TR
-
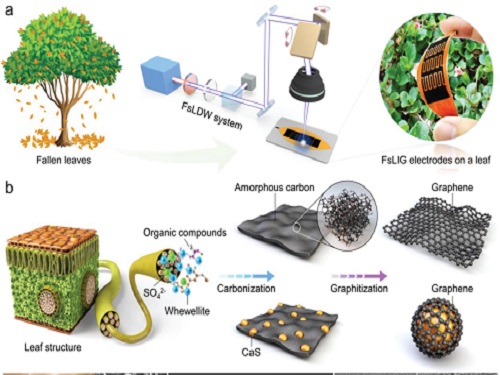 Eco-Friendly Micro-Supercapacitors Using Fallen Leaves
Green micro-supercapacitors on a single leaf could easily be applied in wearable electronics, smart houses, and IoTs
A KAIST research team has developed graphene-inorganic-hybrid micro-supercapacitors made of fallen leaves using femtosecond laser direct writing. The rapid development of wearable electronics requires breakthrough innovations in flexible energy storage devices in which micro-supercapacitors have drawn a great deal of interest due to their high power density, long lifetimes, and short charging times. Recently, there has been an enormous increase in waste batteries owing to the growing demand and the shortened replacement cycle in consumer electronics. The safety and environmental issues involved in the collection, recycling, and processing of such waste batteries are creating a number of challenges.
Forests cover about 30 percent of the Earth’s surface and produce a huge amount of fallen leaves. This naturally occurring biomass comes in large quantities and is completely biodegradable, which makes it an attractive sustainable resource. Nevertheless, if the fallen leaves are left neglected instead of being used efficiently, they can contribute to fire hazards, air pollution, and global warming.
To solve both problems at once, a research team led by Professor Young-Jin Kim from the Department of Mechanical Engineering and Dr. Hana Yoon from the Korea Institute of Energy Research developed a novel technology that can create 3D porous graphene microelectrodes with high electrical conductivity by irradiating femtosecond laser pulses on the leaves in ambient air. This one-step fabrication does not require any additional materials or pre-treatment.
They showed that this technique could quickly and easily produce porous graphene electrodes at a low price, and demonstrated potential applications by fabricating graphene micro-supercapacitors to power an LED and an electronic watch. These results open up a new possibility for the mass production of flexible and green graphene-based electronic devices.
Professor Young-Jin Kim said, “Leaves create forest biomass that comes in unmanageable quantities, so using them for next-generation energy storage devices makes it possible for us to reuse waste resources, thereby establishing a virtuous cycle.”
This research was published in Advanced Functional Materials last month and was sponsored by the Ministry of Agriculture Food and Rural Affairs, the Korea Forest Service, and the Korea Institute of Energy Research.
-Publication
Truong-Son Dinh Le, Yeong A. Lee, Han Ku Nam, Kyu Yeon Jang, Dongwook Yang, Byunggi Kim, Kanghoon Yim, Seung Woo Kim, Hana Yoon, and Young-jin Kim, “Green Flexible Graphene-Inorganic-Hybrid Micro-Supercapacitors Made of Fallen Leaves Enabled by Ultrafast Laser Pulses," December 05, 2021, Advanced Functional Materials (doi.org/10.1002/adfm.202107768)
-ProfileProfessor Young-Jin KimUltra-Precision Metrology and Manufacturing (UPM2) LaboratoryDepartment of Mechanical EngineeringKAIST
2022.01.27 View 13947
Eco-Friendly Micro-Supercapacitors Using Fallen Leaves
Green micro-supercapacitors on a single leaf could easily be applied in wearable electronics, smart houses, and IoTs
A KAIST research team has developed graphene-inorganic-hybrid micro-supercapacitors made of fallen leaves using femtosecond laser direct writing. The rapid development of wearable electronics requires breakthrough innovations in flexible energy storage devices in which micro-supercapacitors have drawn a great deal of interest due to their high power density, long lifetimes, and short charging times. Recently, there has been an enormous increase in waste batteries owing to the growing demand and the shortened replacement cycle in consumer electronics. The safety and environmental issues involved in the collection, recycling, and processing of such waste batteries are creating a number of challenges.
Forests cover about 30 percent of the Earth’s surface and produce a huge amount of fallen leaves. This naturally occurring biomass comes in large quantities and is completely biodegradable, which makes it an attractive sustainable resource. Nevertheless, if the fallen leaves are left neglected instead of being used efficiently, they can contribute to fire hazards, air pollution, and global warming.
To solve both problems at once, a research team led by Professor Young-Jin Kim from the Department of Mechanical Engineering and Dr. Hana Yoon from the Korea Institute of Energy Research developed a novel technology that can create 3D porous graphene microelectrodes with high electrical conductivity by irradiating femtosecond laser pulses on the leaves in ambient air. This one-step fabrication does not require any additional materials or pre-treatment.
They showed that this technique could quickly and easily produce porous graphene electrodes at a low price, and demonstrated potential applications by fabricating graphene micro-supercapacitors to power an LED and an electronic watch. These results open up a new possibility for the mass production of flexible and green graphene-based electronic devices.
Professor Young-Jin Kim said, “Leaves create forest biomass that comes in unmanageable quantities, so using them for next-generation energy storage devices makes it possible for us to reuse waste resources, thereby establishing a virtuous cycle.”
This research was published in Advanced Functional Materials last month and was sponsored by the Ministry of Agriculture Food and Rural Affairs, the Korea Forest Service, and the Korea Institute of Energy Research.
-Publication
Truong-Son Dinh Le, Yeong A. Lee, Han Ku Nam, Kyu Yeon Jang, Dongwook Yang, Byunggi Kim, Kanghoon Yim, Seung Woo Kim, Hana Yoon, and Young-jin Kim, “Green Flexible Graphene-Inorganic-Hybrid Micro-Supercapacitors Made of Fallen Leaves Enabled by Ultrafast Laser Pulses," December 05, 2021, Advanced Functional Materials (doi.org/10.1002/adfm.202107768)
-ProfileProfessor Young-Jin KimUltra-Precision Metrology and Manufacturing (UPM2) LaboratoryDepartment of Mechanical EngineeringKAIST
2022.01.27 View 13947 -
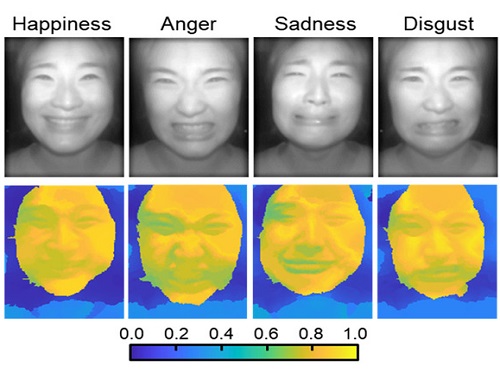 AI Light-Field Camera Reads 3D Facial Expressions
Machine-learned, light-field camera reads facial expressions from high-contrast illumination invariant 3D facial images
A joint research team led by Professors Ki-Hun Jeong and Doheon Lee from the KAIST Department of Bio and Brain Engineering reported the development of a technique for facial expression detection by merging near-infrared light-field camera techniques with artificial intelligence (AI) technology.
Unlike a conventional camera, the light-field camera contains micro-lens arrays in front of the image sensor, which makes the camera small enough to fit into a smart phone, while allowing it to acquire the spatial and directional information of the light with a single shot. The technique has received attention as it can reconstruct images in a variety of ways including multi-views, refocusing, and 3D image acquisition, giving rise to many potential applications.
However, the optical crosstalk between shadows caused by external light sources in the environment and the micro-lens has limited existing light-field cameras from being able to provide accurate image contrast and 3D reconstruction.
The joint research team applied a vertical-cavity surface-emitting laser (VCSEL) in the near-IR range to stabilize the accuracy of 3D image reconstruction that previously depended on environmental light. When an external light source is shone on a face at 0-, 30-, and 60-degree angles, the light field camera reduces 54% of image reconstruction errors. Additionally, by inserting a light-absorbing layer for visible and near-IR wavelengths between the micro-lens arrays, the team could minimize optical crosstalk while increasing the image contrast by 2.1 times.
Through this technique, the team could overcome the limitations of existing light-field cameras and was able to develop their NIR-based light-field camera (NIR-LFC), optimized for the 3D image reconstruction of facial expressions. Using the NIR-LFC, the team acquired high-quality 3D reconstruction images of facial expressions expressing various emotions regardless of the lighting conditions of the surrounding environment.
The facial expressions in the acquired 3D images were distinguished through machine learning with an average of 85% accuracy – a statistically significant figure compared to when 2D images were used. Furthermore, by calculating the interdependency of distance information that varies with facial expression in 3D images, the team could identify the information a light-field camera utilizes to distinguish human expressions.
Professor Ki-Hun Jeong said, “The sub-miniature light-field camera developed by the research team has the potential to become the new platform to quantitatively analyze the facial expressions and emotions of humans.” To highlight the significance of this research, he added, “It could be applied in various fields including mobile healthcare, field diagnosis, social cognition, and human-machine interactions.”
This research was published in Advanced Intelligent Systems online on December 16, under the title, “Machine-Learned Light-field Camera that Reads Facial Expression from High-Contrast and Illumination Invariant 3D Facial Images.” This research was funded by the Ministry of Science and ICT and the Ministry of Trade, Industry and Energy.
