%43%65%6c%6c%73
-
 Breakthrough in Intractable Intestinal Disease Treatment Using Xenogeneic-Free Intestinal Stem Cells
< (From left) Professor Sung Gap Im (KAIST), Dr. Seonghyeon Park (KAIST), M.S candidate Sang Yu Sun (KAIST), Dr. Mi-Young Son (KRIBB), (Top right) Dr. Tae Geol Lee (KRISS), Dr. Jin Gyeong Son (KRISS) >
Intestinal Stem Cells (ISCs) derived from a patient's own cells have garnered significant attention as a new alternative for treating intractable intestinal diseases due to their low risk of rejection. However, clinical application has been limited by safety and regulatory issues arising from conventional culture methods that rely on animal-derived components (xenogeneic components). A KAIST research team has developed an advanced culture technology that stably grows ISCs without animal components while simultaneously enhancing their migration to damaged tissues and regenerative capabilities.
KAIST announced on December 23rd that a joint research team—led by Professor Sung Gap Im from the Department of Chemical and Biomolecular Engineering, Dr. Tae Geol Lee from the Nano-Bio Measurement Group at the Korea Research Institute of Standards and Science and Dr. Mi-Young Son from the Stem Cell Convergence Research Center at the Korea Research Institute of Bioscience and Biotechnology has developed a polymer-based culture platform that dramatically improves the migration and regeneration of ISCs in a xenogeneic-free environment.
To overcome obstacles in the clinical application of stem cell therapies—such as the risk of virus transmission to patients when using substances derived from mouse fibroblasts or Matrigel—the joint research team developed "PLUS" (Polymer-coated Ultra-stable Surface). This polymer-based culture surface technology functions effectively without any animal-derived materials.
< Figure 1. Precise control of polymer coating and surface modification via initiated Chemical Vapor Deposition (iCVD) process >
PLUS is a synthetic polymer surface coated via a vapor deposition method. By precisely controlling surface energy and chemical composition, it significantly enhances the adhesion and mass-culture efficiency of ISCs. Notably, it maintains identical culture performance even after being stored at room temperature for three years, securing industrial scalability and storage convenience for stem cell therapeutics.
Through proteomics analysis*, the research team identified that the expression of proteins related to cytoskeletal reorganization significantly increased in ISCs cultured on the PLUS environment.
Proteomics Analysis: A method used to simultaneously analyze the types and quantitative changes of all proteins present within a cell or tissue.
Specifically, the team confirmed that increased expression of cytoskeleton-binding and actin-binding proteins leads to a stable restructuring of the internal cellular architecture. This provides the power source for stem cells to move faster and more actively across the substrate.
< Figure 2. Elucidation of the mechanism for enhanced ISC migration through precision proteomics analysis >
Real-time observations using holotomography microscopy revealed that ISCs cultured on PLUS exhibited a migration speed approximately twice as fast as those on conventional surfaces. Furthermore, in a damaged tissue model, the cells demonstrated outstanding regenerative performance, repairing more than half of the damage within a single week. This proves that PLUS activates the cytoskeletal activity of stem cells, thereby boosting their practical tissue regeneration capabilities.
The newly developed PLUS culture platform is evaluated as a technology that will significantly enhance the safety, mass production, and clinical feasibility of ISCs derived from human pluripotent stem cells (hPSCs). By elucidating the mechanism that simultaneously strengthens the survival, migration, and regeneration of stem cells in a xenogeneic-free environment, the team has established a foundation to fundamentally resolve safety, regulatory, and productivity issues in stem cell therapy.
Professor Sung Gap Im of KAIST stated, "This research provides a synthetic culture platform that eliminates the dependence on xenogeneic components—which has hindered the clinical application of stem cell therapies—while maximizing the migration and regenerative capacity of stem cells. It will serve as a catalyst for a paradigm shift in the field of regenerative medicine."
Dr. Seonghyeon Park (KAIST), Sang Yu Sun (KAIST), and Dr. Jin Gyeong Son (KRISS) participated as first authors. The research findings were published online on November 26th in Advanced Materials, the leading academic journal in materials science.
Paper Title: Tailored Xenogeneic-Free Polymer Surface Promotes Dynamic Migration of Intestinal Stem Cells
DOI: 10.1002/adma.202513371
This research was conducted with support from the Ministry of Science and ICT, the Ministry of SMEs and Startups, the National Research Foundation of Korea, the National Council of Science and Technology Research, KRISS, KRIBB, and the National NanoFab Center.
2025.12.23 View 1730
Breakthrough in Intractable Intestinal Disease Treatment Using Xenogeneic-Free Intestinal Stem Cells
< (From left) Professor Sung Gap Im (KAIST), Dr. Seonghyeon Park (KAIST), M.S candidate Sang Yu Sun (KAIST), Dr. Mi-Young Son (KRIBB), (Top right) Dr. Tae Geol Lee (KRISS), Dr. Jin Gyeong Son (KRISS) >
Intestinal Stem Cells (ISCs) derived from a patient's own cells have garnered significant attention as a new alternative for treating intractable intestinal diseases due to their low risk of rejection. However, clinical application has been limited by safety and regulatory issues arising from conventional culture methods that rely on animal-derived components (xenogeneic components). A KAIST research team has developed an advanced culture technology that stably grows ISCs without animal components while simultaneously enhancing their migration to damaged tissues and regenerative capabilities.
KAIST announced on December 23rd that a joint research team—led by Professor Sung Gap Im from the Department of Chemical and Biomolecular Engineering, Dr. Tae Geol Lee from the Nano-Bio Measurement Group at the Korea Research Institute of Standards and Science and Dr. Mi-Young Son from the Stem Cell Convergence Research Center at the Korea Research Institute of Bioscience and Biotechnology has developed a polymer-based culture platform that dramatically improves the migration and regeneration of ISCs in a xenogeneic-free environment.
To overcome obstacles in the clinical application of stem cell therapies—such as the risk of virus transmission to patients when using substances derived from mouse fibroblasts or Matrigel—the joint research team developed "PLUS" (Polymer-coated Ultra-stable Surface). This polymer-based culture surface technology functions effectively without any animal-derived materials.
< Figure 1. Precise control of polymer coating and surface modification via initiated Chemical Vapor Deposition (iCVD) process >
PLUS is a synthetic polymer surface coated via a vapor deposition method. By precisely controlling surface energy and chemical composition, it significantly enhances the adhesion and mass-culture efficiency of ISCs. Notably, it maintains identical culture performance even after being stored at room temperature for three years, securing industrial scalability and storage convenience for stem cell therapeutics.
Through proteomics analysis*, the research team identified that the expression of proteins related to cytoskeletal reorganization significantly increased in ISCs cultured on the PLUS environment.
Proteomics Analysis: A method used to simultaneously analyze the types and quantitative changes of all proteins present within a cell or tissue.
Specifically, the team confirmed that increased expression of cytoskeleton-binding and actin-binding proteins leads to a stable restructuring of the internal cellular architecture. This provides the power source for stem cells to move faster and more actively across the substrate.
< Figure 2. Elucidation of the mechanism for enhanced ISC migration through precision proteomics analysis >
Real-time observations using holotomography microscopy revealed that ISCs cultured on PLUS exhibited a migration speed approximately twice as fast as those on conventional surfaces. Furthermore, in a damaged tissue model, the cells demonstrated outstanding regenerative performance, repairing more than half of the damage within a single week. This proves that PLUS activates the cytoskeletal activity of stem cells, thereby boosting their practical tissue regeneration capabilities.
The newly developed PLUS culture platform is evaluated as a technology that will significantly enhance the safety, mass production, and clinical feasibility of ISCs derived from human pluripotent stem cells (hPSCs). By elucidating the mechanism that simultaneously strengthens the survival, migration, and regeneration of stem cells in a xenogeneic-free environment, the team has established a foundation to fundamentally resolve safety, regulatory, and productivity issues in stem cell therapy.
Professor Sung Gap Im of KAIST stated, "This research provides a synthetic culture platform that eliminates the dependence on xenogeneic components—which has hindered the clinical application of stem cell therapies—while maximizing the migration and regenerative capacity of stem cells. It will serve as a catalyst for a paradigm shift in the field of regenerative medicine."
Dr. Seonghyeon Park (KAIST), Sang Yu Sun (KAIST), and Dr. Jin Gyeong Son (KRISS) participated as first authors. The research findings were published online on November 26th in Advanced Materials, the leading academic journal in materials science.
Paper Title: Tailored Xenogeneic-Free Polymer Surface Promotes Dynamic Migration of Intestinal Stem Cells
DOI: 10.1002/adma.202513371
This research was conducted with support from the Ministry of Science and ICT, the Ministry of SMEs and Startups, the National Research Foundation of Korea, the National Council of Science and Technology Research, KRISS, KRIBB, and the National NanoFab Center.
2025.12.23 View 1730 -
 KAIST Uncovers the Mechanism Behind Overactive Immune Cells
<(From Right) Professor Eui-Cheol Shin, Ph.D candidate So-Young Kim, Professor Su-Hyung Park, Professor Hyuk Soo Eun, Dr. Hoyoung Lee>
“Why do immune cells that are supposed to eliminate viruses suddenly turn against our own body?”
There are instances where killer T cells—which are meant to precisely remove virus-infected cells—malfunction like overheated engines, attacking even healthy cells and damaging tissues. A KAIST research team has now identified the key mechanism that regulates this excessive activation of killer T cells, offering new insights into controlling immune overreactions and developing therapies for immune-related diseases.
KAIST (President Kwang Hyung Lee) announced on November 5 that a research team led by Professors Eui-Cheol Shin and Su-Hyung Park from the Graduate School of Medical Science and Engineering, in collaboration with Professor Hyuk Soo Eun from Chungnam National University College of Medicine, has uncovered the molecular basis of nonspecific activation in killer T cells and proposed a new therapeutic strategy to control it.
Killer T cells (CD8⁺ T cells) selectively eliminate infected cells to prevent viral spread. However, when excessively activated, they can attack uninfected cells, causing inflammation and tissue damage. Such overactive immune responses can lead to severe viral infections and autoimmune diseases.
In 2018, Professor Shin’s team was the first in the world to discover that killer T cells can be nonspecifically activated by cytokines and randomly attack host cells—a phenomenon they termed “bystander activation of T cells”. The current study builds on that discovery by revealing the molecular mechanism driving this abnormal process.
The team focused on a cytokine called interleukin-15 (IL-15). Experiments showed that IL-15 can abnormally excite killer T cells by a bystander activation mechanism, causing them to attack uninfected host cells. However, when there is a concurrent antigen-specific stimulation, IL-15-induced bystander activation is suppressed.
The researchers further identified that this suppression occurs through an intracellular signaling process. When the concentration of calcium ions (Ca²⁺) changes, a protein called calcineurin activates, which in turn triggers a regulatory protein known as NFAT, suppressing IL-15-induced bystander activation of killer T cells. In other words, the calcineurin–NFAT pathway activated by antigen stimulation acts as a brake on overactivation by a bystander mechanism.
The team also discovered that some immunosuppressants, which are known to block the calcineurin pathway, may not always suppress immune responses—in certain contexts, they can instead promote IL-15-induced bystander activation of killer T cells. This finding underscores that not all immunosuppressants work the same way and that treatments must be carefully tailored to each patient’s immune response.
Through gene expression analysis, the researchers identified a gene set that increase only in abnormally activated killer T cells induced by IL-15 as markers. They further confirmed that these same markers were elevated in bystander killer T cells from patients with acute hepatitis A, suggesting that the markers could be used for disease diagnosis.
