research
- The somatic nuclear protein kinase VRK-1 increases the worm’s lifespan through AMPK activation, and this mechanism can be applied to promoting human longevity, the study reveals. -

< Sangsoon Park (left) and Professor Seung-Jae V. Lee (right) >
KAIST researchers have been able to dial up and down creatures’ lifespans by altering the activity of proteins found in roundworm cells that tell them to convert sugar into energy when their cellular energy is running low. Humans also have these proteins, offering up the intriguing possibilities for developing longevity-promoting drugs. These new findings were published on July 1 in Science Advances.
The roundworm Caenorhabditis elegans (C. elegans), a millimeter-long nematode commonly used in lab testing, enjoyed a boost in its lifespan when researchers tinkered with a couple of proteins involved in monitoring the energy use by its cells.
The proteins VRK-1 and AMPK work in tandem in roundworm cells, with the former telling the latter to get to work by sticking a phosphate molecule, composed of one phosphorus and four oxygen atoms, on it. In turn, AMPK’s role is to monitor energy levels in cells, when cellular energy is running low. In essence, VRK-1 regulates AMPK, and AMPK regulates the cellular energy status.
Using a range of different biological research tools, including introducing foreign genes into the worm, a group of researchers led by Professor Seung-Jae V. Lee from the Department of Biological Sciences at KAIST were able to dial up and down the activity of the gene that tells cells to produce the VRK-1 protein. This gene has remained pretty much unchanged throughout evolution. Most complex organisms have this same gene, including humans.
Lead author of the study Sangsoon Park and his colleagues confirmed that the overexpression, or increased production, of the VRK-1 protein boosted the lifespan of the C. elegans, which normally lives just two to three weeks, and the inhibition of VRK-1 production reduced its lifespan.
The research team found that the activity of the VRK-1-to-AMPK cellular-energy monitoring process is increased in low cellular energy status by reduced mitochondrial respiration, the set of metabolic chemical reactions that make use of the oxygen the worm breathes to convert macronutrients from food into the energy “currency” that cells spend to do everything they need to do.
It is already known that mitochondria, the energy-producing engine rooms in cells, play a crucial role in aging, and declines in the functioning of mitochondria are associated with age-related diseases. At the same time, the mild inhibition of mitochondrial respiration has been shown to promote longevity in a range of species, including flies and mammals.
When the research team performed similar tinkering with cultured human cells, they found they could also replicate this ramping up and down of the VRK-1-to-AMPK process that occurs in roundworms.
“This raises the intriguing possibility that VRK-1 also functions as a factor in governing human longevity, and so perhaps we can start developing longevity-promoting drugs that alter the activity of VRK-1,” explained Professor Lee.
At the very least, the research points us in an interesting direction for investigating new therapeutic strategies to combat metabolic disorders by targeting the modulation of VRK-1. Metabolic disorders involve the disruption of chemical reactions in the body, including diseases of the mitochondria.
But before metabolic disorder therapeutics or longevity drugs can be contemplated by scientists, further research still needs to be carried out to better understand how VRK-1 works to activate AMPK, as well as figure out the precise mechanics of how AMPK controls cellular energy.
This work was supported by the National Research Foundation (NRF), and the Ministry of Science and ICT (MSIT) of Korea.
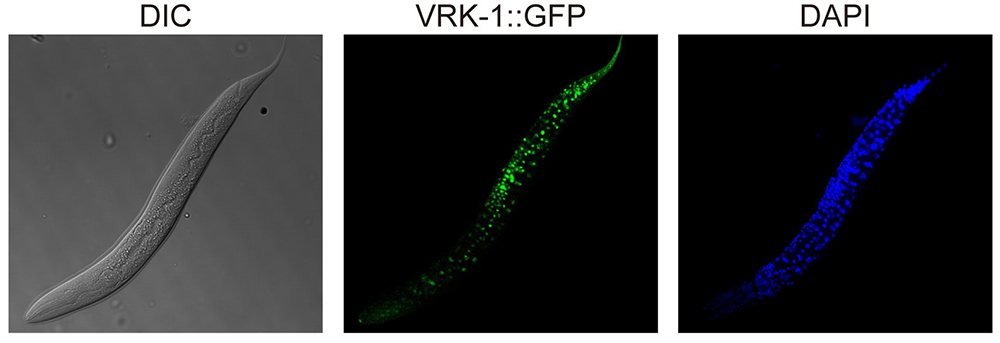
< Image. VRK-1 that was visualized by tagging with green fluorescence protein (GFP) in C. elegans. >
Image credit: Seung-Jae V. LEE, KAIST.