-Publication“Machine-learned light-field camera that reads fascial expression from high-contrast and illumination invariant 3D facial images,” Sang-In Bae, Sangyeon Lee, Jae-Myeong Kwon, Hyun-Kyung Kim. Kyung-Won Jang, Doheon Lee, Ki-Hun Jeong, Advanced Intelligent Systems, December 16, 2021 (doi.org/10.1002/aisy.202100182)
ProfileProfessor Ki-Hun JeongBiophotonic LaboratoryDepartment of Bio and Brain EngineeringKAIST
Professor Doheon LeeDepartment of Bio and Brain EngineeringKAIST
2022.01.21 View 13921
AI Light-Field Camera Reads 3D Facial Expressions
Machine-learned, light-field camera reads facial expressions from high-contrast illumination invariant 3D facial images
A joint research team led by Professors Ki-Hun Jeong and Doheon Lee from the KAIST Department of Bio and Brain Engineering reported the development of a technique for facial expression detection by merging near-infrared light-field camera techniques with artificial intelligence (AI) technology.
Unlike a conventional camera, the light-field camera contains micro-lens arrays in front of the image sensor, which makes the camera small enough to fit into a smart phone, while allowing it to acquire the spatial and directional information of the light with a single shot. The technique has received attention as it can reconstruct images in a variety of ways including multi-views, refocusing, and 3D image acquisition, giving rise to many potential applications.
However, the optical crosstalk between shadows caused by external light sources in the environment and the micro-lens has limited existing light-field cameras from being able to provide accurate image contrast and 3D reconstruction.
The joint research team applied a vertical-cavity surface-emitting laser (VCSEL) in the near-IR range to stabilize the accuracy of 3D image reconstruction that previously depended on environmental light. When an external light source is shone on a face at 0-, 30-, and 60-degree angles, the light field camera reduces 54% of image reconstruction errors. Additionally, by inserting a light-absorbing layer for visible and near-IR wavelengths between the micro-lens arrays, the team could minimize optical crosstalk while increasing the image contrast by 2.1 times.
Through this technique, the team could overcome the limitations of existing light-field cameras and was able to develop their NIR-based light-field camera (NIR-LFC), optimized for the 3D image reconstruction of facial expressions. Using the NIR-LFC, the team acquired high-quality 3D reconstruction images of facial expressions expressing various emotions regardless of the lighting conditions of the surrounding environment.
The facial expressions in the acquired 3D images were distinguished through machine learning with an average of 85% accuracy – a statistically significant figure compared to when 2D images were used. Furthermore, by calculating the interdependency of distance information that varies with facial expression in 3D images, the team could identify the information a light-field camera utilizes to distinguish human expressions.
Professor Ki-Hun Jeong said, “The sub-miniature light-field camera developed by the research team has the potential to become the new platform to quantitatively analyze the facial expressions and emotions of humans.” To highlight the significance of this research, he added, “It could be applied in various fields including mobile healthcare, field diagnosis, social cognition, and human-machine interactions.”
This research was published in Advanced Intelligent Systems online on December 16, under the title, “Machine-Learned Light-field Camera that Reads Facial Expression from High-Contrast and Illumination Invariant 3D Facial Images.” This research was funded by the Ministry of Science and ICT and the Ministry of Trade, Industry and Energy.
-Publication“Machine-learned light-field camera that reads fascial expression from high-contrast and illumination invariant 3D facial images,” Sang-In Bae, Sangyeon Lee, Jae-Myeong Kwon, Hyun-Kyung Kim. Kyung-Won Jang, Doheon Lee, Ki-Hun Jeong, Advanced Intelligent Systems, December 16, 2021 (doi.org/10.1002/aisy.202100182)
ProfileProfessor Ki-Hun JeongBiophotonic LaboratoryDepartment of Bio and Brain EngineeringKAIST
Professor Doheon LeeDepartment of Bio and Brain EngineeringKAIST
2022.01.21 View 13921 -
 Team KAIST Makes Its Presence Felt in the Self-Driving Tech Industry
Team KAIST finishes 4th at the inaugural CES Autonomous Racing Competition
Team KAIST led by Professor Hyunchul Shim and Unmanned Systems Research Group (USRG) placed fourth in an autonomous race car competition in Las Vegas last week, making its presence felt in the self-driving automotive tech industry.
Team KAIST, beat its first competitor, Auburn University, with speeds of up to 131 mph at the Autonomous Challenge at CES held at the Las Vegas Motor Speedway. However, the team failed to advance to the final round when it lost to PoliMOVE, comprised of the Polytechnic University of Milan and the University of Alabama, the final winner of the $150,000 USD race.
A total of eight teams competed in the self-driving race. The race was conducted as a single elimination tournament consisting of multiple rounds of matches. Two cars took turns playing the role of defender and attacker, and each car attempted to outpace the other until one of them was unable to complete the mission.
Each team designed the algorithm to control its racecar, the Dallara-built AV-21, which can reach a speed of up to 173 mph, and make it safely drive around the track at high speeds without crashing into the other.
The event is the CES version of the Indy Autonomous Challenge, a competition that took place for the first time in October last year to encourage university students from around the world to develop complicated software for autonomous driving and advance relevant technologies. Team KAIST placed 4th at the Indy Autonomous Challenge, which qualified it to participate in this race.
“The technical level of the CES race is much higher than last October’s and we had a very tough race. We advanced to the semifinals for two consecutive races. I think our autonomous vehicle technology is proving itself to the world,” said Professor Shim.
Professor Shim’s research group has been working on the development of autonomous aerial and ground vehicles for the past 12 years. A self-driving car developed by the lab was certified by the South Korean government to run on public roads.
The vehicle the team used cost more than 1 million USD to build. Many of the other teams had to repair their vehicle more than once due to accidents and had to spend a lot on repairs. “We are the only one who did not have any accidents, and this is a testament to our technological prowess,” said Professor Shim.
He said the financial funding to purchase pricy parts and equipment for the racecar is always a challenge given the very tight research budget and absence of corporate sponsorships.
However, Professor Shim and his research group plan to participate in the next race in September and in the 2023 CES race.
“I think we need more systemic and proactive research and support systems to earn better results but there is nothing better than the group of passionate students who are taking part in this project with us,” Shim added.
2022.01.12 View 12087
Team KAIST Makes Its Presence Felt in the Self-Driving Tech Industry
Team KAIST finishes 4th at the inaugural CES Autonomous Racing Competition
Team KAIST led by Professor Hyunchul Shim and Unmanned Systems Research Group (USRG) placed fourth in an autonomous race car competition in Las Vegas last week, making its presence felt in the self-driving automotive tech industry.
Team KAIST, beat its first competitor, Auburn University, with speeds of up to 131 mph at the Autonomous Challenge at CES held at the Las Vegas Motor Speedway. However, the team failed to advance to the final round when it lost to PoliMOVE, comprised of the Polytechnic University of Milan and the University of Alabama, the final winner of the $150,000 USD race.
A total of eight teams competed in the self-driving race. The race was conducted as a single elimination tournament consisting of multiple rounds of matches. Two cars took turns playing the role of defender and attacker, and each car attempted to outpace the other until one of them was unable to complete the mission.
Each team designed the algorithm to control its racecar, the Dallara-built AV-21, which can reach a speed of up to 173 mph, and make it safely drive around the track at high speeds without crashing into the other.
The event is the CES version of the Indy Autonomous Challenge, a competition that took place for the first time in October last year to encourage university students from around the world to develop complicated software for autonomous driving and advance relevant technologies. Team KAIST placed 4th at the Indy Autonomous Challenge, which qualified it to participate in this race.
“The technical level of the CES race is much higher than last October’s and we had a very tough race. We advanced to the semifinals for two consecutive races. I think our autonomous vehicle technology is proving itself to the world,” said Professor Shim.
Professor Shim’s research group has been working on the development of autonomous aerial and ground vehicles for the past 12 years. A self-driving car developed by the lab was certified by the South Korean government to run on public roads.
The vehicle the team used cost more than 1 million USD to build. Many of the other teams had to repair their vehicle more than once due to accidents and had to spend a lot on repairs. “We are the only one who did not have any accidents, and this is a testament to our technological prowess,” said Professor Shim.
He said the financial funding to purchase pricy parts and equipment for the racecar is always a challenge given the very tight research budget and absence of corporate sponsorships.
However, Professor Shim and his research group plan to participate in the next race in September and in the 2023 CES race.
“I think we need more systemic and proactive research and support systems to earn better results but there is nothing better than the group of passionate students who are taking part in this project with us,” Shim added.
2022.01.12 View 12087 -
 Team KAIST to Race at CES 2022 Autonomous Challenge
Five top university autonomous racing teams will compete in a head-to-head passing competition in Las Vegas
A self-driving racing team from the KAIST Unmanned System Research Group (USRG) advised by Professor Hyunchul Shim will compete at the Autonomous Challenge at the Consumer Electronic Show (CES) on January 7, 2022. The head-to-head, high speed autonomous racecar passing competition at the Las Vegas Motor Speedway will feature the finalists and semifinalists from the Indy Autonomous Challenge in October of this year. Team KAIST qualified as a semifinalist at the Indy Autonomous Challenge and will join four other university teams including the winner of the competition, Technische Universität München.
Team KAIST’s AV-21 vehicle is capable of driving on its own at more than 200km/h will be expected to show a speed of more than 300 km/h at the race.The participating teams are:1. KAIST2. EuroRacing : University of Modena and Reggio Emilia (Italy), University of Pisa (Italy), ETH Zürich (Switzerland), Polish Academy of Sciences (Poland) 3. MIT-PITT-RW, Massachusetts Institute of Technology, University of Pittsburgh, Rochester Institute of Technology, University of Waterloo (Canada)4.PoliMOVE – Politecnico di Milano (Italy), University of Alabama 5.TUM Autonomous Motorsport – Technische Universität München (Germany)
Professor Shim’s team is dedicated to the development and validation of cutting edge technologies for highly autonomous vehicles. In recognition of his pioneering research in unmanned system technologies, Professor Shim was honored with the Grand Prize of the Minister of Science and ICT on December 9.
