cells, and used this to successfully develop and verify a deep-learning algorithm that could detect the differences in gene expression patterns between tumor cells and normal cells through a logical circuit. CAR immune cells that were fitted with the logic circuits discovered through this methodology could distinguish between tumorous and normal cells as a computer would, and therefore showed potentials to strike only on tumor cells accurately without causing unwanted side effects.
This research, conducted by co-first authors Dr. Joonha Kwon of the KAIST Department of Bio and Brain Engineering and Ph.D. candidate Junho Kang of KAIST GSMSE, was published by Nature Biotechnology on February 16, under the title Single-cell mapping of combinatorial target antigens for CAR switches using logic gates.
An area in cancer research where the most attempts and advances have been made in recent years is immunotherapy. This field of treatment, which utilizes the patient’s own immune system in order to overcome cancer, has several methods including immune checkpoint inhibitors, cancer vaccines and cellular treatments. Immune cells like CAR-T or CAR-NK equipped with chimera antigen receptors, in particular, can recognize cancer antigens and directly destroy cancer cells.
Starting with its success in blood cancer treatment, scientists have been trying to expand the application of CAR cell therapy to treat solid cancer. But there have been difficulties to develop CAR cells with effective killing abilities against solid cancer cells with minimized side effects. Accordingly, in recent years, the development of smarter CAR engineering technologies, i.e., computational logic gates such as AND, OR, and NOT, to effectively target cancer cells has been underway.
At this point in time, the research team built a large-scale database for cancer and normal cells to discover the exact genes that are expressed only from cancer cells at a single-cell level. The team followed this up by developing an AI algorithm that could search for a combination of genes that best distinguishes cancer cells from normal cells. This algorithm, in particular, has been used to find a logic circuit that can specifically target cancer cells through cell-level simulations of all gene combinations. CAR-T cells equipped with logic circuits discovered through this methodology are expected to distinguish cancerous cells from normal cells like computers, thereby minimizing side effects and maximizing the effects of chemotherapy.
Dr. Joonha Kwon, who is the first author of this paper, said, “this research suggests a new method that hasn’t been tried before. What’s particularly noteworthy is the process in which we found the optimal CAR cell circuit through simulations of millions of individual tumors and normal cells.” He added, “This is an innovative technology that can apply AI and computer logic circuits to immune cell engineering. It would contribute greatly to expanding CAR therapy, which is being successfully used for blood cancer, to solid cancers as well.”
This research was funded by the Original Technology Development Project and Research Program for Next Generation Applied Omic of the Korea Research Foundation.
Figure 1. A schematic diagram of manufacturing and administration process of CAR therapy and of cancer cell-specific dual targeting using CAR.
Figure 2. Deep learning (convolutional neural networks, CNNs) algorithm for selection of dual targets based on gene combination (left) and algorithm for calculating expressing cell fractions by gene combination according to logical circuit (right).
2023.03.09 View 18686 -
 KAIST researchers discovers the neural circuit that reacts to alarm clock
KAIST (President Kwang Hyung Lee) announced on the 20th that a research team led by Professor Daesoo Kim of the Department of Brain and Cognitive Sciences and Dr. Jeongjin Kim 's team from the Korea Institute of Science and Technology (KIST) have identified the principle of awakening animals by responding to sounds even while sleeping.
Sleep is a very important physiological process that organizes brain activity and maintains health. During sleep, the function of sensory nerves is blocked, so the ability to detect danger in the proximity is reduced. However, many animals detect approaching predators and respond even while sleeping. Scientists thought that animals ready for danger by alternating between deep sleep and light sleep.
A research team led by Professor Daesoo Kim at KAIST discovered that animals have neural circuits that respond to sounds even during deep sleep. While awake, the medial geniculate thalamus responds to sounds, but during deep sleep, or Non-REM sleep, the Mediodorsal thalamus responds to sounds to wake up the brain.
As a result of the study, when the rats fell into deep sleep, the nerves of the medial geniculate thalamus were also sleeping, but the nerves of mediodorsal thalamus were awake and responded immediately to sounds. In addition, it was observed that when mediodorsal thalamus was inhibited, the rats could not wake up even when a sound was heard, and when the mediodorsal thalamus was stimulated, the rats woke up within a few seconds without sound.
This is the first study to show that sleep and wakefulness can transmit auditory signals through different neural circuits, and was reported in the international journal, Current Biology on February 7, and was highlighted by Nature. (https://www.nature.com/articles/d41586-023-00354-0)
Professor Daesoo Kim explained, “The findings of this study can used in developing digital healthcare technologies to be used to improve understanding of disorders of senses and wakefulness seen in various brain diseases and to control the senses in the future.”
This research was carried out with the support from the National Research Foundation of Korea's Mid-Career Research Foundation Program.
Figure 1. Traditionally, sound signals were thought to be propagated from the auditory nerve to the auditory thalamus. However, while in slow-wave sleep, the auditory nerve sends sound signals to the mediodorsal thalamic neurons via the brainstem nerve to induce arousal in the brain.
Figure 2. GRIK4 dorsomedial nerve in response to sound stimulation. The awakening effect is induced as the activity of the GRIK4 dorsal medial nerve increases based on the time when sound stimulation is given.
2023.03.03 View 10436
KAIST researchers discovers the neural circuit that reacts to alarm clock
KAIST (President Kwang Hyung Lee) announced on the 20th that a research team led by Professor Daesoo Kim of the Department of Brain and Cognitive Sciences and Dr. Jeongjin Kim 's team from the Korea Institute of Science and Technology (KIST) have identified the principle of awakening animals by responding to sounds even while sleeping.
