BIO
-
 KAIST Researchers Receive Awards at the 13th Asian Congress on Biotechnology
(From left: Seon Young Park, Dr. So Young Choi, and Yoojin Choi)
Researchers in the laboratory of KAIST Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering swept awards at the 13th Asian Congress on Biotechnology held in Thailand last month. The conference awarded a total of eight prizes in the areas of best research and best poster presentation. This is an exceptional case in which members of one research team received almost half of the awards at an international conference.
Dr. So Young Choi received the Best Research Award, while Ph.D. candidates Yoojin Choi and Seon Young Park each received the Best Poster Presentation Award at the conference held in Khon Kaen, Thailand from July 23 to 27.
The Asian Congress on Biotechnology is an international conference in which scientists and industry experts in Asia and from around the world gather to present recent research findings in the field of biotechnology. At the conference, around 400 researchers in biotechnology from 25 countries, including Korea, gathered to present and discuss various research findings under the theme of “Bioinnovation and Bioeconomy.”
Distinguished Professor Sang Yup Lee attended the conference to give the opening plenary lecture on the topic of ‘Systems Strategies in Biotechnology.’ Professor Lee announced, “I have attended international conferences with students for the last 20 years, but this is the first in which my team received three awards at an international conference that only honors a total of eight awards, three for Best Research and five for Best Presentation.”
Dr. Choi presented research results on poly (lactate-co-glycolate) (PLGA) synthesis through a biological method using micro-organisms and received the Best Research Award. PLGA is a random copolymer of DL-lactic and glycolic acids and is a biopolymer widely used for biomedical applications. PLGA is biodegradable, biocompatible, and nontoxic, and thus has been approved by the US Food and Drug Administration (FDA) for its use in implants, drug delivery, and sutures.
Dr. Choi’s research was deemed to be innovative for synthesizing PLGA from glucose and xylose in cells through metabolic engineering of E.Coli. Dr. Choi received her Ph.D. under the supervision of Distinguished Professor Lee this February and is currently conducting post-doc research.
Ph.D. candidate Choi presented her research on the use of recombinant E.Coli for the biological synthesis of various nanoparticles and received the Best Poster Presentation award. Choi used recombinant E.Coli-expressing proteins and peptides that adsorb to heavy metals to biologically synthesize diverse metal nanoparticles such as single-nanoparticle including gold and silver, quantum dots, and magnetic nanoparticles for the first time. The synthesized nanoparticles can be used in the fields of bio-imaging, diagnosis, environment, and energy.
Ph.D. candidate Park, who also received the Best Poster Presentation award, synthesized and increased production of astanxanthin, a strong antioxidant found in nature, in E.Coli using metabolic engineering. Astanxanthin is a carotenoid pigment found in salmon and shrimp that widely used in health products and cosmetics.
2017.08.01 View 11476
KAIST Researchers Receive Awards at the 13th Asian Congress on Biotechnology
(From left: Seon Young Park, Dr. So Young Choi, and Yoojin Choi)
Researchers in the laboratory of KAIST Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering swept awards at the 13th Asian Congress on Biotechnology held in Thailand last month. The conference awarded a total of eight prizes in the areas of best research and best poster presentation. This is an exceptional case in which members of one research team received almost half of the awards at an international conference.
Dr. So Young Choi received the Best Research Award, while Ph.D. candidates Yoojin Choi and Seon Young Park each received the Best Poster Presentation Award at the conference held in Khon Kaen, Thailand from July 23 to 27.
The Asian Congress on Biotechnology is an international conference in which scientists and industry experts in Asia and from around the world gather to present recent research findings in the field of biotechnology. At the conference, around 400 researchers in biotechnology from 25 countries, including Korea, gathered to present and discuss various research findings under the theme of “Bioinnovation and Bioeconomy.”
Distinguished Professor Sang Yup Lee attended the conference to give the opening plenary lecture on the topic of ‘Systems Strategies in Biotechnology.’ Professor Lee announced, “I have attended international conferences with students for the last 20 years, but this is the first in which my team received three awards at an international conference that only honors a total of eight awards, three for Best Research and five for Best Presentation.”
Dr. Choi presented research results on poly (lactate-co-glycolate) (PLGA) synthesis through a biological method using micro-organisms and received the Best Research Award. PLGA is a random copolymer of DL-lactic and glycolic acids and is a biopolymer widely used for biomedical applications. PLGA is biodegradable, biocompatible, and nontoxic, and thus has been approved by the US Food and Drug Administration (FDA) for its use in implants, drug delivery, and sutures.
Dr. Choi’s research was deemed to be innovative for synthesizing PLGA from glucose and xylose in cells through metabolic engineering of E.Coli. Dr. Choi received her Ph.D. under the supervision of Distinguished Professor Lee this February and is currently conducting post-doc research.