“We began autonomous vehicle research in 2009 when we signed up for Hyundai Motor Company’s Autonomous Driving Challenge. For this, we developed a complete set of in-house technologies such as low-level vehicle control, perception, localization, and decision making.” In 2019, the team came in third place in the Challenge and they finally won this year.
For years, his team has participated in many unmanned systems challenges at home and abroad, gaining recognition around the world. The team won the inaugural 2016 IROS autonomous drone racing and placed second in the 2018 IROS Autonomous Drone Racing Competition. They also competed in 2017 MBZIRC, ranking fourth in Missions 2 and 3, and fifth in the Grand Challenge.
Most recently, the team won the first round of Lockheed Martin’s Alpha Pilot AI Drone Innovation Challenge. The team is now participating in the DARPA Subterranean Challenge as a member of Team CoSTAR with NASA JPL, MIT, and Caltech.
“We have accumulated plenty of first-hand experience developing autonomous vehicles with the support of domestic companies such as Hyundai Motor Company, Samsung, LG, and NAVER. In 2017, the autonomous vehicle platform “EureCar” that we developed in-house was authorized by the Korean government to lawfully conduct autonomous driving experiment on public roads,” said Professor Shim.
The team has developed various key technologies and algorithms related to unmanned systems that can be categorized into three major components: perception, planning, and control. Considering the characteristics of the algorithms that make up each module, their technology operates using a distributed computing system.
Since 2015, the team has been actively using deep learning algorithms in the form of perception subsystems. Contextual information extracted from multi-modal sensory data gathered via cameras, lidar, radar, GPS, IMU, etc. is forwarded to the planning subsystem. The planning module is responsible for the decision making and planning required for autonomous driving such as lane change determination and trajectory planning, emergency stops, and velocity command generation. The results from the planner are fed into the controller to follow the planned high-level command. The team has also developed and verified the possibility of an end-to-end deep learning based autonomous driving approach that replaces a complex system with one single AI network.
2021.12.17 View 12620
Team KAIST to Race at CES 2022 Autonomous Challenge
Five top university autonomous racing teams will compete in a head-to-head passing competition in Las Vegas
A self-driving racing team from the KAIST Unmanned System Research Group (USRG) advised by Professor Hyunchul Shim will compete at the Autonomous Challenge at the Consumer Electronic Show (CES) on January 7, 2022. The head-to-head, high speed autonomous racecar passing competition at the Las Vegas Motor Speedway will feature the finalists and semifinalists from the Indy Autonomous Challenge in October of this year. Team KAIST qualified as a semifinalist at the Indy Autonomous Challenge and will join four other university teams including the winner of the competition, Technische Universität München.
Team KAIST’s AV-21 vehicle is capable of driving on its own at more than 200km/h will be expected to show a speed of more than 300 km/h at the race.The participating teams are:1. KAIST2. EuroRacing : University of Modena and Reggio Emilia (Italy), University of Pisa (Italy), ETH Zürich (Switzerland), Polish Academy of Sciences (Poland) 3. MIT-PITT-RW, Massachusetts Institute of Technology, University of Pittsburgh, Rochester Institute of Technology, University of Waterloo (Canada)4.PoliMOVE – Politecnico di Milano (Italy), University of Alabama 5.TUM Autonomous Motorsport – Technische Universität München (Germany)
Professor Shim’s team is dedicated to the development and validation of cutting edge technologies for highly autonomous vehicles. In recognition of his pioneering research in unmanned system technologies, Professor Shim was honored with the Grand Prize of the Minister of Science and ICT on December 9.
“We began autonomous vehicle research in 2009 when we signed up for Hyundai Motor Company’s Autonomous Driving Challenge. For this, we developed a complete set of in-house technologies such as low-level vehicle control, perception, localization, and decision making.” In 2019, the team came in third place in the Challenge and they finally won this year.
For years, his team has participated in many unmanned systems challenges at home and abroad, gaining recognition around the world. The team won the inaugural 2016 IROS autonomous drone racing and placed second in the 2018 IROS Autonomous Drone Racing Competition. They also competed in 2017 MBZIRC, ranking fourth in Missions 2 and 3, and fifth in the Grand Challenge.
Most recently, the team won the first round of Lockheed Martin’s Alpha Pilot AI Drone Innovation Challenge. The team is now participating in the DARPA Subterranean Challenge as a member of Team CoSTAR with NASA JPL, MIT, and Caltech.
“We have accumulated plenty of first-hand experience developing autonomous vehicles with the support of domestic companies such as Hyundai Motor Company, Samsung, LG, and NAVER. In 2017, the autonomous vehicle platform “EureCar” that we developed in-house was authorized by the Korean government to lawfully conduct autonomous driving experiment on public roads,” said Professor Shim.
The team has developed various key technologies and algorithms related to unmanned systems that can be categorized into three major components: perception, planning, and control. Considering the characteristics of the algorithms that make up each module, their technology operates using a distributed computing system.
Since 2015, the team has been actively using deep learning algorithms in the form of perception subsystems. Contextual information extracted from multi-modal sensory data gathered via cameras, lidar, radar, GPS, IMU, etc. is forwarded to the planning subsystem. The planning module is responsible for the decision making and planning required for autonomous driving such as lane change determination and trajectory planning, emergency stops, and velocity command generation. The results from the planner are fed into the controller to follow the planned high-level command. The team has also developed and verified the possibility of an end-to-end deep learning based autonomous driving approach that replaces a complex system with one single AI network.
2021.12.17 View 12620 -
 A Study Shows Reactive Electrolyte Additives Improve Lithium Metal Battery Performance
Stable electrode-electrolyte interfaces constructed by fluorine- and nitrogen-donating ionic additives provide an opportunity to improve high-performance lithium metal batteries
A research team showed that electrolyte additives increase the lifetime of lithium metal batteries and remarkably improve the performance of fast charging and discharging. Professor Nam-Soon Choi’s team from the Department of Chemical and Biomolecular Engineering at KAIST hierarchized the solid electrolyte interphase to make a dual-layer structure and showed groundbreaking run times for lithium metal batteries.
The team applied two electrolyte additives that have different reduction and adsorption properties to improve the functionality of the dual-layer solid electrolyte interphase. In addition, the team has confirmed that the structural stability of the nickel-rich cathode was achieved through the formation of a thin protective layer on the cathode. This study was reported in Energy Storage Materials.
Securing high-energy-density lithium metal batteries with a long lifespan and fast charging performance is vital for realizing their ubiquitous use as superior power sources for electric vehicles. Lithium metal batteries comprise a lithium metal anode that delivers 10 times higher capacity than the graphite anodes in lithium-ion batteries. Therefore, lithium metal is an indispensable anode material for realizing high-energy rechargeable batteries. However, undesirable reactions among the electrolytes with lithium metal anodes can reduce the power and this remains an impediment to achieving a longer battery lifespan. Previous studies only focused on the formation of the solid electrolyte interphase on the surface of the lithium metal anode.
The team designed a way to create a dual-layer solid electrolyte interphase to resolve the instability of the lithium metal anode by using electrolyte additives, depending on their electron accepting ability and adsorption tendencies. This hierarchical structure of the solid electrolyte interphase on the lithium metal anode has the potential to be further applied to lithium-alloy anodes, lithium storage structures, and anode-free technology to meet market expectations for electrolyte technology.
The batteries with lithium metal anodes and nickel-rich cathodes represented 80.9% of the initial capacity after 600 cycles and achieved a high Coulombic efficiency of 99.94%. These remarkable results contributed to the development of protective dual-layer solid electrolyte interphase technology for lithium metal anodes.
Professor Choi said that the research suggests a new direction for the development of electrolyte additives to regulate the unstable lithium metal anode-electrolyte interface, the biggest hurdle in research on lithium metal batteries.
She added that anode-free secondary battery technology is expected to be a game changer in the secondary battery market and electrolyte additive technology will contribute to the enhancement of anode-free secondary batteries through the stabilization of lithium metal anodes.
This research was funded by the Technology Development Program to Solve Climate Change of the National Research Foundation in Korea funded by the Ministry of Science, ICT & Future Planning and the Technology Innovation Program funded by the Ministry of Trade, Industry & Energy, and Hyundai Motor Company.
- PublicationSaehun Kim, Sung O Park, Min-Young Lee, Jeong-A Lee, Imanuel Kristanto, Tae Kyung Lee, Daeyeon Hwang, Juyoung Kim, Tae-Ung Wi, Hyun-Wook Lee, Sang Kyu Kwak, and NamSoon Choi, “Stable electrode-electrolyte interfaces constructed by fluorine- and nitrogen-donating ionic additives for high-performance lithium metal batteries,” Energy Storage Materials,45, 1-13 (2022), (doi: https://doi.org/10.1016/j.ensm.2021.10.031)
- ProfileProfessor Nam-Soon ChoiEnergy Materials LaboratoryDepartment of Chemical and Biomolecular EngineeringKAIST
2021.12.16 View 10486
A Study Shows Reactive Electrolyte Additives Improve Lithium Metal Battery Performance
Stable electrode-electrolyte interfaces constructed by fluorine- and nitrogen-donating ionic additives provide an opportunity to improve high-performance lithium metal batteries
A research team showed that electrolyte additives increase the lifetime of lithium metal batteries and remarkably improve the performance of fast charging and discharging. Professor Nam-Soon Choi’s team from the Department of Chemical and Biomolecular Engineering at KAIST hierarchized the solid electrolyte interphase to make a dual-layer structure and showed groundbreaking run times for lithium metal batteries.
