Seyun+Kim
-
 Study Identifies the Novel Molecular Signal for Triggering Septic Shock
Professor Seyun Kim’s team at the Department of Biological Sciences reported the mechanism by which cellular signaling transduction networks are precisely controlled in mediating innate immune responses, such as sepsis, by the enzyme IPMK (Inositol polyphosphate multikinase) which is essential for inositol biosynthesis metabolism.
In collaboration with Professor Hyun Seong Roh at Seoul National University, the study’s first author, Eunha Kim, a Ph.D. candidate in Department of Biological Sciences, performed a series of cellular, biochemical, and physiological experiments searching for the new function of IPMK enzymes in macrophages. The research findings were published in Science Advances on April 21.
Professor Kim’s team has been investigating various inositol metabolites and their biosynthesis metabolism for several years and has multilaterally identified the signaling actions of IPMK for controlling cellular growth and energy homeostasis.
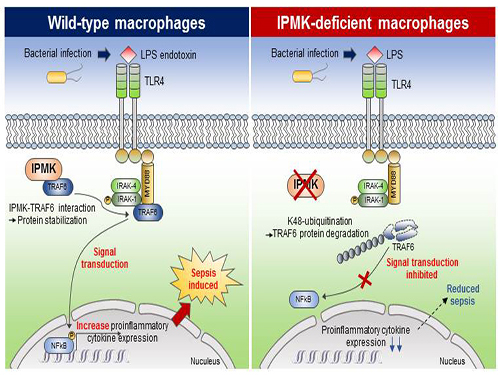
This research showed that the specific deletion of IPMK enzymes in macrophages could significantly reduce levels of inflammation and increase survival rates in mice when they were challenged by microbial septic shock and endotoxins. This suggests a role for IPMK enzymes in mediating innate inflammatory responses that are directly related to a host’s defense against pathogenic bacterial infection.
The team further discovered that IPMK enzymes directly bind to TRAF6 proteins, a key player in immune signaling, thus protecting TRAF6 proteins from ubiquitination reactions that are involved in protein degradation. In addition, Kim and his colleagues successfully verified this IPMK-dependent immune control by employing short peptides which can specifically interfere with the binding between IPMK enzymes and TRAF6 proteins in macrophage cells.
This research revealed a novel function of IPMK enzymes in the fine tuning of innate immune signaling networks, suggesting a new direction for developing therapeutics targeting serious medical conditions such as neuroinflammation, type 2 diabetes, as well as polymicrobial sepsis that are developed from uncontrolled host immune responses. This research was funded by the Ministry of Science, ICT and Future Planning.
(Figure: Deletion of IPMK (inositol polyphosphate multikinase) in macrophages reduces the stability of TRAF6 protein which is the key to innate immune signaling, thereby blocking excessive inflammation in response to pathological bacterial infection.)
2017.05.11 View 9959
Study Identifies the Novel Molecular Signal for Triggering Septic Shock
Professor Seyun Kim’s team at the Department of Biological Sciences reported the mechanism by which cellular signaling transduction networks are precisely controlled in mediating innate immune responses, such as sepsis, by the enzyme IPMK (Inositol polyphosphate multikinase) which is essential for inositol biosynthesis metabolism.
In collaboration with Professor Hyun Seong Roh at Seoul National University, the study’s first author, Eunha Kim, a Ph.D. candidate in Department of Biological Sciences, performed a series of cellular, biochemical, and physiological experiments searching for the new function of IPMK enzymes in macrophages. The research findings were published in Science Advances on April 21.
Professor Kim’s team has been investigating various inositol metabolites and their biosynthesis metabolism for several years and has multilaterally identified the signaling actions of IPMK for controlling cellular growth and energy homeostasis.
This research showed that the specific deletion of IPMK enzymes in macrophages could significantly reduce levels of inflammation and increase survival rates in mice when they were challenged by microbial septic shock and endotoxins. This suggests a role for IPMK enzymes in mediating innate inflammatory responses that are directly related to a host’s defense against pathogenic bacterial infection.
The team further discovered that IPMK enzymes directly bind to TRAF6 proteins, a key player in immune signaling, thus protecting TRAF6 proteins from ubiquitination reactions that are involved in protein degradation. In addition, Kim and his colleagues successfully verified this IPMK-dependent immune control by employing short peptides which can specifically interfere with the binding between IPMK enzymes and TRAF6 proteins in macrophage cells.
This research revealed a novel function of IPMK enzymes in the fine tuning of innate immune signaling networks, suggesting a new direction for developing therapeutics targeting serious medical conditions such as neuroinflammation, type 2 diabetes, as well as polymicrobial sepsis that are developed from uncontrolled host immune responses. This research was funded by the Ministry of Science, ICT and Future Planning.
(Figure: Deletion of IPMK (inositol polyphosphate multikinase) in macrophages reduces the stability of TRAF6 protein which is the key to innate immune signaling, thereby blocking excessive inflammation in response to pathological bacterial infection.)
2017.05.11 View 9959 -
 Professor Seyun Kim Identifies a Neuron Signal Controlling Molecule
A research team led by Professor Seyun Kim of the Department of Biological Sciences at KAIST has identified inositol pyrophosphates as the molecule that strongly controls neuron signaling via synaptotagmin.