Sleep is a very important physiological process that organizes brain activity and maintains health. During sleep, the function of sensory nerves is blocked, so the ability to detect danger in the proximity is reduced. However, many animals detect approaching predators and respond even while sleeping. Scientists thought that animals ready for danger by alternating between deep sleep and light sleep.
A research team led by Professor Daesoo Kim at KAIST discovered that animals have neural circuits that respond to sounds even during deep sleep. While awake, the medial geniculate thalamus responds to sounds, but during deep sleep, or Non-REM sleep, the Mediodorsal thalamus responds to sounds to wake up the brain.
As a result of the study, when the rats fell into deep sleep, the nerves of the medial geniculate thalamus were also sleeping, but the nerves of mediodorsal thalamus were awake and responded immediately to sounds. In addition, it was observed that when mediodorsal thalamus was inhibited, the rats could not wake up even when a sound was heard, and when the mediodorsal thalamus was stimulated, the rats woke up within a few seconds without sound.
This is the first study to show that sleep and wakefulness can transmit auditory signals through different neural circuits, and was reported in the international journal, Current Biology on February 7, and was highlighted by Nature. (https://www.nature.com/articles/d41586-023-00354-0)
Professor Daesoo Kim explained, “The findings of this study can used in developing digital healthcare technologies to be used to improve understanding of disorders of senses and wakefulness seen in various brain diseases and to control the senses in the future.”
This research was carried out with the support from the National Research Foundation of Korea's Mid-Career Research Foundation Program.
Figure 1. Traditionally, sound signals were thought to be propagated from the auditory nerve to the auditory thalamus. However, while in slow-wave sleep, the auditory nerve sends sound signals to the mediodorsal thalamic neurons via the brainstem nerve to induce arousal in the brain.
Figure 2. GRIK4 dorsomedial nerve in response to sound stimulation. The awakening effect is induced as the activity of the GRIK4 dorsal medial nerve increases based on the time when sound stimulation is given.
2023.03.03 View 10436 -
 Professor Jae-Woong Jeong Receives Hyonwoo KAIST Academic Award
Professor Jae-Woong Jeong from the School of Electrical Engineering was selected for the Hyonwoo KAIST Academic Award, funded by the HyonWoo Cultural Foundation (Chairman Soo-il Kwak, honorary professor at Seoul National University Business School).
The Hyonwoo KAIST Academic Award, presented for the first time in 2021, is an award newly founded by the donations of Chairman Soo-il Kwak of the HyonWoo Cultural Foundation, who aims to reward excellent KAIST scholars who have made outstanding academic achievements.
Every year, through the strict evaluations of the selection committee of the HyonWoo Cultural Foundation and the faculty reward recommendation board, KAIST will choose one faculty member that may represent the school with their excellent academic achievement, and reward them with a plaque and 100 million won.
Professor Jae-Woong Jeong, the winner of this year’s award, developed the first IoT-based wireless remote brain neural network control system to overcome brain diseases, and has been leading the field. The research was published in 2021 in Nature Biomedical Engineering, one of world’s best scientific journals, and has been recognized as a novel technology that suggested a new vision for the automation of brain research and disease treatment. This study, led by Professor Jeong’s research team, was part of the KAIST College of Engineering Global Initiative Interdisciplinary Research Project, and was jointly studied by Washington University School of Medicine through an international research collaboration. The technology was introduced more than 60 times through both domestic and international media, including Medical Xpress, MBC News, and Maeil Business News.
Professor Jeong has also developed a wirelessly chargeable soft machine for brain transplants, and the results were published in Nature Communications. He thereby opened a new paradigm for implantable semi-permanent devices for transplants, and is making unprecedented research achievements.
2022.06.13 View 14244
Professor Jae-Woong Jeong Receives Hyonwoo KAIST Academic Award
Professor Jae-Woong Jeong from the School of Electrical Engineering was selected for the Hyonwoo KAIST Academic Award, funded by the HyonWoo Cultural Foundation (Chairman Soo-il Kwak, honorary professor at Seoul National University Business School).
The Hyonwoo KAIST Academic Award, presented for the first time in 2021, is an award newly founded by the donations of Chairman Soo-il Kwak of the HyonWoo Cultural Foundation, who aims to reward excellent KAIST scholars who have made outstanding academic achievements.
Every year, through the strict evaluations of the selection committee of the HyonWoo Cultural Foundation and the faculty reward recommendation board, KAIST will choose one faculty member that may represent the school with their excellent academic achievement, and reward them with a plaque and 100 million won.
Professor Jae-Woong Jeong, the winner of this year’s award, developed the first IoT-based wireless remote brain neural network control system to overcome brain diseases, and has been leading the field. The research was published in 2021 in Nature Biomedical Engineering, one of world’s best scientific journals, and has been recognized as a novel technology that suggested a new vision for the automation of brain research and disease treatment. This study, led by Professor Jeong’s research team, was part of the KAIST College of Engineering Global Initiative Interdisciplinary Research Project, and was jointly studied by Washington University School of Medicine through an international research collaboration. The technology was introduced more than 60 times through both domestic and international media, including Medical Xpress, MBC News, and Maeil Business News.
Professor Jeong has also developed a wirelessly chargeable soft machine for brain transplants, and the results were published in Nature Communications. He thereby opened a new paradigm for implantable semi-permanent devices for transplants, and is making unprecedented research achievements.
2022.06.13 View 14244