The team applied two electrolyte additives that have different reduction and adsorption properties to improve the functionality of the dual-layer solid electrolyte interphase. In addition, the team has confirmed that the structural stability of the nickel-rich cathode was achieved through the formation of a thin protective layer on the cathode. This study was reported in Energy Storage Materials.
Securing high-energy-density lithium metal batteries with a long lifespan and fast charging performance is vital for realizing their ubiquitous use as superior power sources for electric vehicles. Lithium metal batteries comprise a lithium metal anode that delivers 10 times higher capacity than the graphite anodes in lithium-ion batteries. Therefore, lithium metal is an indispensable anode material for realizing high-energy rechargeable batteries. However, undesirable reactions among the electrolytes with lithium metal anodes can reduce the power and this remains an impediment to achieving a longer battery lifespan. Previous studies only focused on the formation of the solid electrolyte interphase on the surface of the lithium metal anode.
The team designed a way to create a dual-layer solid electrolyte interphase to resolve the instability of the lithium metal anode by using electrolyte additives, depending on their electron accepting ability and adsorption tendencies. This hierarchical structure of the solid electrolyte interphase on the lithium metal anode has the potential to be further applied to lithium-alloy anodes, lithium storage structures, and anode-free technology to meet market expectations for electrolyte technology.
The batteries with lithium metal anodes and nickel-rich cathodes represented 80.9% of the initial capacity after 600 cycles and achieved a high Coulombic efficiency of 99.94%. These remarkable results contributed to the development of protective dual-layer solid electrolyte interphase technology for lithium metal anodes.
Professor Choi said that the research suggests a new direction for the development of electrolyte additives to regulate the unstable lithium metal anode-electrolyte interface, the biggest hurdle in research on lithium metal batteries.
She added that anode-free secondary battery technology is expected to be a game changer in the secondary battery market and electrolyte additive technology will contribute to the enhancement of anode-free secondary batteries through the stabilization of lithium metal anodes.
This research was funded by the Technology Development Program to Solve Climate Change of the National Research Foundation in Korea funded by the Ministry of Science, ICT & Future Planning and the Technology Innovation Program funded by the Ministry of Trade, Industry & Energy, and Hyundai Motor Company.
- PublicationSaehun Kim, Sung O Park, Min-Young Lee, Jeong-A Lee, Imanuel Kristanto, Tae Kyung Lee, Daeyeon Hwang, Juyoung Kim, Tae-Ung Wi, Hyun-Wook Lee, Sang Kyu Kwak, and NamSoon Choi, “Stable electrode-electrolyte interfaces constructed by fluorine- and nitrogen-donating ionic additives for high-performance lithium metal batteries,” Energy Storage Materials,45, 1-13 (2022), (doi: https://doi.org/10.1016/j.ensm.2021.10.031)
- ProfileProfessor Nam-Soon ChoiEnergy Materials LaboratoryDepartment of Chemical and Biomolecular EngineeringKAIST
2021.12.16 View 10486 -
 A Team of Three PhD Candidates Wins the Korea Semiconductor Design Contest
“We felt a sense of responsibility to help the nation advance its semiconductor design technology”
A CMOS (complementary metal-oxide semiconductor)-based “ultra-low noise signal chip” for 6G communications designed by three PhD candidates at the KAIST School of Electrical Engineering won the Presidential Award at the 22nd Korea Semiconductor Design Contest.
The winners are PhD candidates Sun-Eui Park, Yoon-Seo Cho, and Ju-Eun Bang from the Integrated Circuits and System Lab run by Professor Jaehyouk Choi. The contest, which is hosted by the Ministry of Trade, Industry and Energy and the Korea Semiconductors Industry Association, is one of the top national semiconductor design contests for college students.
Park said the team felt a sense of responsibility to help advance semiconductor design technology in Korea when deciding to participate the contest. The team expressed deep gratitude to Professor Choi for guiding their research on 6G communications.
“Our colleagues from other labs and seniors who already graduated helped us a great deal, so we owe them a lot,” explained Park. Cho added that their hard work finally got recognized and that acknowledgement pushes her to move forward with her research. Meanwhile, Bang said she is delighted to see that many people seem to be interested in her research topic.
Research for 6G is attempting to reach 1 tera bps (Tbps), 50 times faster than 5G communications with transmission speeds of up to 20 gigabytes. In general, the wider the communication frequency band, the higher the data transmission speed. Thus, the use of frequency bands above 100 gigahertz is essential for delivering high data transmission speeds for 6G communications.
However, it remains a big challenge to make a precise benchmark signal that can be used as a carrier wave in a high frequency band. Despite the advantages of CMOS’s ultra-small and low-power design, it still has limitations at high frequency bands and its operating frequency. Thus, it was difficult to achieve a frequency band above 100 gigahertz.
To overcome these challenges, the three students introduced ultra-low noise signal generation technology that can support high-order modulation technologies. This technology is expected to contribute to increasing the price competitiveness and density of 6G communication chips that will be used in the future.
5G just got started in 2020 and still has long way to go for full commercialization. Nevertheless, many researchers have started preparing for 6G technology, targeting 2030 since a new cellular communication appears in every other decade.
Professor Choi said, “Generating ultra-high frequency signals in bands above 100 GHz with highly accurate timing is one of the key technologies for implementing 6G communication hardware. Our research is significant for the development of the world’s first semiconductor chip that will use the CMOS process to achieve noise performance of less than 80fs in a frequency band above 100 GHz.”
The team members plan to work as circuit designers in Korean semiconductor companies after graduation. “We will continue to research the development of signal generators on the topic of award-winning 6G. We would like to continue our research on high-speed circuit designs such as ultra-fast analog-to-digital converters,” Park added.
2021.11.30 View 10474
A Team of Three PhD Candidates Wins the Korea Semiconductor Design Contest
“We felt a sense of responsibility to help the nation advance its semiconductor design technology”
A CMOS (complementary metal-oxide semiconductor)-based “ultra-low noise signal chip” for 6G communications designed by three PhD candidates at the KAIST School of Electrical Engineering won the Presidential Award at the 22nd Korea Semiconductor Design Contest.
The winners are PhD candidates Sun-Eui Park, Yoon-Seo Cho, and Ju-Eun Bang from the Integrated Circuits and System Lab run by Professor Jaehyouk Choi. The contest, which is hosted by the Ministry of Trade, Industry and Energy and the Korea Semiconductors Industry Association, is one of the top national semiconductor design contests for college students.
Park said the team felt a sense of responsibility to help advance semiconductor design technology in Korea when deciding to participate the contest. The team expressed deep gratitude to Professor Choi for guiding their research on 6G communications.
“Our colleagues from other labs and seniors who already graduated helped us a great deal, so we owe them a lot,” explained Park. Cho added that their hard work finally got recognized and that acknowledgement pushes her to move forward with her research. Meanwhile, Bang said she is delighted to see that many people seem to be interested in her research topic.
Research for 6G is attempting to reach 1 tera bps (Tbps), 50 times faster than 5G communications with transmission speeds of up to 20 gigabytes. In general, the wider the communication frequency band, the higher the data transmission speed. Thus, the use of frequency bands above 100 gigahertz is essential for delivering high data transmission speeds for 6G communications.
However, it remains a big challenge to make a precise benchmark signal that can be used as a carrier wave in a high frequency band. Despite the advantages of CMOS’s ultra-small and low-power design, it still has limitations at high frequency bands and its operating frequency. Thus, it was difficult to achieve a frequency band above 100 gigahertz.
To overcome these challenges, the three students introduced ultra-low noise signal generation technology that can support high-order modulation technologies. This technology is expected to contribute to increasing the price competitiveness and density of 6G communication chips that will be used in the future.
5G just got started in 2020 and still has long way to go for full commercialization. Nevertheless, many researchers have started preparing for 6G technology, targeting 2030 since a new cellular communication appears in every other decade.
Professor Choi said, “Generating ultra-high frequency signals in bands above 100 GHz with highly accurate timing is one of the key technologies for implementing 6G communication hardware. Our research is significant for the development of the world’s first semiconductor chip that will use the CMOS process to achieve noise performance of less than 80fs in a frequency band above 100 GHz.”
The team members plan to work as circuit designers in Korean semiconductor companies after graduation. “We will continue to research the development of signal generators on the topic of award-winning 6G. We would like to continue our research on high-speed circuit designs such as ultra-fast analog-to-digital converters,” Park added.
2021.11.30 View 10474 -
 Scientists Develop Wireless Networks that Allow Brain Circuits to Be Controlled Remotely through the Internet
Wireless implantable devices and IoT could manipulate the brains of animals from anywhere around the world due to their minimalistic hardware, low setup cost, ease of use, and customizable versatility
A new study shows that researchers can remotely control the brain circuits of numerous animals simultaneously and independently through the internet. The scientists believe this newly developed technology can speed up brain research and various neuroscience studies to uncover basic brain functions as well as the underpinnings of various neuropsychiatric and neurological disorders.
A multidisciplinary team of researchers at KAIST, Washington University in St. Louis, and the University of Colorado, Boulder, created a wireless ecosystem with its own wireless implantable devices and Internet of Things (IoT) infrastructure to enable high-throughput neuroscience experiments over the internet. This innovative technology could enable scientists to manipulate the brains of animals from anywhere around the world. The study was published in the journal Nature Biomedical Engineering on November 25
“This novel technology is highly versatile and adaptive. It can remotely control numerous neural implants and laboratory tools in real-time or in a scheduled way without direct human interactions,” said Professor Jae-Woong Jeong of the School of Electrical Engineering at KAIST and a senior author of the study. “These wireless neural devices and equipment integrated with IoT technology have enormous potential for science and medicine.”