Professors Tae-Young Yoon of Yonsei University’s Y-IBS and Sung-Hyun Kim of Kyung Hee University’s Department of Biomedical Science also joined the team.
The results were published in the Proceedings of the National Academy of Sciences of the United States of America (PNAS) on June 30, 2016.
This interdisciplinary research project was conducted by six research teams from four different countries and covered a wide scope of academic fields, from neurobiology to super resolution optic imaging.
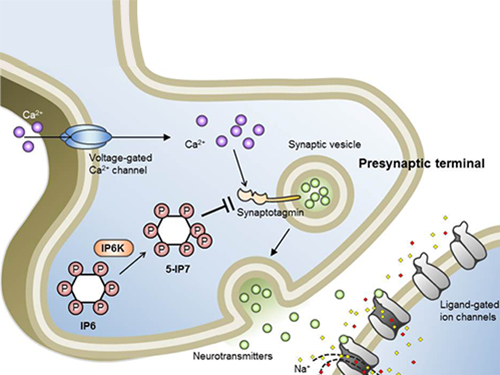
Inositol pyrophosphates such as 5-diphosphoinositol pentakisphos-phate (5-IP7), which naturally occur in corns and beans, are essential metabolites in the body. In particular, inositol hexakisphosphate (IP6) has anti-cancer properties and is thought to have an important role in cell signaling.
Inositol pentakisphosphate (IP7) differs from IP6 by having an additional phosphate group, which was first discovered 20 years ago. IP7 has recently been identified as playing a key role in diabetes and obesity.
Psychopathy and neurodegenerative diseases are known to result from the disrupted balance of inositol pyrophosphates. However, the role and the mechanism of action of IP7 in brain neurons and nerve transmission remained unknown.
Professor Kim’s team has worked on inositol pyrophosphates for several years and discovered that very small quantities of IP7 control cell-signaling transduction. Professor Yoon of Yonsei University identified IP7 as a much stronger inhibitor of neuron signaling compared to IP6. In particular, IP7 directly suppresses synaptotagmin, one of the key proteins in neuron signaling. Moreover, Professor Kim of Kyung Hee University observed IP7 inhibition in sea horse neurons.
Together, the joint research team identified inositol pyrophosphates as the key switch metabolite of brain-signaling transduction.
The researchers hope that future research on synaptotagmin and IP7 will reveal the mechanism of neuron-signal transduction and thus enable the treatment of neurological disorders.
These research findings were the result of cooperation of various science and technology institutes: KAIST, Yonsei-IBS (Institute for Basic Science), Kyung Hee University, Sungkyunkwan University, KIST, University of Zurich in Switzerland, and Albert-Ludwigs-University Freiburg in Germany.
Schematic Image of Controlling the Synaptic Exocytotic Pathway by 5-IP7 , Helping the Understanding of the Signaling Mechanisms of Inositol Pyrophosphates
2016.07.21 View 12857
Professor Seyun Kim Identifies a Neuron Signal Controlling Molecule
A research team led by Professor Seyun Kim of the Department of Biological Sciences at KAIST has identified inositol pyrophosphates as the molecule that strongly controls neuron signaling via synaptotagmin.
Professors Tae-Young Yoon of Yonsei University’s Y-IBS and Sung-Hyun Kim of Kyung Hee University’s Department of Biomedical Science also joined the team.
The results were published in the Proceedings of the National Academy of Sciences of the United States of America (PNAS) on June 30, 2016.
This interdisciplinary research project was conducted by six research teams from four different countries and covered a wide scope of academic fields, from neurobiology to super resolution optic imaging.
Inositol pyrophosphates such as 5-diphosphoinositol pentakisphos-phate (5-IP7), which naturally occur in corns and beans, are essential metabolites in the body. In particular, inositol hexakisphosphate (IP6) has anti-cancer properties and is thought to have an important role in cell signaling.
Inositol pentakisphosphate (IP7) differs from IP6 by having an additional phosphate group, which was first discovered 20 years ago. IP7 has recently been identified as playing a key role in diabetes and obesity.
Psychopathy and neurodegenerative diseases are known to result from the disrupted balance of inositol pyrophosphates. However, the role and the mechanism of action of IP7 in brain neurons and nerve transmission remained unknown.
Professor Kim’s team has worked on inositol pyrophosphates for several years and discovered that very small quantities of IP7 control cell-signaling transduction. Professor Yoon of Yonsei University identified IP7 as a much stronger inhibitor of neuron signaling compared to IP6. In particular, IP7 directly suppresses synaptotagmin, one of the key proteins in neuron signaling. Moreover, Professor Kim of Kyung Hee University observed IP7 inhibition in sea horse neurons.
Together, the joint research team identified inositol pyrophosphates as the key switch metabolite of brain-signaling transduction.
The researchers hope that future research on synaptotagmin and IP7 will reveal the mechanism of neuron-signal transduction and thus enable the treatment of neurological disorders.
These research findings were the result of cooperation of various science and technology institutes: KAIST, Yonsei-IBS (Institute for Basic Science), Kyung Hee University, Sungkyunkwan University, KIST, University of Zurich in Switzerland, and Albert-Ludwigs-University Freiburg in Germany.
Schematic Image of Controlling the Synaptic Exocytotic Pathway by 5-IP7 , Helping the Understanding of the Signaling Mechanisms of Inositol Pyrophosphates
2016.07.21 View 12857