<In a normal immune response, killer T cells are activated by antigen stimulation and selectively eliminate only virus-infected cells, thereby controlling viral replication and promoting the patient’s rapid recovery. However, when killer T cells are nonspecifically overactivated by interleukin-15, they may randomly attack normal cells as well, causing excessive tissue damage and leading to severe disease. Future research may identify diseases in which such nonspecific hyperimmune responses occur, making it possible to develop new drugs to control them>
This study provides crucial clues for understanding the pathogenesis of various immune-related diseases, including severe viral infections, chronic inflammatory disorders, autoimmune diseases, and organ transplant rejection. It also paves the way for developing novel immunoregulatory therapies targeting IL-15 signaling.
Professor Eui-Cheol Shin explained that, “this study shows that killer T cells are not merely defenders—they can transform into ‘nonspecific attackers’ depending on the inflammatory environment. By precisely regulating this abnormal activation, we may be able to develop new treatments for intractable immune diseases.”
This research was published in the journal Immunity on October 31, with Dr. Hoyoung Lee and Ph.D. candidate So-Young Kim as co–first authors.
Title: “TCR signaling via NFATc1 constrains IL-15-induced bystander activation of human memory CD8⁺ T cells”, DOI: doi.org/10.1016/j.immuni.2025.10.002
The study was supported by the National Research Foundation of Korea (NRF), the Korea Health Industry Development Institute (KHIDI), and the Institute for Basic Science (IBS).
2025.11.06 View 2921
KAIST Uncovers the Mechanism Behind Overactive Immune Cells
<(From Right) Professor Eui-Cheol Shin, Ph.D candidate So-Young Kim, Professor Su-Hyung Park, Professor Hyuk Soo Eun, Dr. Hoyoung Lee>
“Why do immune cells that are supposed to eliminate viruses suddenly turn against our own body?”
There are instances where killer T cells—which are meant to precisely remove virus-infected cells—malfunction like overheated engines, attacking even healthy cells and damaging tissues. A KAIST research team has now identified the key mechanism that regulates this excessive activation of killer T cells, offering new insights into controlling immune overreactions and developing therapies for immune-related diseases.
KAIST (President Kwang Hyung Lee) announced on November 5 that a research team led by Professors Eui-Cheol Shin and Su-Hyung Park from the Graduate School of Medical Science and Engineering, in collaboration with Professor Hyuk Soo Eun from Chungnam National University College of Medicine, has uncovered the molecular basis of nonspecific activation in killer T cells and proposed a new therapeutic strategy to control it.
Killer T cells (CD8⁺ T cells) selectively eliminate infected cells to prevent viral spread. However, when excessively activated, they can attack uninfected cells, causing inflammation and tissue damage. Such overactive immune responses can lead to severe viral infections and autoimmune diseases.
In 2018, Professor Shin’s team was the first in the world to discover that killer T cells can be nonspecifically activated by cytokines and randomly attack host cells—a phenomenon they termed “bystander activation of T cells”. The current study builds on that discovery by revealing the molecular mechanism driving this abnormal process.
The team focused on a cytokine called interleukin-15 (IL-15). Experiments showed that IL-15 can abnormally excite killer T cells by a bystander activation mechanism, causing them to attack uninfected host cells. However, when there is a concurrent antigen-specific stimulation, IL-15-induced bystander activation is suppressed.
The researchers further identified that this suppression occurs through an intracellular signaling process. When the concentration of calcium ions (Ca²⁺) changes, a protein called calcineurin activates, which in turn triggers a regulatory protein known as NFAT, suppressing IL-15-induced bystander activation of killer T cells. In other words, the calcineurin–NFAT pathway activated by antigen stimulation acts as a brake on overactivation by a bystander mechanism.
The team also discovered that some immunosuppressants, which are known to block the calcineurin pathway, may not always suppress immune responses—in certain contexts, they can instead promote IL-15-induced bystander activation of killer T cells. This finding underscores that not all immunosuppressants work the same way and that treatments must be carefully tailored to each patient’s immune response.
Through gene expression analysis, the researchers identified a gene set that increase only in abnormally activated killer T cells induced by IL-15 as markers. They further confirmed that these same markers were elevated in bystander killer T cells from patients with acute hepatitis A, suggesting that the markers could be used for disease diagnosis.
<In a normal immune response, killer T cells are activated by antigen stimulation and selectively eliminate only virus-infected cells, thereby controlling viral replication and promoting the patient’s rapid recovery. However, when killer T cells are nonspecifically overactivated by interleukin-15, they may randomly attack normal cells as well, causing excessive tissue damage and leading to severe disease. Future research may identify diseases in which such nonspecific hyperimmune responses occur, making it possible to develop new drugs to control them>
This study provides crucial clues for understanding the pathogenesis of various immune-related diseases, including severe viral infections, chronic inflammatory disorders, autoimmune diseases, and organ transplant rejection. It also paves the way for developing novel immunoregulatory therapies targeting IL-15 signaling.
Professor Eui-Cheol Shin explained that, “this study shows that killer T cells are not merely defenders—they can transform into ‘nonspecific attackers’ depending on the inflammatory environment. By precisely regulating this abnormal activation, we may be able to develop new treatments for intractable immune diseases.”
This research was published in the journal Immunity on October 31, with Dr. Hoyoung Lee and Ph.D. candidate So-Young Kim as co–first authors.
Title: “TCR signaling via NFATc1 constrains IL-15-induced bystander activation of human memory CD8⁺ T cells”, DOI: doi.org/10.1016/j.immuni.2025.10.002
The study was supported by the National Research Foundation of Korea (NRF), the Korea Health Industry Development Institute (KHIDI), and the Institute for Basic Science (IBS).
2025.11.06 View 2921 -
 KAIST Develops AI Technology That Predicts and Assembles Cell Drug Responses Like Lego Blocks
<(From left) Dr. Younghyun Han, (top center) Dr. Chun-Kyung Lee, (bottom center) Prof. Kwang-Hyun Cho,Ph.D. candidate Hyunjin Kim>
Controlling the state of a cell in a desired direction is one of the central challenges in life sciences, including drug development, cancer treatment, and regenerative medicine. However, identifying the right drug or genetic target for that purpose is extremely difficult. To address this, researchers at KAIST have mathematically modeled the interaction between cells and drugs in a modular “Lego block” manner—breaking them down and recombining them—to develop a new AI technology that can predict not only new cell–drug reactions never before tested but also the effects of arbitrary genetic perturbations.
KAIST (President Kwang Hyung Lee) announced on the 16th of October that a research team led by Professor Kwang-Hyun Cho of the Department of Bio and Brain Engineering has developed a generative AI-based technology capable of identifying drugs and genetic targets that can guide cells toward a desired state.
“Latent space” is an invisible mathematical map used by image-generating AI to organize the essential features of objects or cells. The research team succeeded in separating the representations of cell states and drug effects within this space and then recombining them to predict the reactions of previously untested cell–drug combinations. They further extended this principle to show that the model can also predict how a cell’s state would change when a specific gene is regulated.
The team validated this approach using real experimental data. As a result, the AI identified molecular targets capable of reverting colorectal cancer cells toward a normal-like state, which the team later confirmed through cell experiments.
This finding demonstrates that the method is not limited to cancer treatment—it serves as a general platform capable of predicting various untrained cell-state transitions and drug responses. In other words, the technology not only determines whether or not a drug works but also reveals how it functions inside the cell, making the achievement particularly meaningful.
<Latent Space Direction Vector–Based Cell Transition Modeling>
The research provides a powerful tool for designing methods to induce desired cell-state changes. It is expected to have broad applications in drug discovery, cancer therapy, and regenerative medicine, such as restoring damaged cells to a healthy state.
Professor Kwang-Hyun Cho stated, “Inspired by image-generation AI, we applied the concept of a ‘direction vector,’ an idea that allows us to transform cells in a desired direction.” He added, “This technology enables quantitative analysis of how specific drugs or genes affect cells and even predicts previously unknown reactions, making it a highly generalizable AI framework.”
The study was conducted with Dr. Younghyun Han, Ph.D. candidate Hyunjin Kim, and Dr. Chun-Kyung Lee of KAIST. The research findings were published online in Cell Systems, a journal by Cell Press, on October 15.
※ Paper title: “Identifying an Optimal Perturbation to Induce a Desired Cell State by Generative Deep Learning” (DOI: 10.1016/j.cels.2025.101405)
The study was supported by the National Research Foundation of Korea (NRF) through the Ministry of Science and ICT’s Mid-Career Researcher Program and the Basic Research Laboratory (BRL) Program.
2025.10.16 View 3487
KAIST Develops AI Technology That Predicts and Assembles Cell Drug Responses Like Lego Blocks
<(From left) Dr. Younghyun Han, (top center) Dr. Chun-Kyung Lee, (bottom center) Prof. Kwang-Hyun Cho,Ph.D. candidate Hyunjin Kim>
Controlling the state of a cell in a desired direction is one of the central challenges in life sciences, including drug development, cancer treatment, and regenerative medicine. However, identifying the right drug or genetic target for that purpose is extremely difficult. To address this, researchers at KAIST have mathematically modeled the interaction between cells and drugs in a modular “Lego block” manner—breaking them down and recombining them—to develop a new AI technology that can predict not only new cell–drug reactions never before tested but also the effects of arbitrary genetic perturbations.
KAIST (President Kwang Hyung Lee) announced on the 16th of October that a research team led by Professor Kwang-Hyun Cho of the Department of Bio and Brain Engineering has developed a generative AI-based technology capable of identifying drugs and genetic targets that can guide cells toward a desired state.
“Latent space” is an invisible mathematical map used by image-generating AI to organize the essential features of objects or cells. The research team succeeded in separating the representations of cell states and drug effects within this space and then recombining them to predict the reactions of previously untested cell–drug combinations. They further extended this principle to show that the model can also predict how a cell’s state would change when a specific gene is regulated.
The team validated this approach using real experimental data. As a result, the AI identified molecular targets capable of reverting colorectal cancer cells toward a normal-like state, which the team later confirmed through cell experiments.
This finding demonstrates that the method is not limited to cancer treatment—it serves as a general platform capable of predicting various untrained cell-state transitions and drug responses. In other words, the technology not only determines whether or not a drug works but also reveals how it functions inside the cell, making the achievement particularly meaningful.
<Latent Space Direction Vector–Based Cell Transition Modeling>
The research provides a powerful tool for designing methods to induce desired cell-state changes. It is expected to have broad applications in drug discovery, cancer therapy, and regenerative medicine, such as restoring damaged cells to a healthy state.
Professor Kwang-Hyun Cho stated, “Inspired by image-generation AI, we applied the concept of a ‘direction vector,’ an idea that allows us to transform cells in a desired direction.” He added, “This technology enables quantitative analysis of how specific drugs or genes affect cells and even predicts previously unknown reactions, making it a highly generalizable AI framework.”
The study was conducted with Dr. Younghyun Han, Ph.D. candidate Hyunjin Kim, and Dr. Chun-Kyung Lee of KAIST. The research findings were published online in Cell Systems, a journal by Cell Press, on October 15.
※ Paper title: “Identifying an Optimal Perturbation to Induce a Desired Cell State by Generative Deep Learning” (DOI: 10.1016/j.cels.2025.101405)
The study was supported by the National Research Foundation of Korea (NRF) through the Ministry of Science and ICT’s Mid-Career Researcher Program and the Basic Research Laboratory (BRL) Program.
2025.10.16 View 3487 -
 KAIST Discovers Role of Huntingtin Protein in Building the Cell Skeleton
<(From Left) Professor Ji-Joon Song, Ph.D candidate Jaesung Kim, Dr. Hyeongju Kim of KAIST’s Department of Biological Sciences>
Huntington’s disease is a rare genetic disorder and a representative neurodegenerative disease, characterized by loss of motor control, cognitive decline, and psychiatric problems. An international research team has discovered that the “huntingtin protein,” the causal protein of Huntington’s disease (whose mutations are the direct cause of the disease), also performs a new function: directly organizing the cytoskeleton, the fine structural framework inside cells. This discovery is expected to contribute not only to understanding the pathogenic mechanism of Huntington’s disease, but also to research on neurodevelopmental disorders such as Alzheimer’s disease and Parkinson’s disease, as well as muscle- or mobility-related diseases such as muscular dystrophy.