The wireless ecosystem only requires a mini-computer that can be purchased for under $45, which connects to the internet and communicates with wireless multifunctional brain probes or other types of conventional laboratory equipment using IoT control modules. By optimally integrating the versatility and modular construction of both unique IoT hardware and software within a single ecosystem, this wireless technology offers new applications that have not been demonstrated before by a single standalone technology. This includes, but is not limited to minimalistic hardware, global remote access, selective and scheduled experiments, customizable automation, and high-throughput scalability.
“As long as researchers have internet access, they are able to trigger, customize, stop, validate, and store the outcomes of large experiments at any time and from anywhere in the world. They can remotely perform large-scale neuroscience experiments in animals deployed in multiple countries,” said one of the lead authors, Dr. Raza Qazi, a researcher with KAIST and the University of Colorado, Boulder. “The low cost of this system allows it to be easily adopted and can further fuel innovation across many laboratories,” Dr. Qazi added.
One of the significant advantages of this IoT neurotechnology is its ability to be mass deployed across the globe due to its minimalistic hardware, low setup cost, ease of use, and customizable versatility. Scientists across the world can quickly implement this technology within their existing laboratories with minimal budget concerns to achieve globally remote access, scalable experimental automation, or both, thus potentially reducing the time needed to unravel various neuroscientific challenges such as those associated with intractable neurological conditions.
Another senior author on the study, Professor Jordan McCall from the Department of Anesthesiology and Center for Clinical Pharmacology at Washington University in St. Louis, said this technology has the potential to change how basic neuroscience studies are performed. “One of the biggest limitations when trying to understand how the mammalian brain works is that we have to study these functions in unnatural conditions. This technology brings us one step closer to performing important studies without direct human interaction with the study subjects.”
The ability to remotely schedule experiments moves toward automating these types of experiments. Dr. Kyle Parker, an instructor at Washington University in St. Louis and another lead author on the study added, “This experimental automation can potentially help us reduce the number of animals used in biomedical research by reducing the variability introduced by various experimenters. This is especially important given our moral imperative to seek research designs that enable this reduction.”
The researchers believe this wireless technology may open new opportunities for many applications including brain research, pharmaceuticals, and telemedicine to treat diseases in the brain and other organs remotely. This remote automation technology could become even more valuable when many labs need to shut down, such as during the height of the COVID-19 pandemic.
This work was supported by grants from the KAIST Global Singularity Research Program, the National Research Foundation of Korea, the United States National Institute of Health, and Oak Ridge Associated Universities.
-PublicationRaza Qazi, Kyle Parker, Choong Yeon Kim, Jordan McCall, Jae-Woong Jeong et al. “Scalable and modular wireless-network infrastructure for large-scale behavioral neuroscience,” Nature Biomedical Engineering, November 25 2021 (doi.org/10.1038/s41551-021-00814-w)
-ProfileProfessor Jae-Woong JeongBio-Integrated Electronics and Systems LabSchool of Electrical EngineeringKAIST
2021.11.29 View 17108
Scientists Develop Wireless Networks that Allow Brain Circuits to Be Controlled Remotely through the Internet
Wireless implantable devices and IoT could manipulate the brains of animals from anywhere around the world due to their minimalistic hardware, low setup cost, ease of use, and customizable versatility
A new study shows that researchers can remotely control the brain circuits of numerous animals simultaneously and independently through the internet. The scientists believe this newly developed technology can speed up brain research and various neuroscience studies to uncover basic brain functions as well as the underpinnings of various neuropsychiatric and neurological disorders.
A multidisciplinary team of researchers at KAIST, Washington University in St. Louis, and the University of Colorado, Boulder, created a wireless ecosystem with its own wireless implantable devices and Internet of Things (IoT) infrastructure to enable high-throughput neuroscience experiments over the internet. This innovative technology could enable scientists to manipulate the brains of animals from anywhere around the world. The study was published in the journal Nature Biomedical Engineering on November 25
“This novel technology is highly versatile and adaptive. It can remotely control numerous neural implants and laboratory tools in real-time or in a scheduled way without direct human interactions,” said Professor Jae-Woong Jeong of the School of Electrical Engineering at KAIST and a senior author of the study. “These wireless neural devices and equipment integrated with IoT technology have enormous potential for science and medicine.”
The wireless ecosystem only requires a mini-computer that can be purchased for under $45, which connects to the internet and communicates with wireless multifunctional brain probes or other types of conventional laboratory equipment using IoT control modules. By optimally integrating the versatility and modular construction of both unique IoT hardware and software within a single ecosystem, this wireless technology offers new applications that have not been demonstrated before by a single standalone technology. This includes, but is not limited to minimalistic hardware, global remote access, selective and scheduled experiments, customizable automation, and high-throughput scalability.
“As long as researchers have internet access, they are able to trigger, customize, stop, validate, and store the outcomes of large experiments at any time and from anywhere in the world. They can remotely perform large-scale neuroscience experiments in animals deployed in multiple countries,” said one of the lead authors, Dr. Raza Qazi, a researcher with KAIST and the University of Colorado, Boulder. “The low cost of this system allows it to be easily adopted and can further fuel innovation across many laboratories,” Dr. Qazi added.
One of the significant advantages of this IoT neurotechnology is its ability to be mass deployed across the globe due to its minimalistic hardware, low setup cost, ease of use, and customizable versatility. Scientists across the world can quickly implement this technology within their existing laboratories with minimal budget concerns to achieve globally remote access, scalable experimental automation, or both, thus potentially reducing the time needed to unravel various neuroscientific challenges such as those associated with intractable neurological conditions.
Another senior author on the study, Professor Jordan McCall from the Department of Anesthesiology and Center for Clinical Pharmacology at Washington University in St. Louis, said this technology has the potential to change how basic neuroscience studies are performed. “One of the biggest limitations when trying to understand how the mammalian brain works is that we have to study these functions in unnatural conditions. This technology brings us one step closer to performing important studies without direct human interaction with the study subjects.”
The ability to remotely schedule experiments moves toward automating these types of experiments. Dr. Kyle Parker, an instructor at Washington University in St. Louis and another lead author on the study added, “This experimental automation can potentially help us reduce the number of animals used in biomedical research by reducing the variability introduced by various experimenters. This is especially important given our moral imperative to seek research designs that enable this reduction.”
The researchers believe this wireless technology may open new opportunities for many applications including brain research, pharmaceuticals, and telemedicine to treat diseases in the brain and other organs remotely. This remote automation technology could become even more valuable when many labs need to shut down, such as during the height of the COVID-19 pandemic.
This work was supported by grants from the KAIST Global Singularity Research Program, the National Research Foundation of Korea, the United States National Institute of Health, and Oak Ridge Associated Universities.
-PublicationRaza Qazi, Kyle Parker, Choong Yeon Kim, Jordan McCall, Jae-Woong Jeong et al. “Scalable and modular wireless-network infrastructure for large-scale behavioral neuroscience,” Nature Biomedical Engineering, November 25 2021 (doi.org/10.1038/s41551-021-00814-w)
-ProfileProfessor Jae-Woong JeongBio-Integrated Electronics and Systems LabSchool of Electrical EngineeringKAIST
2021.11.29 View 17108 -
 Renault 5 EV and Canoo’s Pickup Truck Win the 2021 FMOTY Awards
KAIST Future Mobility of the Year Awards recognize the most innovative concept cars of the year
The Renault 5 EV from France and a pickup truck from the US startup Canoo won the 2021 Future Mobility of the Year Awards (FMOTY) hosted by the Cho Chun Shik Graduate School of Green Transportation at KAIST. The awards ceremony was held at Renault Samsung Motors in Seoul on November 25.
KAIST began the FMOTY in 2019 to advance future car technology and stimulate growth in the industry. The award recognizes the most innovative ideas for making the most futuristic concept car and improving the technological and social value of the industry.
The awards ceremony was attended by KAIST President Kwang Hyung Lee, the dean of the Cho Chun Shik Graduate School of Green Transportation In Gwun Jang, CEO of Renault Samsung Motors Dominique Signora, and CEO of Canoo Tony Aquila. President Lee said, “The new world order will be impacted by new technology developers who envision the future. Their innovation and creative ideas will open a new world of sustainable future transportation.”
Out of the 46 concept cars revealed at global motor exhibitions between last year and the first quarter of this year, models demonstrating transport technology useful for future society and innovative service were selected in the categories of passenger cars and commercial vehicles.
Sixteen automotive journalists from 11 countries, including the chief editor of Car Magazine in Germany Georg Kacher and editorial director of BBC Top Gear Charlie Turner, participated as judges.
This year’s award for the best concept car for a passenger vehicle went to an electric vehicle, the Renault 5 EV. The compact electric car was highly regarded for its practicality and environmental friendliness. A pickup truck by Canoo, an American EV manufacturing start-up, won the award in the commercial vehicle category. The pickup features an innovative design allowing for a variety of functions topped with a competitive price and it received overwhelming support from the judges.
While Hyundai Motors swept both prizes at the awards last year and demonstrated the potential of Korean concept cars, Canoo’s win in the commercial vehicle section as a young American venture company brought attention to the changing dynamics in the automotive market. This shows that young EV start-ups can compete with existing car companies as the automotive paradigm is shifting from those with internal combustion engines to EVs.
The awards organizers said that the Cho Chun Shik Graduate School of Green Transportation will continue to hold the FMOTY to lead the fast-changing global mobility market. For more information, please visit www.fmoty.org.