Image usage restrictions: News organizations may use or redistribute this image, with proper attribution, as part of news coverage of this paper only.
Publication:
Park, S., et al. (2020) ‘VRK-1 extends life span by activation of AMPK via phosphorylation’. Science Advances, Volume 6. No. 27, eaaw7824. Available online at https://doi.org/10.1126/sciadv.aaw7824
Profile: Seung-Jae V. Lee, Ph.D.
Professor
seungjaevlee@kaist.ac.kr
https://sites.google.com/view/mgakaist
Molecular Genetics of Aging Laboratory
Department of Biological Sciences
Korea Advanced Institute of Science and Technology (KAIST)
https://www.kaist.ac.kr
Daejeon 34141, Korea
(END)
-
research Longevity mediated by suppressing age-associated circRNA
< (Back row from left) Prof. Yoon Ki Kim, Prof. Seung-Jae V. Lee, and Gwangrog Lee; (Front row from left) Dr. Sung Ho Boo, Sieun S. Kim, Seokjin Ham, and (top) Donghun Lee > Cells in our bodies produce RNA based on genetic information stored in DNA, and RNA serves as a blueprint for making proteins. Researchers at our university have discovered a new phenomenon: removing 'circular RNA' that accumulates in cells as we age can slow down aging and extend lifespan. This study provides cru
2026-03-18 -
research KAIST Develops mRNA Platform That Remains Effective Even in Aging and Obesity
<(From Left) Dr. Subin Yoon, Ph.D candidate Hyeonggon Cho, Prof. Jae-Hwan Nam, Prof. Young-suk Lee> Since the COVID-19 pandemic, mRNA vaccines have gained attention as a next-generation pharmaceutical technology. mRNA therapeutics work by delivering genetic instructions that enable cells to produce specific proteins for therapeutic effects. However, their efficacy has been reported to decline in elderly individuals or patients with obesity. To address this limitation, Korean researchers
2026-03-10 -
event MICCAI 2025 Eve KAIST Day Successfully Held
< Scene of the KAIST Day Symposium Lectures > KAIST announced on the September 23rd that the 'KAIST Day' special symposium, held on the eve of 'MICCAI 2025' at the Jeong Geun-mo Conference Hall of the KAIST Academic and Cultural Center on September 22, was successfully held with the attendance of more than 30 overseas scholars and 200 domestic researchers and students. This event was a special program prepared to commemorate the hosting of MICCAI 2025 (The 28th International Confe
2025-09-23 -
research KAIST succeeds in controlling complex altered gene networks to restore them to normal
Previously, research on controlling gene networks has been carried out based on a single stimulus-response of cells. More recently, studies have been proposed to precisely analyze complex gene networks to identify control targets. A KAIST research team has succeeded in developing a universal technology that identifies gene control targets in altered cellular gene networks and restores them. This achievement is expected to be widely applied to new anticancer therapies such as cancer reversibili
2025-08-29 -
research KAIST Develops Customized Tactile Sensor That Can Detect Light Breath, Pressure and Sound
< Photo 1. (From left) Professor Inkyu Park of KAIST Department of Mechanical Engineering (ME), Dr. Jungrak Choi of ETRI, Ph.D. Candidate Donho Lee and M.S. Graduate Chankyu Han of KAIST ME > When a robot grabs an object or a medical device detects a pulse, the tactile sensor is the technology that senses pressure like a fingertip. Existing sensors had disadvantages, such as slow responses or declining accuracy after repeated use, but Korean researchers have succeeded in developing a
2025-06-23