KAIST (President Kwang Hyung Lee) announced on September 30 that a research team led by Professor Ji-Joon Song of the Department of Biological Sciences, in collaboration with the Institute of Science and Technology Austria (ISTA), Sorbonne University/Paris Brain Institute, and the Swiss Federal Institute of Technology Lausanne (EPFL), has uncovered—through cryo-electron microscopy (cryo-EM) and cell biology methods—the structural principle by which the huntingtin protein arranges cytoskeletal microfilaments (F-actin) into bundles.
Until now, the huntingtin protein was known only to “use” the cytoskeleton, being involved in vesicle transport or microtubule-based transport. The team, however, demonstrated that huntingtin physically organizes the cytoskeleton itself. This study is considered the first in the world to prove this new role of the huntingtin protein at the molecular level.
The researchers confirmed that huntingtin binds directly to cytoskeletal microfilaments (F-actin), and that pairs of huntingtin proteins bundle the cytoskeleton into arrays at intervals of about 20 nanometers.
Such cytoskeletal bundles play a crucial role in the development of neural connectivity. Indeed, structural development of neurons was found to be impaired in nerve cells deficient in the huntingtin protein.
<Elucidation of the Mechanism of Cytoskeletal Microfilament Bundle Formation by Huntingtin Protein and Its Impact on Neuronal Development>
First author Jaesung Kim, a PhD candidate at KAIST, stated, “This study provides a new perspective for understanding the molecular mechanism of the huntingtin protein, the cause of an incurable disease that has long remained a mystery.”
Professor Ji-Joon Song of KAIST’s Department of Biological Sciences explained, “This achievement not only provides an important clue to understanding the pathogenic mechanism of Huntington’s disease, but is also expected to have a far-reaching impact on research into cytoskeleton-related diseases,” and added that “it opens new avenues for exploring the role of the huntingtin protein in diverse biological phenomena such as cell division, migration, and mechanical signal transduction.”
This research was conducted with Jaesung Kim (PhD candidate, KAIST), Hyeongju Kim (now at Harvard University), Rémi Carpentier (Paris Brain Institute), Mariacristina Capizzi (Paris Brain Institute), and others as co-first authors, and was published on September 19 in Science Advances, a sister journal of Science.
※ Paper title: “Structure of the Huntingtin F-actin complex reveals its role in cytoskeleton organization,” DOI: https://doi.org/10.1126/sciadv.adw4124※ Co-corresponding authors: Ji-Joon Song (KAIST), Florian Schur (ISTA), and Sandrine Humbert (Sorbonne University/Paris Brain Institute).
This research was supported by the Ministry of Health and Welfare’s Global Research Collaboration Program (Korea–Switzerland Biohealth International Joint Research) and the Korea–Austria Cooperation Program.
2025.09.30 View 3040
KAIST Discovers Role of Huntingtin Protein in Building the Cell Skeleton
<(From Left) Professor Ji-Joon Song, Ph.D candidate Jaesung Kim, Dr. Hyeongju Kim of KAIST’s Department of Biological Sciences>
Huntington’s disease is a rare genetic disorder and a representative neurodegenerative disease, characterized by loss of motor control, cognitive decline, and psychiatric problems. An international research team has discovered that the “huntingtin protein,” the causal protein of Huntington’s disease (whose mutations are the direct cause of the disease), also performs a new function: directly organizing the cytoskeleton, the fine structural framework inside cells. This discovery is expected to contribute not only to understanding the pathogenic mechanism of Huntington’s disease, but also to research on neurodevelopmental disorders such as Alzheimer’s disease and Parkinson’s disease, as well as muscle- or mobility-related diseases such as muscular dystrophy.
KAIST (President Kwang Hyung Lee) announced on September 30 that a research team led by Professor Ji-Joon Song of the Department of Biological Sciences, in collaboration with the Institute of Science and Technology Austria (ISTA), Sorbonne University/Paris Brain Institute, and the Swiss Federal Institute of Technology Lausanne (EPFL), has uncovered—through cryo-electron microscopy (cryo-EM) and cell biology methods—the structural principle by which the huntingtin protein arranges cytoskeletal microfilaments (F-actin) into bundles.
Until now, the huntingtin protein was known only to “use” the cytoskeleton, being involved in vesicle transport or microtubule-based transport. The team, however, demonstrated that huntingtin physically organizes the cytoskeleton itself. This study is considered the first in the world to prove this new role of the huntingtin protein at the molecular level.
The researchers confirmed that huntingtin binds directly to cytoskeletal microfilaments (F-actin), and that pairs of huntingtin proteins bundle the cytoskeleton into arrays at intervals of about 20 nanometers.
Such cytoskeletal bundles play a crucial role in the development of neural connectivity. Indeed, structural development of neurons was found to be impaired in nerve cells deficient in the huntingtin protein.
<Elucidation of the Mechanism of Cytoskeletal Microfilament Bundle Formation by Huntingtin Protein and Its Impact on Neuronal Development>
First author Jaesung Kim, a PhD candidate at KAIST, stated, “This study provides a new perspective for understanding the molecular mechanism of the huntingtin protein, the cause of an incurable disease that has long remained a mystery.”
Professor Ji-Joon Song of KAIST’s Department of Biological Sciences explained, “This achievement not only provides an important clue to understanding the pathogenic mechanism of Huntington’s disease, but is also expected to have a far-reaching impact on research into cytoskeleton-related diseases,” and added that “it opens new avenues for exploring the role of the huntingtin protein in diverse biological phenomena such as cell division, migration, and mechanical signal transduction.”
This research was conducted with Jaesung Kim (PhD candidate, KAIST), Hyeongju Kim (now at Harvard University), Rémi Carpentier (Paris Brain Institute), Mariacristina Capizzi (Paris Brain Institute), and others as co-first authors, and was published on September 19 in Science Advances, a sister journal of Science.
※ Paper title: “Structure of the Huntingtin F-actin complex reveals its role in cytoskeleton organization,” DOI: https://doi.org/10.1126/sciadv.adw4124※ Co-corresponding authors: Ji-Joon Song (KAIST), Florian Schur (ISTA), and Sandrine Humbert (Sorbonne University/Paris Brain Institute).
This research was supported by the Ministry of Health and Welfare’s Global Research Collaboration Program (Korea–Switzerland Biohealth International Joint Research) and the Korea–Austria Cooperation Program.
2025.09.30 View 3040 -
 KAIST Develops Bioelectrosynthesis Platform for Switch-Like Precision Control of Cell Signaling
<(From left)Professor Jimin Park, Ph.D candidate Myeongeun Lee, Ph.D cadidate Jaewoong Lee,Professor Jihan Kim>
Cells use various signaling molecules to regulate the nervous, immune, and vascular systems. Among these, nitric oxide (NO) and ammonia (NH₃) play important roles, but their chemical instability and gaseous nature make them difficult to generate or control externally. A KAIST research team has developed a platform that generates specific signaling molecules in situ from a single precursor under an applied electrical signal, enabling switch-like, precise spatiotemporal control of cellular responses. This approach could provide a foundation for future medical technologies such as electroceuticals, electrogenetics, and personalized cell therapies.
KAIST (President Kwang Hyung Lee) announced on August 11 that a research team led by Professor Jimin Park from the Department of Chemical and Biomolecular Engineering, in collaboration with Professor Jihan Kim's group, has developed a 'Bioelectrosynthesis Platform' capable of producing either nitric oxide or ammonia on demand using only an electrical signal. The platform allows control over the timing, spatial range, and duration of cell responses.
Inspired by enzymes involved in nitrite reduction, the researchers implemented an electrochemical strategy that selectively produces nitric oxide or ammonia from a single precursor, nitrite (NO₂⁻). By changing the catalyst, the team generated ammonia or nitric oxide from nitrite using a copper-molybdenum-sulfur catalyst (Cu2MoS4) and an iron-incorporated catalyst (FeCuMS4), respectively.
Through electrochemical measurements and computer simulations, the team revealed that Fe sites in the FeCuMoS4 catalyst bind nitric oxide intermediates more strongly, shifting product selectivity toward nitric oxide. Under the same electrical conditions, the Fe-containing catalyst preferentially produces nitric oxide, whereas the Cu2MoS4 catalyst favors ammonia production.
<Figure 1. Schematic diagram of a bio-electrosynthesis platform that synthesizes a desired signaling substance with an electrical signal (left) and the results of precise cell control using it (right)>
The research team demonstrated biological functionality by using the platform to activate ion channels in human cells. Specifically, electrochemically produced nitric oxide activated TRPV1 channels (responsive to heat and chemical stimuli), while electrochemically produced ammonia induced intracellular alkalinization and activated OTOP1 proton channels. By tuning the applied voltage and electrolysis duration, the team modulated the onset time, spatial extent, and termination of cellular responses, which effectively turned cellular signaling on and off like a switch.
<Figure 2. Experimental results showing the change in the production ratio of nitric oxide and ammonia signaling substances according to the type of catalyst (left) and computational simulation results showing the strong bond between iron and nitric oxide (right)>
Professor Jimin Park said, "This work is significant because it enables precise cellular control by selectively producing signaling molecules with electricity. We believe it has strong potential for applications in electroceutical technologies targeting the nervous system or metabolic disorders."
Myeongeun Lee and Jaewoong Lee, Ph.D. students in the Department of Chemical and Biomolecular Engineering at KAIST, served as the co-first authors. Professor Jihan Kim is a co-author. The paper was published online in 'Angewandte Chemie International Edition' on July 8, 2025 (DOI: 10.1002/ange.202508192).
Reference: https://doi.org/10.1002/ange.202508192
Authors: Myeongeun Lee†, Jaewoong Lee†, Yongha Kim, Changho Lee, Sang Yeon Oh, Prof. Jihan Kim, Prof. Jimin Park*
†These authors contributed equally. *Corresponding author.
2025.08.12 View 4002
KAIST Develops Bioelectrosynthesis Platform for Switch-Like Precision Control of Cell Signaling
<(From left)Professor Jimin Park, Ph.D candidate Myeongeun Lee, Ph.D cadidate Jaewoong Lee,Professor Jihan Kim>
Cells use various signaling molecules to regulate the nervous, immune, and vascular systems. Among these, nitric oxide (NO) and ammonia (NH₃) play important roles, but their chemical instability and gaseous nature make them difficult to generate or control externally. A KAIST research team has developed a platform that generates specific signaling molecules in situ from a single precursor under an applied electrical signal, enabling switch-like, precise spatiotemporal control of cellular responses. This approach could provide a foundation for future medical technologies such as electroceuticals, electrogenetics, and personalized cell therapies.
KAIST (President Kwang Hyung Lee) announced on August 11 that a research team led by Professor Jimin Park from the Department of Chemical and Biomolecular Engineering, in collaboration with Professor Jihan Kim's group, has developed a 'Bioelectrosynthesis Platform' capable of producing either nitric oxide or ammonia on demand using only an electrical signal. The platform allows control over the timing, spatial range, and duration of cell responses.
Inspired by enzymes involved in nitrite reduction, the researchers implemented an electrochemical strategy that selectively produces nitric oxide or ammonia from a single precursor, nitrite (NO₂⁻). By changing the catalyst, the team generated ammonia or nitric oxide from nitrite using a copper-molybdenum-sulfur catalyst (Cu2MoS4) and an iron-incorporated catalyst (FeCuMS4), respectively.
Through electrochemical measurements and computer simulations, the team revealed that Fe sites in the FeCuMoS4 catalyst bind nitric oxide intermediates more strongly, shifting product selectivity toward nitric oxide. Under the same electrical conditions, the Fe-containing catalyst preferentially produces nitric oxide, whereas the Cu2MoS4 catalyst favors ammonia production.