2021.11.26 View 8438
Renault 5 EV and Canoo’s Pickup Truck Win the 2021 FMOTY Awards
KAIST Future Mobility of the Year Awards recognize the most innovative concept cars of the year
The Renault 5 EV from France and a pickup truck from the US startup Canoo won the 2021 Future Mobility of the Year Awards (FMOTY) hosted by the Cho Chun Shik Graduate School of Green Transportation at KAIST. The awards ceremony was held at Renault Samsung Motors in Seoul on November 25.
KAIST began the FMOTY in 2019 to advance future car technology and stimulate growth in the industry. The award recognizes the most innovative ideas for making the most futuristic concept car and improving the technological and social value of the industry.
The awards ceremony was attended by KAIST President Kwang Hyung Lee, the dean of the Cho Chun Shik Graduate School of Green Transportation In Gwun Jang, CEO of Renault Samsung Motors Dominique Signora, and CEO of Canoo Tony Aquila. President Lee said, “The new world order will be impacted by new technology developers who envision the future. Their innovation and creative ideas will open a new world of sustainable future transportation.”
Out of the 46 concept cars revealed at global motor exhibitions between last year and the first quarter of this year, models demonstrating transport technology useful for future society and innovative service were selected in the categories of passenger cars and commercial vehicles.
Sixteen automotive journalists from 11 countries, including the chief editor of Car Magazine in Germany Georg Kacher and editorial director of BBC Top Gear Charlie Turner, participated as judges.
This year’s award for the best concept car for a passenger vehicle went to an electric vehicle, the Renault 5 EV. The compact electric car was highly regarded for its practicality and environmental friendliness. A pickup truck by Canoo, an American EV manufacturing start-up, won the award in the commercial vehicle category. The pickup features an innovative design allowing for a variety of functions topped with a competitive price and it received overwhelming support from the judges.
While Hyundai Motors swept both prizes at the awards last year and demonstrated the potential of Korean concept cars, Canoo’s win in the commercial vehicle section as a young American venture company brought attention to the changing dynamics in the automotive market. This shows that young EV start-ups can compete with existing car companies as the automotive paradigm is shifting from those with internal combustion engines to EVs.
The awards organizers said that the Cho Chun Shik Graduate School of Green Transportation will continue to hold the FMOTY to lead the fast-changing global mobility market. For more information, please visit www.fmoty.org.
2021.11.26 View 8438 -
 Marien Buissonniere Awarded the 9th Grand Award for Future Strategy
Global healthcare and humanitarian activist honored by the Grand Award for Future Strategy
The Moon Soul Graduate School of Future Strategy awarded the 9th Grand Award for Future Strategy to Marine Buissonniere, an independent advisor and practitioner in the fields of global health and humanitarian action. She currently works as a senior advisor to the Prevent Epidemics team at Resolve to Save Lives. She also co-chairs Doctors Without Borders’ Transformational Investment Capacity.
Buissonniere was recognized for designing and implementing global response strategies in global strife and disaster stricken areas over the 25 years while serving as secretary general of Doctors Without Borders. She has been working with various government agencies around the world including Resolve to Save Lives to respond to the Covid-19 pandemic and preparing global future strategies for the post-pandemic era.
The Grand Award for Future Strategy recognizes individual and organization who have contributed to the nation and humanity through future research and strategies in the fields of science and technology, economy and industry, society and culture, politics and governance, and resources and environment.
The selection committee place particular emphasis on her humanitarian efforts toward North Korea. She was in charge of the task force for resuming the health project in North Korea and facilitated the North Korean program in 2002. She also played a significant role in raising awareness of North Korea’s humanitarian issues in the international community by lecturing at Columbia and Princeton.
Buissonniere said during the awards ceremony held online on November 5, “I am very grateful to receive this award from KAIST, a world’s top-flight university as well as from South Korea related to the Korean Peninsula and North Korea, where I have spent most of my life. What makes this award even more special is it is about the international medical relief activities and system innovations that I’ve devoted my life to over the last 25 years. I am going to continue this journey to help many people in difficult situations. Eventually, I would like to make it possible for those people in need to make their own future by themselves.”
2021.11.11 View 7422
Marien Buissonniere Awarded the 9th Grand Award for Future Strategy
Global healthcare and humanitarian activist honored by the Grand Award for Future Strategy
The Moon Soul Graduate School of Future Strategy awarded the 9th Grand Award for Future Strategy to Marine Buissonniere, an independent advisor and practitioner in the fields of global health and humanitarian action. She currently works as a senior advisor to the Prevent Epidemics team at Resolve to Save Lives. She also co-chairs Doctors Without Borders’ Transformational Investment Capacity.
Buissonniere was recognized for designing and implementing global response strategies in global strife and disaster stricken areas over the 25 years while serving as secretary general of Doctors Without Borders. She has been working with various government agencies around the world including Resolve to Save Lives to respond to the Covid-19 pandemic and preparing global future strategies for the post-pandemic era.
The Grand Award for Future Strategy recognizes individual and organization who have contributed to the nation and humanity through future research and strategies in the fields of science and technology, economy and industry, society and culture, politics and governance, and resources and environment.
The selection committee place particular emphasis on her humanitarian efforts toward North Korea. She was in charge of the task force for resuming the health project in North Korea and facilitated the North Korean program in 2002. She also played a significant role in raising awareness of North Korea’s humanitarian issues in the international community by lecturing at Columbia and Princeton.
Buissonniere said during the awards ceremony held online on November 5, “I am very grateful to receive this award from KAIST, a world’s top-flight university as well as from South Korea related to the Korean Peninsula and North Korea, where I have spent most of my life. What makes this award even more special is it is about the international medical relief activities and system innovations that I’ve devoted my life to over the last 25 years. I am going to continue this journey to help many people in difficult situations. Eventually, I would like to make it possible for those people in need to make their own future by themselves.”
2021.11.11 View 7422 -
 Nanoscale Self-Assembling Salt-Crystal ‘Origami’ Balls Envelop Liquids
Mechanical engineers have devised a ‘crystal capillary origami’ technique where salt crystals spontaneously encapsulate liquid droplets
Researchers have developed a technique whereby they can spontaneously encapsulate microscopic droplets of water and oil emulsion in a tiny sphere made of salt crystals—sort of like a minute, self-constructing origami soccer ball filled with liquid. The process, which they are calling ‘crystal capillary origami,’ could be used in a range of fields from more precise drug delivery to nanoscale medical devices.The technique is described in a paper appearing in the journal Nanoscale on September 21.
Capillary action, or ‘capillarity,’ will be familiar to most people as the way that water or other liquids can move up narrow tubes or other porous materials seemingly in defiance of gravity (for example within the vascular systems of plants, or even more simply, the drawing up of paint between the hairs of a paintbrush). This effect is due to the forces of cohesion (the tendency of a liquid’s molecules to stick together), which results in surface tension, and adhesion (their tendency to stick to the surface of other substances). The strength of the capillarity depends on the chemistry of the liquid, the chemistry of the porous material, and on the other forces acting on them both. For example, a liquid with lower surface tension than water would not be able to hold up a water strider insect.
Less well known is a related phenomenon, elasto-capillarity, that takes advantage of the relationship between capillarity and the elasticity of a very tiny flat sheet of a solid material. In certain circumstances, the capillary forces can overcome the elastic bending resistance of the sheet.
This relationship can be exploited to create ‘capillary origami,’ or three-dimensional structures. When a liquid droplet is placed on the flat sheet, the latter can spontaneously encapsulate the former due to surface tension. Capillary origami can take on other forms including wrinkling, buckling, or self-folding into other shapes. The specific geometrical shape that the 3D capillary origami structure ends up taking is determined by both the chemistry of the flat sheet and that of the liquid, and by carefully designing the shape and size of the sheet.
There is one big problem with these small devices, however. “These conventional self-assembled origami structures cannot be completely spherical and will always have discontinuous boundaries, or what you might call ‘edges,’ as a result of the original two-dimensional shape of the sheet,” said Kwangseok Park, a lead researcher on the project. He added, “These edges could turn out to be future defects with the potential for failure in the face of increased stress.” Non-spherical particles are also known to be more disadvantageous than spherical particles in terms of cellular uptake.
Professor Hyoungsoo Kim from the Department of Mechanical Engineering explained, “This is why researchers have long been on the hunt for substances that could produce a fully spherical capillary origami structure.”
The authors of the study have demonstrated such an origami sphere for the first time. They showed how instead of a flat sheet, the growth of salt-crystals can perform capillary origami action in a similar manner. What they call ‘crystal capillary origami’ spontaneously constructs a smooth spherical shell capsule from these same surface tension effects, but now the spontaneous encapsulation of a liquid is determined by the elasto-capillary conditions of growing crystals.
Here, the term ‘salt’ refers to a compound of one positively charged ion and another negatively charged. Table salt, or sodium chloride, is just one example of a salt. The researchers used four other salts: calcium propionate, sodium salicylate, calcium nitrate tetrahydrate, and sodium bicarbonate to envelop a water-oil emulsion. Normally, a salt such as sodium chloride has a cubical crystal structure, but these four salts form plate-like structures as crystallites or ‘grains’ (the microscopic shape that forms when a crystal first starts to grow) instead. These plates then self-assemble into perfect spheres.
Using scanning electron microscopy and X-ray diffraction analysis, they investigated the mechanism of such formation and concluded that it was ‘Laplace pressure’ that drives the crystallite plates to cover the emulsion surface. Laplace pressure describes the pressure difference between the interior and exterior of a curved surface caused by the surface tension at the interface between the two substances, in this case between the salt water and the oil.
The researchers hope that these self-assembling nanostructures can be used for encapsulation applications in a range of sectors, from the food industry and cosmetics to drug delivery and even tiny medical devices.