<Figure 1. Schematic diagram of a bio-electrosynthesis platform that synthesizes a desired signaling substance with an electrical signal (left) and the results of precise cell control using it (right)>
The research team demonstrated biological functionality by using the platform to activate ion channels in human cells. Specifically, electrochemically produced nitric oxide activated TRPV1 channels (responsive to heat and chemical stimuli), while electrochemically produced ammonia induced intracellular alkalinization and activated OTOP1 proton channels. By tuning the applied voltage and electrolysis duration, the team modulated the onset time, spatial extent, and termination of cellular responses, which effectively turned cellular signaling on and off like a switch.
<Figure 2. Experimental results showing the change in the production ratio of nitric oxide and ammonia signaling substances according to the type of catalyst (left) and computational simulation results showing the strong bond between iron and nitric oxide (right)>
Professor Jimin Park said, "This work is significant because it enables precise cellular control by selectively producing signaling molecules with electricity. We believe it has strong potential for applications in electroceutical technologies targeting the nervous system or metabolic disorders."
Myeongeun Lee and Jaewoong Lee, Ph.D. students in the Department of Chemical and Biomolecular Engineering at KAIST, served as the co-first authors. Professor Jihan Kim is a co-author. The paper was published online in 'Angewandte Chemie International Edition' on July 8, 2025 (DOI: 10.1002/ange.202508192).
Reference: https://doi.org/10.1002/ange.202508192
Authors: Myeongeun Lee†, Jaewoong Lee†, Yongha Kim, Changho Lee, Sang Yeon Oh, Prof. Jihan Kim, Prof. Jimin Park*
†These authors contributed equally. *Corresponding author.
2025.08.12 View 4002 -
 KAIST Unveils New Possibilities for Treating Intractable Brain Tumors
< Photo 1. (From left) Professor Heung Kyu Lee, KAIST Department of Biological Sciences, and Dr. Keun Bon Ku >
Immunotherapy, which enhances the immune system's T cell response to eliminate cancer cells, has emerged as a key approach in cancer treatment. However, in the case of glioblastoma, an aggressive and treatment-resistant brain tumor, numerous clinical trials have failed to confirm their efficacy. Korean researchers have recently analyzed the mechanisms that cause T cell exhaustion, which is characterized by a loss of function or a weakened response following prolonged exposure to antigens in such intractable cancers, identifying key control factors in T cell activation and clarifying the mechanisms that enhance therapeutic effectiveness.
KAIST (represented by President Kwang Hyung Lee) announced on the 6th of November that Professor Heung Kyu Lee’s team from the Department of Biological Sciences, in collaboration with the Korea Research Institute of Chemical Technology (represented by President Young Kuk Lee), has confirmed improved survival rates in a glioblastoma mouse model. By removing the inhibitory Fc gamma receptor (FcγRIIB), the research team was able to restore the responsiveness of cytotoxic T cells to immune checkpoint inhibitors, leading to enhanced anticancer activity.
The research team examined the effect of FcγRIIB, an inhibitory receptor recently found in cytotoxic T cells, on tumor-infiltrating T cells and the therapeutic effectiveness of the anti-PD-1 immune checkpoint inhibitor.
< Figure 1. Study results on improved survival rate due to increased antitumor activity of anti-PD-1 treatment in inhibitory Fc gamma receptor(Fcgr2b) ablation mice with murine glioblastoma. >
Their findings showed that deleting FcγRIIB induced the increase of tumor antigen-specific memory T cells, which helps to suppress exhaustion, enhances stem-like qualities, and reactivates T cell-mediated antitumor immunity, particularly in response to anti-PD-1 treatment. Furthermore, FcγRIIB deletion led to an increase in antigen-specific memory T cells that maintained continuous infiltration into the tumor tissue.
This study presents a new therapeutic target for tumors unresponsive to immune checkpoint inhibitors and demonstrates that combining FcγRIIB inhibition with anti-PD-1 treatment can produce synergistic effects, potentially improving therapeutic outcomes for tumors like glioblastoma, which typically show resistance to anti-PD-1 therapy.
< Figure 2. Overview of the study on the enhanced response to anti-PD-1 therapy for glioblastoma brain tumors upon deletion of the inhibitory Fc gamma receptor (FcγRIIB) in tumor microenvironment. When the inhibitory Fc gamma receptor (FcγRIIB) of cytotoxic T cells is deleted, an increase in tumor-specific memory T cells (Ttsms) was observed. In addition, this T cell subset is identified as originating from the tumor-draining lymph nodes(TdLNs) and leads to persistent infiltration into the tumor tissue. Anti-PD-1 therapy leads to an increased anti-tumor immune response via Ttsms, which is confirmed by increased tumor cell toxicity and increased cell division and decreased cell de-migration indices. Ultimately, the increased cytotoxic T cell immune response leads to an increase in the survival rate of glioblastoma. >
Professor Heung Kyu Lee explained, "This study offers a way to overcome clinical failures in treating brain tumors with immune checkpoint therapy and opens possibilities for broader applications to other intractable cancers. It also highlights the potential of utilizing cytotoxic T cells for tumor cell therapy."
The study, led by Dr. Keun Bon Ku of KAIST (currently a senior researcher at the Korea Research Institute of Chemical Technology's Center for Infectious Disease Diagnosis and Prevention), along with Chae Won Kim, Yumin Kim, Byeong Hoon Kang, Jeongwoo La, In Kang, Won Hyung Park, Stephen Ahn, and Sung Ki Lee, was published online on October 26 in the Journal for ImmunoTherapy of Cancer, an international journal in tumor immunology and therapy from the Society for Immunotherapy of Cancer. (Paper title: “Inhibitory Fcγ receptor deletion enhances CD8 T cell stemness increasing anti-PD-1 therapy responsiveness against glioblastoma,” http://dx.doi.org/10.1136/jitc-2024-009449).
This research received support from the National Research Foundation of Korea, the Bio & Medical Technology Development Program, and the Samsung Science & Technology Foundation.
2024.11.15 View 11650
KAIST Unveils New Possibilities for Treating Intractable Brain Tumors
< Photo 1. (From left) Professor Heung Kyu Lee, KAIST Department of Biological Sciences, and Dr. Keun Bon Ku >
Immunotherapy, which enhances the immune system's T cell response to eliminate cancer cells, has emerged as a key approach in cancer treatment. However, in the case of glioblastoma, an aggressive and treatment-resistant brain tumor, numerous clinical trials have failed to confirm their efficacy. Korean researchers have recently analyzed the mechanisms that cause T cell exhaustion, which is characterized by a loss of function or a weakened response following prolonged exposure to antigens in such intractable cancers, identifying key control factors in T cell activation and clarifying the mechanisms that enhance therapeutic effectiveness.
KAIST (represented by President Kwang Hyung Lee) announced on the 6th of November that Professor Heung Kyu Lee’s team from the Department of Biological Sciences, in collaboration with the Korea Research Institute of Chemical Technology (represented by President Young Kuk Lee), has confirmed improved survival rates in a glioblastoma mouse model. By removing the inhibitory Fc gamma receptor (FcγRIIB), the research team was able to restore the responsiveness of cytotoxic T cells to immune checkpoint inhibitors, leading to enhanced anticancer activity.
The research team examined the effect of FcγRIIB, an inhibitory receptor recently found in cytotoxic T cells, on tumor-infiltrating T cells and the therapeutic effectiveness of the anti-PD-1 immune checkpoint inhibitor.
< Figure 1. Study results on improved survival rate due to increased antitumor activity of anti-PD-1 treatment in inhibitory Fc gamma receptor(Fcgr2b) ablation mice with murine glioblastoma. >
Their findings showed that deleting FcγRIIB induced the increase of tumor antigen-specific memory T cells, which helps to suppress exhaustion, enhances stem-like qualities, and reactivates T cell-mediated antitumor immunity, particularly in response to anti-PD-1 treatment. Furthermore, FcγRIIB deletion led to an increase in antigen-specific memory T cells that maintained continuous infiltration into the tumor tissue.
This study presents a new therapeutic target for tumors unresponsive to immune checkpoint inhibitors and demonstrates that combining FcγRIIB inhibition with anti-PD-1 treatment can produce synergistic effects, potentially improving therapeutic outcomes for tumors like glioblastoma, which typically show resistance to anti-PD-1 therapy.
< Figure 2. Overview of the study on the enhanced response to anti-PD-1 therapy for glioblastoma brain tumors upon deletion of the inhibitory Fc gamma receptor (FcγRIIB) in tumor microenvironment. When the inhibitory Fc gamma receptor (FcγRIIB) of cytotoxic T cells is deleted, an increase in tumor-specific memory T cells (Ttsms) was observed. In addition, this T cell subset is identified as originating from the tumor-draining lymph nodes(TdLNs) and leads to persistent infiltration into the tumor tissue. Anti-PD-1 therapy leads to an increased anti-tumor immune response via Ttsms, which is confirmed by increased tumor cell toxicity and increased cell division and decreased cell de-migration indices. Ultimately, the increased cytotoxic T cell immune response leads to an increase in the survival rate of glioblastoma. >
Professor Heung Kyu Lee explained, "This study offers a way to overcome clinical failures in treating brain tumors with immune checkpoint therapy and opens possibilities for broader applications to other intractable cancers. It also highlights the potential of utilizing cytotoxic T cells for tumor cell therapy."
The study, led by Dr. Keun Bon Ku of KAIST (currently a senior researcher at the Korea Research Institute of Chemical Technology's Center for Infectious Disease Diagnosis and Prevention), along with Chae Won Kim, Yumin Kim, Byeong Hoon Kang, Jeongwoo La, In Kang, Won Hyung Park, Stephen Ahn, and Sung Ki Lee, was published online on October 26 in the Journal for ImmunoTherapy of Cancer, an international journal in tumor immunology and therapy from the Society for Immunotherapy of Cancer. (Paper title: “Inhibitory Fcγ receptor deletion enhances CD8 T cell stemness increasing anti-PD-1 therapy responsiveness against glioblastoma,” http://dx.doi.org/10.1136/jitc-2024-009449).
This research received support from the National Research Foundation of Korea, the Bio & Medical Technology Development Program, and the Samsung Science & Technology Foundation.
2024.11.15 View 11650 -
 KAIST presents strategies for Holotomography in advanced bio research
Measuring and analyzing three-dimensional (3D) images of live cells and tissues is considered crucial in advanced fields of biology and medicine. Organoids, which are 3D structures that mimic organs, are particular examples that significantly benefits 3D live imaging. Organoids provide effective alternatives to animal testing in the drug development processes, and can rapidly determine personalized medicine. On the other hand, active researches are ongoing to utilize organoids for organ replacement.
< Figure 1. Schematic illustration of holotomography compared to X-ray CT. Similar to CT, they share the commonality of measuring the optical properties of an unlabeled specimen in three dimensions. Instead of X-rays, holotomography irradiates light in the visible range, and provides refractive index measurements of transparent specimens rather than absorptivity. While CT obtains three-dimensional information only through mechanical rotation of the irradiating light, holotomography can replace this by applying wavefront control technology in the visible range. >
Organelle-level observation of 3D biological specimens such as organoids and stem cell colonies without staining or preprocessing holds significant implications for both innovating basic research and bioindustrial applications related to regenerative medicine and bioindustrial applications.
Holotomography (HT) is a 3D optical microscopy that implements 3D reconstruction analogous to that of X-ray computed tomography (CT). Although HT and CT share a similar theoretical background, HT facilitates high-resolution examination inside cells and tissues, instead of the human body. HT obtains 3D images of cells and tissues at the organelle level without chemical or genetic labeling, thus overcomes various challenges of existing methods in bio research and industry. Its potential is highlighted in research fields where sample physiology must not be disrupted, such as regenerative medicine, personalized medicine, and infertility treatment.
< Figure 2. Label-free 3D imaging of diverse live cells. Time-lapse image of Hep3B cells illustrating subcellular morphology changes upon H2O2 treatment, followed by cellular recovery after returning to the regular cell culture medium. >
This paper introduces the advantages and broad applicability of HT to biomedical researchers, while presenting an overview of principles and future technical challenges to optical researchers. It showcases various cases of applying HT in studies such as 3D biology, regenerative medicine, and cancer research, as well as suggesting future optical development. Also, it categorizes HT based on the light source, to describe the principles, limitations, and improvements of each category in detail. Particularly, the paper addresses strategies for deepening cell and organoid studies by introducing artificial intelligence (AI) to HT.
Due to its potential to drive advanced bioindustry, HT is attracting interest and investment from universities and corporates worldwide. The KAIST research team has been leading this international field by developing core technologies and carrying out key application researches throughout the last decade.
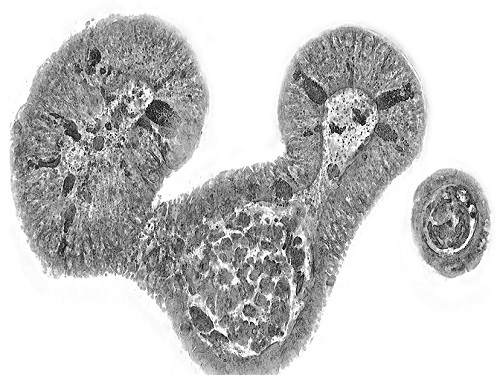
< Figure 3. Various types of cells and organelles that make up the imaging barrier of a living intestinal organoid can be observed using holotomography. >
This paper, co-authored by Dr. Geon Kim from KAIST Research Center for Natural Sciences, Professor Ki-Jun Yoon's team from the Department of Biological Sciences, Director Bon-Kyoung Koo's team from the Institute for Basic Science (IBS) Center for Genome Engineering, and Dr. Seongsoo Lee's team from the Korea Basic Science Institute (KBSI), was published in 'Nature Reviews Methods Primers' on the 25th of July. This research was supported by the Leader Grant and Basic Science Research Program of the National Research Foundation, the Hologram Core Technology Development Grant of the Ministry of Science and ICT, the Nano and Material Technology Development Project, and the Health and Medical R&D Project of the Ministry of Health and Welfare.
2024.07.30 View 11479
KAIST presents strategies for Holotomography in advanced bio research
Measuring and analyzing three-dimensional (3D) images of live cells and tissues is considered crucial in advanced fields of biology and medicine. Organoids, which are 3D structures that mimic organs, are particular examples that significantly benefits 3D live imaging. Organoids provide effective alternatives to animal testing in the drug development processes, and can rapidly determine personalized medicine. On the other hand, active researches are ongoing to utilize organoids for organ replacement.
< Figure 1. Schematic illustration of holotomography compared to X-ray CT. Similar to CT, they share the commonality of measuring the optical properties of an unlabeled specimen in three dimensions. Instead of X-rays, holotomography irradiates light in the visible range, and provides refractive index measurements of transparent specimens rather than absorptivity. While CT obtains three-dimensional information only through mechanical rotation of the irradiating light, holotomography can replace this by applying wavefront control technology in the visible range. >
Organelle-level observation of 3D biological specimens such as organoids and stem cell colonies without staining or preprocessing holds significant implications for both innovating basic research and bioindustrial applications related to regenerative medicine and bioindustrial applications.
Holotomography (HT) is a 3D optical microscopy that implements 3D reconstruction analogous to that of X-ray computed tomography (CT). Although HT and CT share a similar theoretical background, HT facilitates high-resolution examination inside cells and tissues, instead of the human body. HT obtains 3D images of cells and tissues at the organelle level without chemical or genetic labeling, thus overcomes various challenges of existing methods in bio research and industry. Its potential is highlighted in research fields where sample physiology must not be disrupted, such as regenerative medicine, personalized medicine, and infertility treatment.
< Figure 2. Label-free 3D imaging of diverse live cells. Time-lapse image of Hep3B cells illustrating subcellular morphology changes upon H2O2 treatment, followed by cellular recovery after returning to the regular cell culture medium. >
This paper introduces the advantages and broad applicability of HT to biomedical researchers, while presenting an overview of principles and future technical challenges to optical researchers. It showcases various cases of applying HT in studies such as 3D biology, regenerative medicine, and cancer research, as well as suggesting future optical development. Also, it categorizes HT based on the light source, to describe the principles, limitations, and improvements of each category in detail. Particularly, the paper addresses strategies for deepening cell and organoid studies by introducing artificial intelligence (AI) to HT.
Due to its potential to drive advanced bioindustry, HT is attracting interest and investment from universities and corporates worldwide. The KAIST research team has been leading this international field by developing core technologies and carrying out key application researches throughout the last decade.
< Figure 3. Various types of cells and organelles that make up the imaging barrier of a living intestinal organoid can be observed using holotomography. >
This paper, co-authored by Dr. Geon Kim from KAIST Research Center for Natural Sciences, Professor Ki-Jun Yoon's team from the Department of Biological Sciences, Director Bon-Kyoung Koo's team from the Institute for Basic Science (IBS) Center for Genome Engineering, and Dr. Seongsoo Lee's team from the Korea Basic Science Institute (KBSI), was published in 'Nature Reviews Methods Primers' on the 25th of July. This research was supported by the Leader Grant and Basic Science Research Program of the National Research Foundation, the Hologram Core Technology Development Grant of the Ministry of Science and ICT, the Nano and Material Technology Development Project, and the Health and Medical R&D Project of the Ministry of Health and Welfare.
2024.07.30 View 11479 -
 KAIST presents a fundamental technology to remove metastatic traits from lung cancer cells
KAIST (President Kwang Hyung Lee) announced on January 30th that a research team led by Professor Kwang-Hyun Cho from the Department of Bio and Brain Engineering succeeded in using systems biology research to change the properties of carcinogenic cells in the lungs and eliminate both drug resistance and their ability to proliferate out to other areas of the body.
As the incidences of cancer increase within aging populations, cancer has become the most lethal disease threatening healthy life. Fatality rates are especially high when early detection does not happen in time and metastasis has occurred in various organs. In order to resolve this problem, a series of attempts were made to remove or lower the ability of cancer cells to spread, but they resulted in cancer cells in the intermediate state becoming more unstable and even more malignant, which created serious treatment challenges.
Professor Kwang-Hyun Cho's research team simulated various cancer cell states in the Epithelial-to-Mesenchymal Transition (EMT) of lung cancer cells, between epithelial cells without metastatic ability and mesenchymal cells with metastatic ability. A mathematical model of molecular network was established, and key regulators that could reverse the state of invasive and drug resistant mesenchymal cells back to the epithelial state were discovered through computer simulation analysis and molecular cell experiments. In particular, this process succeeded in properly reverting the mesenchymal lung cancer cells to a state where they were sensitive to chemotherapy treatment while avoiding the unstable EMT hybrid cell state in the middle process, which had remained a difficult problem.
The results of this research, in which KAIST Ph.D. student Namhee Kim, Dr. Chae Young Hwang, Researcher Taeyoung Kim, and Ph.D. student Hyunjin Kim participated, were published as an online paper in the international journal “Cancer Research” published by the American Association for Cancer Research (AACR) on January 30th. (Paper title: A cell fate reprogramming strategy reverses epithelial-to-mesenchymal transition of lung cancer cells while avoiding hybrid states)
Cells in an EMT hybrid state, which are caused by incomplete transitions during the EMT process in cancer cells, have the characteristics of both epithelial cells and mesenchymal cells, and are known to have high drug resistance and metastatic potential by acquiring high stem cell capacity. In particular, EMT is further enhanced through factors such as transforming growth factor-beta (TGF-β) secreted from the tumor microenvironment (TME) and, as a result, various cell states with high plasticity appear. Due to the complexity of EMT, it has been very difficult to completely reverse the transitional process of the mesenchymal cancer cells to an epithelial cell state in which metastatic ability and drug resistance are eliminated while avoiding the EMT hybrid cell state with high metastatic ability and drug resistance.
Professor Kwang-Hyun Cho's research team established a mathematical model of the gene regulation network that governs the complex process of EMT, and then applied large-scale computer simulation analysis and complex system network control technology to identify and verify 'p53', 'SMAD4', and 'ERK1' and 'ERK 2' (collectively ERKs) through molecular cell experiments as the three key molecular targets that can transform lung cancer cells in the mesenchymal cell state, reversed back to an epithelial cell state that no longer demonstrates the ability to metastasize, while avoiding the EMT hybrid cell state.
In particular, by analyzing the molecular regulatory mechanism of the complex EMT process at the system level, the key pathways were identified that were linked to the positive feedback that plays an important role in completely returning cancer cells to an epithelial cell state in which metastatic ability and drug resistance are removed.
This discovery is significant in that it proved that mesenchymal cells can be reverted to the state of epithelial cells under conditions where TGF-β stimulation are present, like they are in the actual environment where cancer tissue forms in the human body.
Abnormal EMT in cancer cells leads to various malignant traits such as the migration and invasion of cancer cells, changes in responsiveness to chemotherapy treatment, enhanced stem cell function, and the dissemination of cancer. In particular, the acquisition of the metastatic ability of cancer cells is a key determinant factor for the prognosis of cancer patients. The EMT reversal technology in lung cancer cells developed in this research is a new anti-cancer treatment strategy that reprograms cancer cells to eliminate their high plasticity and metastatic potential and increase their responsiveness to chemotherapy.
Professor Kwang-Hyun Cho said, "By succeeding in reversing the state of lung cancer cells that acquired high metastatic traits and resistance to drugs and reverting them to a treatable epithelial cell state with renewed sensitivity to chemotherapy, the research findings propose a new strategy for treatments that can improve the prognosis of cancer patients.”
Professor Kwang-Hyun Cho's research team was the first to present the principle of reversal treatment to revert cancer cells to normal cells, following through with the announcement of the results of their study that reverted colon cancer cells to normal colon cells in January of 2020, and also presenting successful re-programming research where the most malignant basal type breast cancer cells turned into less-malignant luminal type breast cancer cells that were treatable with hormonal therapies in January of 2022. This latest research result is the third in the development of reversal technology where lung cancer cells that had acquired metastatic traits returned to a state in which their metastatic ability was removed and drug sensitivity was enhanced.
This research was carried out with support from the Ministry of Science and ICT and the National Research Foundation of Korea's Basic Research in Science & Engineering Program for Mid-Career Researchers.
< Figure 1. Construction of the mathematical model of the regulatory network to represent the EMT phenotype based on the interaction between various molecules related to EMT.
(A) Professor Kwang-Hyun Cho's research team investigated numerous literatures and databases related to complex EMT, and based on comparative analysis of cell line data showing epithelial and mesenchymal cell conditions, they extracted key signaling pathways related to EMT and built a mathematical model of regulatory network (B) By comparing the results of computer simulation analysis and the molecular cell experiments, it was verified how well the constructed mathematical model simulated the actual cellular phenomena. >
< Figure 2. Understanding of various EMT phenotypes through large-scale computer simulation analysis and complex system network control technology.
(A) Through computer simulation analysis and experiments, Professor Kwang-Hyun Cho's research team found that complete control of EMT is impossible with single-molecule control alone. In particular, through comparison of the relative stability of attractors, it was revealed that the cell state exhibiting EMT hybrid characteristics has unstable properties. (B), (C) Based on these results, Prof. Cho’s team identified two feedbacks (positive feedback consisting of Snail-miR-34 and ZEB1-miR-200) that play an important role in avoiding the EMT hybrid state that appeared in the TGF-β-ON state. It was found through computer simulation analysis that the two feedbacks restore relatively high stability when the excavated p53 and SMAD4 are regulated. In addition, molecular cell experiments demonstrated that the expression levels of E-cad and ZEB1, which are representative phenotypic markers of EMT, changed similarly to the expression profile in the epithelial cell state, despite the TGF-β-ON state. >
< Figure 3. Complex molecular network analysis and discovery of reprogramming molecular targets for intact elimination of EMT hybrid features.