-Publication
Kwangseok Park, Hyoungsoo Kim “Crystal capillary origami capsule with self-assembled nanostructure,” Nanoscale, 13(35), 14656-14665 (DOI: 10.1039/d1nr02456f)
-Profile
Professor Hyoungsoo Kim
Fluid and Interface Laboratory
http://fil.kaist.ac.kr
Department of Mechanical Engineering
KAIST
2021.11.04 View 10837
Nanoscale Self-Assembling Salt-Crystal ‘Origami’ Balls Envelop Liquids
Mechanical engineers have devised a ‘crystal capillary origami’ technique where salt crystals spontaneously encapsulate liquid droplets
Researchers have developed a technique whereby they can spontaneously encapsulate microscopic droplets of water and oil emulsion in a tiny sphere made of salt crystals—sort of like a minute, self-constructing origami soccer ball filled with liquid. The process, which they are calling ‘crystal capillary origami,’ could be used in a range of fields from more precise drug delivery to nanoscale medical devices.The technique is described in a paper appearing in the journal Nanoscale on September 21.
Capillary action, or ‘capillarity,’ will be familiar to most people as the way that water or other liquids can move up narrow tubes or other porous materials seemingly in defiance of gravity (for example within the vascular systems of plants, or even more simply, the drawing up of paint between the hairs of a paintbrush). This effect is due to the forces of cohesion (the tendency of a liquid’s molecules to stick together), which results in surface tension, and adhesion (their tendency to stick to the surface of other substances). The strength of the capillarity depends on the chemistry of the liquid, the chemistry of the porous material, and on the other forces acting on them both. For example, a liquid with lower surface tension than water would not be able to hold up a water strider insect.
Less well known is a related phenomenon, elasto-capillarity, that takes advantage of the relationship between capillarity and the elasticity of a very tiny flat sheet of a solid material. In certain circumstances, the capillary forces can overcome the elastic bending resistance of the sheet.
This relationship can be exploited to create ‘capillary origami,’ or three-dimensional structures. When a liquid droplet is placed on the flat sheet, the latter can spontaneously encapsulate the former due to surface tension. Capillary origami can take on other forms including wrinkling, buckling, or self-folding into other shapes. The specific geometrical shape that the 3D capillary origami structure ends up taking is determined by both the chemistry of the flat sheet and that of the liquid, and by carefully designing the shape and size of the sheet.
There is one big problem with these small devices, however. “These conventional self-assembled origami structures cannot be completely spherical and will always have discontinuous boundaries, or what you might call ‘edges,’ as a result of the original two-dimensional shape of the sheet,” said Kwangseok Park, a lead researcher on the project. He added, “These edges could turn out to be future defects with the potential for failure in the face of increased stress.” Non-spherical particles are also known to be more disadvantageous than spherical particles in terms of cellular uptake.
Professor Hyoungsoo Kim from the Department of Mechanical Engineering explained, “This is why researchers have long been on the hunt for substances that could produce a fully spherical capillary origami structure.”
The authors of the study have demonstrated such an origami sphere for the first time. They showed how instead of a flat sheet, the growth of salt-crystals can perform capillary origami action in a similar manner. What they call ‘crystal capillary origami’ spontaneously constructs a smooth spherical shell capsule from these same surface tension effects, but now the spontaneous encapsulation of a liquid is determined by the elasto-capillary conditions of growing crystals.
Here, the term ‘salt’ refers to a compound of one positively charged ion and another negatively charged. Table salt, or sodium chloride, is just one example of a salt. The researchers used four other salts: calcium propionate, sodium salicylate, calcium nitrate tetrahydrate, and sodium bicarbonate to envelop a water-oil emulsion. Normally, a salt such as sodium chloride has a cubical crystal structure, but these four salts form plate-like structures as crystallites or ‘grains’ (the microscopic shape that forms when a crystal first starts to grow) instead. These plates then self-assemble into perfect spheres.
Using scanning electron microscopy and X-ray diffraction analysis, they investigated the mechanism of such formation and concluded that it was ‘Laplace pressure’ that drives the crystallite plates to cover the emulsion surface. Laplace pressure describes the pressure difference between the interior and exterior of a curved surface caused by the surface tension at the interface between the two substances, in this case between the salt water and the oil.
The researchers hope that these self-assembling nanostructures can be used for encapsulation applications in a range of sectors, from the food industry and cosmetics to drug delivery and even tiny medical devices.
-Publication
Kwangseok Park, Hyoungsoo Kim “Crystal capillary origami capsule with self-assembled nanostructure,” Nanoscale, 13(35), 14656-14665 (DOI: 10.1039/d1nr02456f)
-Profile
Professor Hyoungsoo Kim
Fluid and Interface Laboratory
http://fil.kaist.ac.kr
Department of Mechanical Engineering
KAIST
2021.11.04 View 10837 -
 GSI Forum Highlights Global Collaboration Toward a Sustainable Global Economy
The forum stresses global collaboration to make the global value chain more resilient
Speakers at the 5th Global Strategy Institute International Forum on October 28 stressed the importance of global collaboration for rebuilding the global economy and making innovations in national science and technology governance in order to enhance national competitiveness. The forum entitled “Grand Strategic Shift under Global Techno-Geopolitical Paradigm” examined strategies for making the global supply chain more resilient and rebuild the global economy as well as how Korea could advance in the technology race.
Speakers concurred that technology has become an issue of national security. The global supply chain has been disrupted amid the global pandemic and intense conflict between the U.S. and China. Speakers presented a common solution: global collaboration and innovations in science and technology governance.
KAIST President Kwang Hyung Lee said in his opening remarks that the future ‘world map’ may turn out very differently depending on how we prepare and what we envision for the future. He also stressed the importance of technology sovereignty, adding that only those who can create their own new technology independently will be the future leaders.
Prime Minister Boo Kyum Kim and Vice Minister of Science and ICT Hongtaek Yong delivered congratulatory remarks. Keynote speakers included Professor Scott Stern from the MIT Sloan School of Management, Professor Aaron Chatterji from the Fuqua School of Business at Duke University, Professor Sarah Kreps from the Department of Government at Cornell University, SK Group Chairperson Tae-Won Chey, President Woo Il Lee of the Korean Federation of Science and Technology Societies, Professor Young Kwan Yoon at Seoul National University, President Eun Mee Kim of Ewha Womans University, and President Ieehwan Kim of the University of Science and Technology.
During the first session, Professor Chatterji stressed that how to make supply chains resilience will be the key for making long-term strategy with relevant government policy. He said that AI has become a general purpose technology (GPT) and Korea ranked 4th in AI innovation in the world, but how to translate this innovativeness into national strategic leadership will be a new challenge for Korea. He suggested that Korea strengthen its strategic partnerships with allies such as the U.S. and provide opportunities not only for established players but start-ups and entrepreneurs. Meanwhile, Professor Kreps said that industrial policy should also leverage trust and innovations for building technology alliances with a more longer-term approach, without antagonizing certain groups of nations.
Vice President for Planning and Budget Bowon Kim who joined the forum as a discussant pointed out that in this hyper-connected era, nothing can be manufactured in a single company and country without the global supply chain. “In longer-term policy and strategies, we should embrace China as a global economy partner and include all nations around the world.”
Chairman Chey from SK said that the clear role among universities, industry, and the government doesn’t exist any longer. Now, universities are working hard for the commercialization of technology from their labs. Industry is nurturing the talents inept for future industry, and the government is trying to introduce a more private-sector approach. As such, universities, the government, and industry should embrace all-inclusive approaches encompassing global politics and trade to lead on the global stage.
Meanwhile in the second session, all of the speakers stressed innovation in science and technology governance in order to adopt to the new industrial paradigm. They agreed to make prompt innovations and solid collaborative systems among the government ministries to ensure national competitiveness, especially in the field of science and technology.
President Lee from KOFST said Korea should adopt a first mover strategy and the government should adopt a mission-oriented projects and deregulate more. He pointed out that when mandating more autonomy in decision making, scientists and students can make more creative outcomes.
Professor Yoon at SNU stressed the close alliance with the U.S. in the technology race, but suggested that Korea should also seek ways to help minimize the technology gap between advanced and developing countries. Universities should also be allowed more autonomy in running creative curriculum and academic affairs to in order boost the competitiveness of science and technology.
President Kim from Ewha pointed out the role of education as a public good. In some countries, strengthening science and technology can be accomplished with wider educational opportunities in middle and high schools. President Kim also stressed expanding strategic partnerships. She said Korea should expand its alliances and partnerships, not only with the U.S. but with European countries and other niche countries where certain technologies are superior.
President Kim of UST stressed a new science and technology leadership is required to build technology sovereignty and the government should spearhead the deregulations of the government policy.
This GSI forum was co-hosted by two think-tanks at KAIST, the Korea Policy Center for the Fourth Industrial Revolution (KPC4IR) and the Innovation Strategy and Policy Institute (ISPI).
2021.10.28 View 10256
GSI Forum Highlights Global Collaboration Toward a Sustainable Global Economy
The forum stresses global collaboration to make the global value chain more resilient
Speakers at the 5th Global Strategy Institute International Forum on October 28 stressed the importance of global collaboration for rebuilding the global economy and making innovations in national science and technology governance in order to enhance national competitiveness. The forum entitled “Grand Strategic Shift under Global Techno-Geopolitical Paradigm” examined strategies for making the global supply chain more resilient and rebuild the global economy as well as how Korea could advance in the technology race.
Speakers concurred that technology has become an issue of national security. The global supply chain has been disrupted amid the global pandemic and intense conflict between the U.S. and China. Speakers presented a common solution: global collaboration and innovations in science and technology governance.