(A) Controlling the expression of p53 and SMAD4 in lung cancer cell lines was expected to overcome drug resistance, but contrary to expectations, chemotherapy responsiveness was not restored. (B) Professor Kwang-Hyun Cho's research team additionally analyzed computer simulations, genome data, and experimental results and found that high expression levels of TWIST1 and EPCAM were related to drug resistance. (C) Prof. Cho’s team identified three key molecular targets: p53, SMAD4 and ERK1 & ERK2. (D), (E) Furthermore, they identified a key pathway that plays an important role in completely reversing into epithelial cells while avoiding EMT hybrid characteristics, and confirmed through network analysis and attractor analysis that high stability of the key pathway was restored when the proposed molecular target was controlled. >
< Figure 4. Verification through experiments with lung cancer cell lines.
When p53 was activated and SMAD4 and ERK1/2 were inhibited in lung cancer cell lines, (A), (B) E-cad protein expression increased and ZEB1 protein expression decreased, and (C) mesenchymal cell status including TWIST1 and EPCAM and gene expression of markers related to stem cell potential characteristics were completely inhibited. In addition, (D) it was confirmed that resistance to chemotherapy treatment was also overcome as the cell state was reversed by the regulated target. >
< Figure 5. A schematic representation of the research results.
Prof. Cho’s research team identified key molecular regulatory pathways to avoid high plasticity formed by abnormal EMT of cancer cells and reverse it to an epithelial cell state through systems biology research. From this analysis, a reprogramming molecular target that can reverse the state of mesenchymal cells with acquired invasiveness and drug resistance to the state of epithelial cells with restored drug responsiveness was discovered.
For lung cancer cells, when a drug that enhances the expression of p53, one of the molecular targets discovered, and inhibits the expression of SMAD4 and ERK1 & ERK2 is administered, the molecular network of genes in the state of mesenchymal cells is modified, eventually eliminating metastatic ability and it is reprogrammed to turn into epithelial cells without the resistance to chemotherapy treatments. >
2023.01.30 View 26866
KAIST presents a fundamental technology to remove metastatic traits from lung cancer cells
KAIST (President Kwang Hyung Lee) announced on January 30th that a research team led by Professor Kwang-Hyun Cho from the Department of Bio and Brain Engineering succeeded in using systems biology research to change the properties of carcinogenic cells in the lungs and eliminate both drug resistance and their ability to proliferate out to other areas of the body.
As the incidences of cancer increase within aging populations, cancer has become the most lethal disease threatening healthy life. Fatality rates are especially high when early detection does not happen in time and metastasis has occurred in various organs. In order to resolve this problem, a series of attempts were made to remove or lower the ability of cancer cells to spread, but they resulted in cancer cells in the intermediate state becoming more unstable and even more malignant, which created serious treatment challenges.
Professor Kwang-Hyun Cho's research team simulated various cancer cell states in the Epithelial-to-Mesenchymal Transition (EMT) of lung cancer cells, between epithelial cells without metastatic ability and mesenchymal cells with metastatic ability. A mathematical model of molecular network was established, and key regulators that could reverse the state of invasive and drug resistant mesenchymal cells back to the epithelial state were discovered through computer simulation analysis and molecular cell experiments. In particular, this process succeeded in properly reverting the mesenchymal lung cancer cells to a state where they were sensitive to chemotherapy treatment while avoiding the unstable EMT hybrid cell state in the middle process, which had remained a difficult problem.
The results of this research, in which KAIST Ph.D. student Namhee Kim, Dr. Chae Young Hwang, Researcher Taeyoung Kim, and Ph.D. student Hyunjin Kim participated, were published as an online paper in the international journal “Cancer Research” published by the American Association for Cancer Research (AACR) on January 30th. (Paper title: A cell fate reprogramming strategy reverses epithelial-to-mesenchymal transition of lung cancer cells while avoiding hybrid states)
Cells in an EMT hybrid state, which are caused by incomplete transitions during the EMT process in cancer cells, have the characteristics of both epithelial cells and mesenchymal cells, and are known to have high drug resistance and metastatic potential by acquiring high stem cell capacity. In particular, EMT is further enhanced through factors such as transforming growth factor-beta (TGF-β) secreted from the tumor microenvironment (TME) and, as a result, various cell states with high plasticity appear. Due to the complexity of EMT, it has been very difficult to completely reverse the transitional process of the mesenchymal cancer cells to an epithelial cell state in which metastatic ability and drug resistance are eliminated while avoiding the EMT hybrid cell state with high metastatic ability and drug resistance.
Professor Kwang-Hyun Cho's research team established a mathematical model of the gene regulation network that governs the complex process of EMT, and then applied large-scale computer simulation analysis and complex system network control technology to identify and verify 'p53', 'SMAD4', and 'ERK1' and 'ERK 2' (collectively ERKs) through molecular cell experiments as the three key molecular targets that can transform lung cancer cells in the mesenchymal cell state, reversed back to an epithelial cell state that no longer demonstrates the ability to metastasize, while avoiding the EMT hybrid cell state.
In particular, by analyzing the molecular regulatory mechanism of the complex EMT process at the system level, the key pathways were identified that were linked to the positive feedback that plays an important role in completely returning cancer cells to an epithelial cell state in which metastatic ability and drug resistance are removed.
This discovery is significant in that it proved that mesenchymal cells can be reverted to the state of epithelial cells under conditions where TGF-β stimulation are present, like they are in the actual environment where cancer tissue forms in the human body.
Abnormal EMT in cancer cells leads to various malignant traits such as the migration and invasion of cancer cells, changes in responsiveness to chemotherapy treatment, enhanced stem cell function, and the dissemination of cancer. In particular, the acquisition of the metastatic ability of cancer cells is a key determinant factor for the prognosis of cancer patients. The EMT reversal technology in lung cancer cells developed in this research is a new anti-cancer treatment strategy that reprograms cancer cells to eliminate their high plasticity and metastatic potential and increase their responsiveness to chemotherapy.
Professor Kwang-Hyun Cho said, "By succeeding in reversing the state of lung cancer cells that acquired high metastatic traits and resistance to drugs and reverting them to a treatable epithelial cell state with renewed sensitivity to chemotherapy, the research findings propose a new strategy for treatments that can improve the prognosis of cancer patients.”
Professor Kwang-Hyun Cho's research team was the first to present the principle of reversal treatment to revert cancer cells to normal cells, following through with the announcement of the results of their study that reverted colon cancer cells to normal colon cells in January of 2020, and also presenting successful re-programming research where the most malignant basal type breast cancer cells turned into less-malignant luminal type breast cancer cells that were treatable with hormonal therapies in January of 2022. This latest research result is the third in the development of reversal technology where lung cancer cells that had acquired metastatic traits returned to a state in which their metastatic ability was removed and drug sensitivity was enhanced.
This research was carried out with support from the Ministry of Science and ICT and the National Research Foundation of Korea's Basic Research in Science & Engineering Program for Mid-Career Researchers.
< Figure 1. Construction of the mathematical model of the regulatory network to represent the EMT phenotype based on the interaction between various molecules related to EMT.
(A) Professor Kwang-Hyun Cho's research team investigated numerous literatures and databases related to complex EMT, and based on comparative analysis of cell line data showing epithelial and mesenchymal cell conditions, they extracted key signaling pathways related to EMT and built a mathematical model of regulatory network (B) By comparing the results of computer simulation analysis and the molecular cell experiments, it was verified how well the constructed mathematical model simulated the actual cellular phenomena. >
< Figure 2. Understanding of various EMT phenotypes through large-scale computer simulation analysis and complex system network control technology.
(A) Through computer simulation analysis and experiments, Professor Kwang-Hyun Cho's research team found that complete control of EMT is impossible with single-molecule control alone. In particular, through comparison of the relative stability of attractors, it was revealed that the cell state exhibiting EMT hybrid characteristics has unstable properties. (B), (C) Based on these results, Prof. Cho’s team identified two feedbacks (positive feedback consisting of Snail-miR-34 and ZEB1-miR-200) that play an important role in avoiding the EMT hybrid state that appeared in the TGF-β-ON state. It was found through computer simulation analysis that the two feedbacks restore relatively high stability when the excavated p53 and SMAD4 are regulated. In addition, molecular cell experiments demonstrated that the expression levels of E-cad and ZEB1, which are representative phenotypic markers of EMT, changed similarly to the expression profile in the epithelial cell state, despite the TGF-β-ON state. >
< Figure 3. Complex molecular network analysis and discovery of reprogramming molecular targets for intact elimination of EMT hybrid features.
(A) Controlling the expression of p53 and SMAD4 in lung cancer cell lines was expected to overcome drug resistance, but contrary to expectations, chemotherapy responsiveness was not restored. (B) Professor Kwang-Hyun Cho's research team additionally analyzed computer simulations, genome data, and experimental results and found that high expression levels of TWIST1 and EPCAM were related to drug resistance. (C) Prof. Cho’s team identified three key molecular targets: p53, SMAD4 and ERK1 & ERK2. (D), (E) Furthermore, they identified a key pathway that plays an important role in completely reversing into epithelial cells while avoiding EMT hybrid characteristics, and confirmed through network analysis and attractor analysis that high stability of the key pathway was restored when the proposed molecular target was controlled. >
< Figure 4. Verification through experiments with lung cancer cell lines.
When p53 was activated and SMAD4 and ERK1/2 were inhibited in lung cancer cell lines, (A), (B) E-cad protein expression increased and ZEB1 protein expression decreased, and (C) mesenchymal cell status including TWIST1 and EPCAM and gene expression of markers related to stem cell potential characteristics were completely inhibited. In addition, (D) it was confirmed that resistance to chemotherapy treatment was also overcome as the cell state was reversed by the regulated target. >
< Figure 5. A schematic representation of the research results.
Prof. Cho’s research team identified key molecular regulatory pathways to avoid high plasticity formed by abnormal EMT of cancer cells and reverse it to an epithelial cell state through systems biology research. From this analysis, a reprogramming molecular target that can reverse the state of mesenchymal cells with acquired invasiveness and drug resistance to the state of epithelial cells with restored drug responsiveness was discovered.
For lung cancer cells, when a drug that enhances the expression of p53, one of the molecular targets discovered, and inhibits the expression of SMAD4 and ERK1 & ERK2 is administered, the molecular network of genes in the state of mesenchymal cells is modified, eventually eliminating metastatic ability and it is reprogrammed to turn into epithelial cells without the resistance to chemotherapy treatments. >
2023.01.30 View 26866 -
 A KAIST Research Team Develops Diesel Reforming Catalyst Enabling Hydrogen Production for Future Mobile Fuel Cells
This catalyst capability allowing stable hydrogen production from commercial diesel is expected to be applied in mobile fuel cell systems in the future hydrogen economy
On August 16, a joint research team led by Professors Joongmyeon Bae and Kang Taek Lee of KAIST’s Department of Mechanical Engineering and Dr. Chan-Woo Lee of Korea Institute of Energy Research (KIER) announced the successful development of a highly active and durable reforming catalyst allowing hydrogen production from commercial diesel.
Fuel reforming is a hydrogen production technique that extracts hydrogen from hydrocarbons through catalytic reactions. Diesel, being a liquid fuel, has a high storage density for hydrogen and is easy to transport and store. There have therefore been continuous research efforts to apply hydrogel supply systems using diesel reformation in mobile fuel cells, such as for auxiliary power in heavy trucks or air-independent propulsion (AIP) systems in submarines.