KAIST President Kwang Hyung Lee said in his opening remarks that the future ‘world map’ may turn out very differently depending on how we prepare and what we envision for the future. He also stressed the importance of technology sovereignty, adding that only those who can create their own new technology independently will be the future leaders.
Prime Minister Boo Kyum Kim and Vice Minister of Science and ICT Hongtaek Yong delivered congratulatory remarks. Keynote speakers included Professor Scott Stern from the MIT Sloan School of Management, Professor Aaron Chatterji from the Fuqua School of Business at Duke University, Professor Sarah Kreps from the Department of Government at Cornell University, SK Group Chairperson Tae-Won Chey, President Woo Il Lee of the Korean Federation of Science and Technology Societies, Professor Young Kwan Yoon at Seoul National University, President Eun Mee Kim of Ewha Womans University, and President Ieehwan Kim of the University of Science and Technology.
During the first session, Professor Chatterji stressed that how to make supply chains resilience will be the key for making long-term strategy with relevant government policy. He said that AI has become a general purpose technology (GPT) and Korea ranked 4th in AI innovation in the world, but how to translate this innovativeness into national strategic leadership will be a new challenge for Korea. He suggested that Korea strengthen its strategic partnerships with allies such as the U.S. and provide opportunities not only for established players but start-ups and entrepreneurs. Meanwhile, Professor Kreps said that industrial policy should also leverage trust and innovations for building technology alliances with a more longer-term approach, without antagonizing certain groups of nations.
Vice President for Planning and Budget Bowon Kim who joined the forum as a discussant pointed out that in this hyper-connected era, nothing can be manufactured in a single company and country without the global supply chain. “In longer-term policy and strategies, we should embrace China as a global economy partner and include all nations around the world.”
Chairman Chey from SK said that the clear role among universities, industry, and the government doesn’t exist any longer. Now, universities are working hard for the commercialization of technology from their labs. Industry is nurturing the talents inept for future industry, and the government is trying to introduce a more private-sector approach. As such, universities, the government, and industry should embrace all-inclusive approaches encompassing global politics and trade to lead on the global stage.
Meanwhile in the second session, all of the speakers stressed innovation in science and technology governance in order to adopt to the new industrial paradigm. They agreed to make prompt innovations and solid collaborative systems among the government ministries to ensure national competitiveness, especially in the field of science and technology.
President Lee from KOFST said Korea should adopt a first mover strategy and the government should adopt a mission-oriented projects and deregulate more. He pointed out that when mandating more autonomy in decision making, scientists and students can make more creative outcomes.
Professor Yoon at SNU stressed the close alliance with the U.S. in the technology race, but suggested that Korea should also seek ways to help minimize the technology gap between advanced and developing countries. Universities should also be allowed more autonomy in running creative curriculum and academic affairs to in order boost the competitiveness of science and technology.
President Kim from Ewha pointed out the role of education as a public good. In some countries, strengthening science and technology can be accomplished with wider educational opportunities in middle and high schools. President Kim also stressed expanding strategic partnerships. She said Korea should expand its alliances and partnerships, not only with the U.S. but with European countries and other niche countries where certain technologies are superior.
President Kim of UST stressed a new science and technology leadership is required to build technology sovereignty and the government should spearhead the deregulations of the government policy.
This GSI forum was co-hosted by two think-tanks at KAIST, the Korea Policy Center for the Fourth Industrial Revolution (KPC4IR) and the Innovation Strategy and Policy Institute (ISPI).
2021.10.28 View 10256 -
 Flexible Sensor-Integrated RFA Needle Leads to Smarter Medical Treatment
Clinical trial of flexible sensor-integrated radiofrequency ablation (RFA) needle tip monitors physical changes and steam pop
Researchers have designed a thin polymeric sensor platform on a radiofrequency ablation needle to monitor temperature and pressure in real time. The sensors integrated onto 1.5 mm diameter needle tip have proven their efficacy during clinical tests and expect to provide a new opportunity for safer and more effective medical practices. The research was reported in Advanced Science as the frontispiece on August 5.
Radiofrequency ablation (RFA) is a minimally invasive surgery technique for removing tumors and treating cardiovascular disease. During a procedure, an unintended audible explosion called ‘steam pop’ can occur due to the increased internal steam pressure in the ablation region. This phenomenon has been cited as a cause of various negative thermal and mechanical effects on neighboring tissue. Even more, the relationship between steam pop and cancer recurrence is still being investigated.
Professor Inkyu Park said that his team’s integrated sensors reliably detected the occurrence of steam pop. The sensors also monitor rapidly spreading hot steam in tissue. It is expected that the diverse properties of tissue undergoing RFA could be checked by utilizing the physical sensors integrated on the needle.
“We believe that the integrated sensors can provide useful information about a variety of medical procedures and accompanying environmental changes in the human body, and help develop more effective and safer surgical procedures,” said Professor Park.
Professor Park’s team built a thin film type pressure and temperature sensor stack with a thickness of less than 10 μm using a microfabrication process. For the pressure sensor, the team used contact resistance changes between metal electrodes and a carbon nanotube coated polymeric membrane. The entire sensor array was thoroughly insulated with medical tubes to minimize any exposure of the sensor materials to external tissue and maximize its biocompatibility.
During the clinical trial, the research team found that the accumulated hot steam is suddenly released during steam pops and this hot air spreads to neighboring tissue, which accelerates the ablation process. Furthermore, using in-situ ultrasound imaging and computational simulations, the research team could confirm the non-uniform temperature distribution around the RFA needle can be one of the primary reasons for the steam popping.
Professor Park explained that various physical and chemical sensors for different targets can be added to create other medical devices and industrial tools.
“This result will expand the usability and applicability of current flexible sensor technologies. We are also trying to integrate this sensor onto a 0.3mm diameter needle for in-vivo diagnosis applications and expect that this approach can be applied to other medical treatments as well as the industrial field,” added Professor Park. This study was supported by the National Research Foundation of Korea.
-PublicationJaeho Park, Jinwoo Lee, Hyo Keun Lim, Inkyu Park et al. “Real-Time Internal Steam Pop Detection during Radiofrequency Ablation with a Radiofrequency Ablation Needle Integrated with a Temperature and Pressure Sensor: Preclinical and clinical pilot tests," Advanced Science (https://doi/org/10.1002/advs.202100725) on August 5, 2021
-ProfileProfessor Inkyu ParkMicro & Nano Tranducers Laboratory http://mintlab1.kaist.ac.kr/
Department of Mechanical EngineeringCollege of EngineeringKAIST
2021.10.20 View 10050
Flexible Sensor-Integrated RFA Needle Leads to Smarter Medical Treatment
Clinical trial of flexible sensor-integrated radiofrequency ablation (RFA) needle tip monitors physical changes and steam pop
Researchers have designed a thin polymeric sensor platform on a radiofrequency ablation needle to monitor temperature and pressure in real time. The sensors integrated onto 1.5 mm diameter needle tip have proven their efficacy during clinical tests and expect to provide a new opportunity for safer and more effective medical practices. The research was reported in Advanced Science as the frontispiece on August 5.
Radiofrequency ablation (RFA) is a minimally invasive surgery technique for removing tumors and treating cardiovascular disease. During a procedure, an unintended audible explosion called ‘steam pop’ can occur due to the increased internal steam pressure in the ablation region. This phenomenon has been cited as a cause of various negative thermal and mechanical effects on neighboring tissue. Even more, the relationship between steam pop and cancer recurrence is still being investigated.
Professor Inkyu Park said that his team’s integrated sensors reliably detected the occurrence of steam pop. The sensors also monitor rapidly spreading hot steam in tissue. It is expected that the diverse properties of tissue undergoing RFA could be checked by utilizing the physical sensors integrated on the needle.
“We believe that the integrated sensors can provide useful information about a variety of medical procedures and accompanying environmental changes in the human body, and help develop more effective and safer surgical procedures,” said Professor Park.
Professor Park’s team built a thin film type pressure and temperature sensor stack with a thickness of less than 10 μm using a microfabrication process. For the pressure sensor, the team used contact resistance changes between metal electrodes and a carbon nanotube coated polymeric membrane. The entire sensor array was thoroughly insulated with medical tubes to minimize any exposure of the sensor materials to external tissue and maximize its biocompatibility.
During the clinical trial, the research team found that the accumulated hot steam is suddenly released during steam pops and this hot air spreads to neighboring tissue, which accelerates the ablation process. Furthermore, using in-situ ultrasound imaging and computational simulations, the research team could confirm the non-uniform temperature distribution around the RFA needle can be one of the primary reasons for the steam popping.
Professor Park explained that various physical and chemical sensors for different targets can be added to create other medical devices and industrial tools.
“This result will expand the usability and applicability of current flexible sensor technologies. We are also trying to integrate this sensor onto a 0.3mm diameter needle for in-vivo diagnosis applications and expect that this approach can be applied to other medical treatments as well as the industrial field,” added Professor Park. This study was supported by the National Research Foundation of Korea.
-PublicationJaeho Park, Jinwoo Lee, Hyo Keun Lim, Inkyu Park et al. “Real-Time Internal Steam Pop Detection during Radiofrequency Ablation with a Radiofrequency Ablation Needle Integrated with a Temperature and Pressure Sensor: Preclinical and clinical pilot tests," Advanced Science (https://doi/org/10.1002/advs.202100725) on August 5, 2021
-ProfileProfessor Inkyu ParkMicro & Nano Tranducers Laboratory http://mintlab1.kaist.ac.kr/
Department of Mechanical EngineeringCollege of EngineeringKAIST
2021.10.20 View 10050