However, diesel is a mixture of high hydrocarbons including long-chained paraffin, double-bonded olefin, and aromatic hydrocarbons with benzene groups, and it requires a highly active catalyst to effectively break them down. In addition, the catalyst must be extremely durable against caulking and sintering, as they are often the main causes of catalyst degradation. Such challenges have limited the use of diesel reformation technologies to date.
The joint research team successfully developed a highly active and durable diesel reforming catalyst through elution (a heat treatment method used to uniformly grow active metals retained in an oxide support as ions in the form of metal nanoparticles), forming alloy nanoparticles. The design was based on the fact that eluted nanoparticles strongly interact with the support, allowing a high degree of dispersion at high temperatures, and that producing an alloy from dissimilar metals can increase the performance of catalysts through a synergistic effect.
The research team introduced a solution combustion synthesis method to produce a multi-component catalyst with a trace amount of platinum (Pt) and ruthenium (Ru) penetrated into a ceria (CeO2) lattice, which is a structure commonly used as a support for catalysts in redox reactions. When exposed to a diesel reforming reaction environment, the catalyst induces Pt-Ru alloy nanoparticle formation upon Pt and Ru elution onto the support surface.
In addition to the catalyst analysis, the research team also succeeded in characterizing the behaviour of active metal elution and alloy formation from an energetic perspective using a density functional theory-based calculation. In a performance comparison test between the Pt-Ru alloy catalyst against existing single-metal catalysts, the reforming activity was shown to have improved, as it showed a 100% fuel conversion rate even at a low temperature (600oC, compared to the original 800oC). In a long-term durability test (800oC, 200 hours), the catalyst showed commercial stability by successfully producing hydrogen from commercial diesel without performance degradation.
The study was conducted by Ph.D. candidate Jaemyung Lee of KAIST’s Department of Mechanical Engineering as the first author. Ph.D. candidate Changho Yeon of KIER, Dr. Jiwoo Oh of KAIST’s Department of Mechanical Engineering, Dr. Gwangwoo Han of KIER, Ph.D. candidate Jeong Do Yoo of KAIST’s Department of Mechanical Engineering, and Dr. Hyung Joong Yun of the Korea Basic Science Institute contributed as co-authors. Dr. Chan-Woo Lee of KIER and Professors Kang Taek Lee and Joongmyeon Bae of KAIST’s Department of Mechanical Engineering contributed as corresponding authors. The research was published in the online version of Applied Catalysis B: Environmental (IF 24.319, JCR 0.93%) on June 17, under the title “Highly Active and Stable Catalyst with Exsolved PtRu Alloy Nanoparticles for Hydrogen Production via Commercial Diesel Reforming”.
Professor Joongmyeon Bae said, “The fact that hydrogen can be stably produced from commercial diesel makes this a very meaningful achievement, and we look forward to this technology contributing to the active introduction of mobile fuel cell systems in the early hydrogen economy.” He added, “Our approach to catalyst design may be applied not only to reforming reactions, but also in various other fields.”
This research was supported by the National Research Foundation of Korea through funding from the Ministry of Science, ICT and Future Planning.
Figure. Schematic diagram of high-performance diesel reforming catalyst with eluted platinum-ruthenium alloy nanoparticles and long-term durability verification experiment results for commercial diesel reforming reaction
2022.09.07 View 23607
A KAIST Research Team Develops Diesel Reforming Catalyst Enabling Hydrogen Production for Future Mobile Fuel Cells
This catalyst capability allowing stable hydrogen production from commercial diesel is expected to be applied in mobile fuel cell systems in the future hydrogen economy
On August 16, a joint research team led by Professors Joongmyeon Bae and Kang Taek Lee of KAIST’s Department of Mechanical Engineering and Dr. Chan-Woo Lee of Korea Institute of Energy Research (KIER) announced the successful development of a highly active and durable reforming catalyst allowing hydrogen production from commercial diesel.
Fuel reforming is a hydrogen production technique that extracts hydrogen from hydrocarbons through catalytic reactions. Diesel, being a liquid fuel, has a high storage density for hydrogen and is easy to transport and store. There have therefore been continuous research efforts to apply hydrogel supply systems using diesel reformation in mobile fuel cells, such as for auxiliary power in heavy trucks or air-independent propulsion (AIP) systems in submarines.
However, diesel is a mixture of high hydrocarbons including long-chained paraffin, double-bonded olefin, and aromatic hydrocarbons with benzene groups, and it requires a highly active catalyst to effectively break them down. In addition, the catalyst must be extremely durable against caulking and sintering, as they are often the main causes of catalyst degradation. Such challenges have limited the use of diesel reformation technologies to date.
The joint research team successfully developed a highly active and durable diesel reforming catalyst through elution (a heat treatment method used to uniformly grow active metals retained in an oxide support as ions in the form of metal nanoparticles), forming alloy nanoparticles. The design was based on the fact that eluted nanoparticles strongly interact with the support, allowing a high degree of dispersion at high temperatures, and that producing an alloy from dissimilar metals can increase the performance of catalysts through a synergistic effect.
The research team introduced a solution combustion synthesis method to produce a multi-component catalyst with a trace amount of platinum (Pt) and ruthenium (Ru) penetrated into a ceria (CeO2) lattice, which is a structure commonly used as a support for catalysts in redox reactions. When exposed to a diesel reforming reaction environment, the catalyst induces Pt-Ru alloy nanoparticle formation upon Pt and Ru elution onto the support surface.
In addition to the catalyst analysis, the research team also succeeded in characterizing the behaviour of active metal elution and alloy formation from an energetic perspective using a density functional theory-based calculation. In a performance comparison test between the Pt-Ru alloy catalyst against existing single-metal catalysts, the reforming activity was shown to have improved, as it showed a 100% fuel conversion rate even at a low temperature (600oC, compared to the original 800oC). In a long-term durability test (800oC, 200 hours), the catalyst showed commercial stability by successfully producing hydrogen from commercial diesel without performance degradation.
The study was conducted by Ph.D. candidate Jaemyung Lee of KAIST’s Department of Mechanical Engineering as the first author. Ph.D. candidate Changho Yeon of KIER, Dr. Jiwoo Oh of KAIST’s Department of Mechanical Engineering, Dr. Gwangwoo Han of KIER, Ph.D. candidate Jeong Do Yoo of KAIST’s Department of Mechanical Engineering, and Dr. Hyung Joong Yun of the Korea Basic Science Institute contributed as co-authors. Dr. Chan-Woo Lee of KIER and Professors Kang Taek Lee and Joongmyeon Bae of KAIST’s Department of Mechanical Engineering contributed as corresponding authors. The research was published in the online version of Applied Catalysis B: Environmental (IF 24.319, JCR 0.93%) on June 17, under the title “Highly Active and Stable Catalyst with Exsolved PtRu Alloy Nanoparticles for Hydrogen Production via Commercial Diesel Reforming”.
Professor Joongmyeon Bae said, “The fact that hydrogen can be stably produced from commercial diesel makes this a very meaningful achievement, and we look forward to this technology contributing to the active introduction of mobile fuel cell systems in the early hydrogen economy.” He added, “Our approach to catalyst design may be applied not only to reforming reactions, but also in various other fields.”
This research was supported by the National Research Foundation of Korea through funding from the Ministry of Science, ICT and Future Planning.
Figure. Schematic diagram of high-performance diesel reforming catalyst with eluted platinum-ruthenium alloy nanoparticles and long-term durability verification experiment results for commercial diesel reforming reaction
2022.09.07 View 23607 -
 PICASSO Technique Drives Biological Molecules into Technicolor
The new imaging approach brings current imaging colors from four to more than 15 for mapping overlapping proteins
Pablo Picasso’s surreal cubist artistic style shifted common features into unrecognizable scenes, but a new imaging approach bearing his namesake may elucidate the most complicated subject: the brain. Employing artificial intelligence to clarify spectral color blending of tiny molecules used to stain specific proteins and other items of research interest, the PICASSO technique, allows researchers to use more than 15 colors to image and parse our overlapping proteins.
The PICASSO developers, based in Korea, published their approach on May 5 in Nature Communications.
Fluorophores — the staining molecules — emit specific colors when excited by a light, but if more than four fluorophores are used, their emitted colors overlap and blend. Researchers previously developed techniques to correct this spectral overlap by precisely defining the matrix of mixed and unmixed images. This measurement depends on reference spectra, found by identifying clear images of only one fluorophore-stained specimen or of multiple, identically prepared specimens that only contain a single fluorophore each.
“Such reference spectra measurement could be complicated to perform in highly heterogeneous specimens, such as the brain, due to the highly varied emission spectra of fluorophores depending on the subregions from which the spectra were measured,” said co-corresponding author Young-Gyu Yoon, professor in the School of Electrical Engineering at KAIST. He explained that the subregions would each need their own spectra reference measurements, making for an inefficient, time-consuming process. “To address this problem, we developed an approach that does not require reference spectra measurements.”
The approach is the “Process of ultra-multiplexed Imaging of biomolecules viA the unmixing of the Signals of Spectrally Overlapping fluorophores,” also known as PICASSO. Ultra-multiplexed imaging refers to visualizing the numerous individual components of a unit. Like a cinema multiplex in which each theater plays a different movie, each protein in a cell has a different role. By staining with fluorophores, researchers can begin to understand those roles.
“We devised a strategy based on information theory; unmixing is performed by iteratively minimizing the mutual information between mixed images,” said co-corresponding author Jae-Byum Chang, professor in the Department of Materials Science and Engineering, KAIST. “This allows us to get away with the assumption that the spatial distribution of different proteins is mutually exclusive and enables accurate information unmixing.”
To demonstrate PICASSO’s capabilities, the researchers applied the technique to imaging a mouse brain. With a single round of staining, they performed 15-color multiplexed imaging of a mouse brain. Although small, mouse brains are still complex, multifaceted organs that can take significant resources to map. According to the researchers, PICASSO can improve the capabilities of other imaging techniques and allow for the use of even more fluorophore colors.
Using one such imaging technique in combination with PICASSO, the team achieved 45-color multiplexed imaging of the mouse brain in only three staining and imaging cycles, according to Yoon.
“PICASSO is a versatile tool for the multiplexed biomolecule imaging of cultured cells, tissue slices and clinical specimens,” Chang said. “We anticipate that PICASSO will be useful for a broad range of applications for which biomolecules’ spatial information is important. One such application the tool would be useful for is revealing the cellular heterogeneities of tumor microenvironments, especially the heterogeneous populations of immune cells, which are closely related to cancer prognoses and the efficacy of cancer therapies.”
The Samsung Research Funding & Incubation Center for Future Technology supported this work. Spectral imaging was performed at the Korea Basic Science Institute Western Seoul Center.
-PublicationJunyoung Seo, Yeonbo Sim, Jeewon Kim, Hyunwoo Kim, In Cho, Hoyeon Nam, Yong-Gyu Yoon, Jae-Byum Chang, “PICASSO allows ultra-multiplexed fluorescence imaging of spatiallyoverlapping proteins without reference spectra measurements,” May 5, Nature Communications (doi.org/10.1038/s41467-022-30168-z)
-ProfileProfessor Jae-Byum ChangDepartment of Materials Science and EngineeringCollege of EngineeringKAIST
Professor Young-Gyu YoonSchool of Electrical EngineeringCollege of EngineeringKAIST
2022.06.22 View 17182
PICASSO Technique Drives Biological Molecules into Technicolor
The new imaging approach brings current imaging colors from four to more than 15 for mapping overlapping proteins
Pablo Picasso’s surreal cubist artistic style shifted common features into unrecognizable scenes, but a new imaging approach bearing his namesake may elucidate the most complicated subject: the brain. Employing artificial intelligence to clarify spectral color blending of tiny molecules used to stain specific proteins and other items of research interest, the PICASSO technique, allows researchers to use more than 15 colors to image and parse our overlapping proteins.
The PICASSO developers, based in Korea, published their approach on May 5 in Nature Communications.
Fluorophores — the staining molecules — emit specific colors when excited by a light, but if more than four fluorophores are used, their emitted colors overlap and blend. Researchers previously developed techniques to correct this spectral overlap by precisely defining the matrix of mixed and unmixed images. This measurement depends on reference spectra, found by identifying clear images of only one fluorophore-stained specimen or of multiple, identically prepared specimens that only contain a single fluorophore each.
“Such reference spectra measurement could be complicated to perform in highly heterogeneous specimens, such as the brain, due to the highly varied emission spectra of fluorophores depending on the subregions from which the spectra were measured,” said co-corresponding author Young-Gyu Yoon, professor in the School of Electrical Engineering at KAIST. He explained that the subregions would each need their own spectra reference measurements, making for an inefficient, time-consuming process. “To address this problem, we developed an approach that does not require reference spectra measurements.”
The approach is the “Process of ultra-multiplexed Imaging of biomolecules viA the unmixing of the Signals of Spectrally Overlapping fluorophores,” also known as PICASSO. Ultra-multiplexed imaging refers to visualizing the numerous individual components of a unit. Like a cinema multiplex in which each theater plays a different movie, each protein in a cell has a different role. By staining with fluorophores, researchers can begin to understand those roles.
“We devised a strategy based on information theory; unmixing is performed by iteratively minimizing the mutual information between mixed images,” said co-corresponding author Jae-Byum Chang, professor in the Department of Materials Science and Engineering, KAIST. “This allows us to get away with the assumption that the spatial distribution of different proteins is mutually exclusive and enables accurate information unmixing.”
To demonstrate PICASSO’s capabilities, the researchers applied the technique to imaging a mouse brain. With a single round of staining, they performed 15-color multiplexed imaging of a mouse brain. Although small, mouse brains are still complex, multifaceted organs that can take significant resources to map. According to the researchers, PICASSO can improve the capabilities of other imaging techniques and allow for the use of even more fluorophore colors.
Using one such imaging technique in combination with PICASSO, the team achieved 45-color multiplexed imaging of the mouse brain in only three staining and imaging cycles, according to Yoon.
“PICASSO is a versatile tool for the multiplexed biomolecule imaging of cultured cells, tissue slices and clinical specimens,” Chang said. “We anticipate that PICASSO will be useful for a broad range of applications for which biomolecules’ spatial information is important. One such application the tool would be useful for is revealing the cellular heterogeneities of tumor microenvironments, especially the heterogeneous populations of immune cells, which are closely related to cancer prognoses and the efficacy of cancer therapies.”
The Samsung Research Funding & Incubation Center for Future Technology supported this work. Spectral imaging was performed at the Korea Basic Science Institute Western Seoul Center.
-PublicationJunyoung Seo, Yeonbo Sim, Jeewon Kim, Hyunwoo Kim, In Cho, Hoyeon Nam, Yong-Gyu Yoon, Jae-Byum Chang, “PICASSO allows ultra-multiplexed fluorescence imaging of spatiallyoverlapping proteins without reference spectra measurements,” May 5, Nature Communications (doi.org/10.1038/s41467-022-30168-z)
-ProfileProfessor Jae-Byum ChangDepartment of Materials Science and EngineeringCollege of EngineeringKAIST
Professor Young-Gyu YoonSchool of Electrical EngineeringCollege of EngineeringKAIST
2022.06.22 View 17182 -
 Five Projects Ranked in the Top 100 for National R&D Excellence
Five KAIST research projects were selected as the 2021 Top 100 for National R&D Excellence by the Ministry of Science and ICT and the Korea Institute of Science & Technology Evaluation and Planning.
The five projects are:-The development of E. coli that proliferates with only formic acid and carbon dioxide by Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering
-An original reverse aging technology that restores an old human skin cell into a younger one by Professor Kwang-Hyun Cho from the Department of Bio and Brain Engineering-The development of next-generation high-efficiency perovskite-silicon tandem solar cells by Professor Byungha Shin from the Department of Materials Science and Engineering-Research on the effects of ultrafine dust in the atmosphere has on energy consumption by Professor Jiyong Eom from the School of Business and Technology Management-Research on a molecular trigger that controls the phase transformation of bio materials by Professor Myungchul Kim from the Department of Bio and Brain Engineering
Started in 2006, an Evaluation Committee composed of experts in industries, universities, and research institutes has made the preliminary selections of the most outstanding research projects based on their significance as a scientific and technological development and their socioeconomic effects. The finalists went through an open public evaluation. The final 100 studies are from six fields: 18 from mechanics & materials, 26 from biology & marine sciences, 19 from ICT & electronics, 10 from interdisciplinary research, and nine from natural science and infrastructure.
The selected 100 studies will receive a certificate and an award plaque from the minister of MSIT as well as additional points for business and institutional evaluations according to appropriate regulations, and the selected researchers will be strongly recommended as candidates for national meritorious awards.
In particular, to help the 100 selected research projects become more accessible for the general public, their main contents will be provided in a free e-book ‘The Top 100 for National R&D Excellence of 2021’ that will be available from online booksellers.
2022.02.17 View 16634
Five Projects Ranked in the Top 100 for National R&D Excellence
Five KAIST research projects were selected as the 2021 Top 100 for National R&D Excellence by the Ministry of Science and ICT and the Korea Institute of Science & Technology Evaluation and Planning.
The five projects are:-The development of E. coli that proliferates with only formic acid and carbon dioxide by Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering
-An original reverse aging technology that restores an old human skin cell into a younger one by Professor Kwang-Hyun Cho from the Department of Bio and Brain Engineering-The development of next-generation high-efficiency perovskite-silicon tandem solar cells by Professor Byungha Shin from the Department of Materials Science and Engineering-Research on the effects of ultrafine dust in the atmosphere has on energy consumption by Professor Jiyong Eom from the School of Business and Technology Management-Research on a molecular trigger that controls the phase transformation of bio materials by Professor Myungchul Kim from the Department of Bio and Brain Engineering
Started in 2006, an Evaluation Committee composed of experts in industries, universities, and research institutes has made the preliminary selections of the most outstanding research projects based on their significance as a scientific and technological development and their socioeconomic effects. The finalists went through an open public evaluation. The final 100 studies are from six fields: 18 from mechanics & materials, 26 from biology & marine sciences, 19 from ICT & electronics, 10 from interdisciplinary research, and nine from natural science and infrastructure.
The selected 100 studies will receive a certificate and an award plaque from the minister of MSIT as well as additional points for business and institutional evaluations according to appropriate regulations, and the selected researchers will be strongly recommended as candidates for national meritorious awards.
In particular, to help the 100 selected research projects become more accessible for the general public, their main contents will be provided in a free e-book ‘The Top 100 for National R&D Excellence of 2021’ that will be available from online booksellers.
2022.02.17 View 16634 -
 Hydrogel-Based Flexible Brain-Machine Interface
The interface is easy to insert into the body when dry, but behaves ‘stealthily’ inside the brain when wet
Professor Seongjun Park’s research team and collaborators revealed a newly developed hydrogel-based flexible brain-machine interface. To study the structure of the brain or to identify and treat neurological diseases, it is crucial to develop an interface that can stimulate the brain and detect its signals in real time. However, existing neural interfaces are mechanically and chemically different from real brain tissue. This causes foreign body response and forms an insulating layer (glial scar) around the interface, which shortens its lifespan.
To solve this problem, the research team developed a ‘brain-mimicking interface’ by inserting a custom-made multifunctional fiber bundle into the hydrogel body. The device is composed not only of an optical fiber that controls specific nerve cells with light in order to perform optogenetic procedures, but it also has an electrode bundle to read brain signals and a microfluidic channel to deliver drugs to the brain.
The interface is easy to insert into the body when dry, as hydrogels become solid. But once in the body, the hydrogel will quickly absorb body fluids and resemble the properties of its surrounding tissues, thereby minimizing foreign body response.
The research team applied the device on animal models, and showed that it was possible to detect neural signals for up to six months, which is far beyond what had been previously recorded. It was also possible to conduct long-term optogenetic and behavioral experiments on freely moving mice with a significant reduction in foreign body responses such as glial and immunological activation compared to existing devices.
“This research is significant in that it was the first to utilize a hydrogel as part of a multifunctional neural interface probe, which increased its lifespan dramatically,” said Professor Park. “With our discovery, we look forward to advancements in research on neurological disorders like Alzheimer’s or Parkinson’s disease that require long-term observation.”
The research was published in Nature Communications on June 8, 2021. (Title: Adaptive and multifunctional hydrogel hybrid probes for long-term sensing and modulation of neural activity) The study was conducted jointly with an MIT research team composed of Professor Polina Anikeeva, Professor Xuanhe Zhao, and Dr. Hyunwoo Yook.
This research was supported by the National Research Foundation (NRF) grant for emerging research, Korea Medical Device Development Fund, KK-JRC Smart Project, KAIST Global Initiative Program, and Post-AI Project.
-PublicationPark, S., Yuk, H., Zhao, R. et al. Adaptive and multifunctional hydrogel hybrid probes for long-term sensing and modulation of neural activity. Nat Commun 12, 3435 (2021). https://doi.org/10.1038/s41467-021-23802-9
-ProfileProfessor Seongjun ParkBio and Neural Interfaces LaboratoryDepartment of Bio and Brain EngineeringKAIST
2021.07.13 View 18464
Hydrogel-Based Flexible Brain-Machine Interface
The interface is easy to insert into the body when dry, but behaves ‘stealthily’ inside the brain when wet
Professor Seongjun Park’s research team and collaborators revealed a newly developed hydrogel-based flexible brain-machine interface. To study the structure of the brain or to identify and treat neurological diseases, it is crucial to develop an interface that can stimulate the brain and detect its signals in real time. However, existing neural interfaces are mechanically and chemically different from real brain tissue. This causes foreign body response and forms an insulating layer (glial scar) around the interface, which shortens its lifespan.
To solve this problem, the research team developed a ‘brain-mimicking interface’ by inserting a custom-made multifunctional fiber bundle into the hydrogel body. The device is composed not only of an optical fiber that controls specific nerve cells with light in order to perform optogenetic procedures, but it also has an electrode bundle to read brain signals and a microfluidic channel to deliver drugs to the brain.
The interface is easy to insert into the body when dry, as hydrogels become solid. But once in the body, the hydrogel will quickly absorb body fluids and resemble the properties of its surrounding tissues, thereby minimizing foreign body response.
The research team applied the device on animal models, and showed that it was possible to detect neural signals for up to six months, which is far beyond what had been previously recorded. It was also possible to conduct long-term optogenetic and behavioral experiments on freely moving mice with a significant reduction in foreign body responses such as glial and immunological activation compared to existing devices.
“This research is significant in that it was the first to utilize a hydrogel as part of a multifunctional neural interface probe, which increased its lifespan dramatically,” said Professor Park. “With our discovery, we look forward to advancements in research on neurological disorders like Alzheimer’s or Parkinson’s disease that require long-term observation.”
The research was published in Nature Communications on June 8, 2021. (Title: Adaptive and multifunctional hydrogel hybrid probes for long-term sensing and modulation of neural activity) The study was conducted jointly with an MIT research team composed of Professor Polina Anikeeva, Professor Xuanhe Zhao, and Dr. Hyunwoo Yook.
This research was supported by the National Research Foundation (NRF) grant for emerging research, Korea Medical Device Development Fund, KK-JRC Smart Project, KAIST Global Initiative Program, and Post-AI Project.
-PublicationPark, S., Yuk, H., Zhao, R. et al. Adaptive and multifunctional hydrogel hybrid probes for long-term sensing and modulation of neural activity. Nat Commun 12, 3435 (2021). https://doi.org/10.1038/s41467-021-23802-9
-ProfileProfessor Seongjun ParkBio and Neural Interfaces LaboratoryDepartment of Bio and Brain EngineeringKAIST
2021.07.13 View 18464