Ph.D. candidate Choi presented her research on the use of recombinant E.Coli for the biological synthesis of various nanoparticles and received the Best Poster Presentation award. Choi used recombinant E.Coli-expressing proteins and peptides that adsorb to heavy metals to biologically synthesize diverse metal nanoparticles such as single-nanoparticle including gold and silver, quantum dots, and magnetic nanoparticles for the first time. The synthesized nanoparticles can be used in the fields of bio-imaging, diagnosis, environment, and energy.
Ph.D. candidate Park, who also received the Best Poster Presentation award, synthesized and increased production of astanxanthin, a strong antioxidant found in nature, in E.Coli using metabolic engineering. Astanxanthin is a carotenoid pigment found in salmon and shrimp that widely used in health products and cosmetics.
2017.08.01 View 11476 -
 Top 10 Emerging Technologies of 2017
The World Economic Forum’s Expert Network and Global Future Councils in collaboration with Scientific American and its Board of Advisors announced the top 10 emerging technologies of 2017 on June 26 in Dalian, China where the 2017 Summer Davos Forum is being held. Each technology was chosen for its potential to improve lives, transform industries, and safeguard the planet.
The KAIST delegation, headed by President Sung-Chul Shin, is participating in the forum’s diverse activities including IdeasLab and GULF (Global University Leaders Forum). KAIST is the only Korean representative participating in the IdeasLab. KAIST Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering, director of KAIST Institute, has served as a committee member of the Global Agenda Council on Emerging Technologies since 2012 and Global Future Council on the Fourth Industrial Revolution. He also chairs the Global Future Council on Biotechnologies.
Professor Lee said, “Very diverse technological breakthroughs were proposed for the final list of candidates. We made the final selections through very in-depth discussion with experts in each field.
We focused on the technologies which have a level of maturity that will enable them to be adopted widely within three to five years."
The top 10 emerging technologies are
(courtesy from https://
www.weforum.org/agenda/2017/06/these-are-the-top-10-emerging-technologies-of-2017):
2017 10대 기술.
1. Liquid biopsies
Liquid biopsies mark a step forward in the fight against cancer. First, they are an alternative where traditional tissue-based biopsies are not possible. Second, they provide a full spectrum of information compared to tissue samples, which only reflect the information available in the sample. Lastly, by homing in on circulating-tumor DNA (ctDNA), genetic material that routinely finds its way from cancer cells into the bloodstream, disease progression or resistance to treatment can be spotted much faster than otherwise relying on symptoms or imaging.
2. Harvesting clean water from air
The ability to extract clean water from air is not new, however existing techniques require high moisture levels and a lot of electricity. This is changing. A team from MIT and University of California, Berkeley has successfully tested a process using porous crystals that convert the water using no energy at all.
3. Deep learning for visual tasks
Computers are beginning to recognize images better than humans. Thanks to deep learning, an emerging field of artificial intelligence, computer-vision technologies are increasingly being used in applications as diverse as driving autonomous vehicles, medical diagnostics, damage assessment for insurance claims, and monitoring water levels and crop yield.
4. Liquid fuels from sunshine
Can we mimic the humble leaf to create artificial photosynthesis to generate and store energy? The prospects are looking increasingly positive. The answer lies in using sunlight-activated catalysts to split water molecules into water and hydrogen, and then using the same hydrogen to convert CO2 into hydrocarbons.
5. The Human Cell Atlas
An international collaboration aimed at deciphering the human body, called the Human Cell Atlas, was launched in October 2016. The project aims to identify every cell type in every tissue; learn exactly which genes, proteins, and other molecules are active in each type, and the processes which control that activity.
6. Precision farming
The Fourth Industrial Revolution is providing farmers with a new set of tools to boost crop yield and quality while reducing water and chemical use. Sensors, robots, GPS, mapping tools, and data-analytics software are all being used to customize the care that plants need.
7. Affordable catalysts for green vehicles
Progress is being made on a promising zero-emission technology, the hydrogen-fed fuel cell. Progress to date has been stymied by the high price of catalysts which contain platinum. However, much progress has been made in reducing reliance on this rare and expensive metal, and the latest developments involve catalysts that include no platinum, or in some cases no metal at all.
8. Genomic vaccines
Vaccines based on genes are superior to more conventional ones in a number of ways. They are faster to manufacture, which is crucial during violent outbreaks. Compared to manufacturing proteins in cell cultures or eggs, producing genetic material should also be simpler and less expensive.
9. Sustainable design of communities
Applying green construction to multiple buildings at once has the potential to revolutionize the amount of energy and water we consume. Sending locally-generated solar power to a smart microgrid could reduce electricity consumption by half and reduce carbon emissions to zero if a project currently under development at the University of California at Berkeley goes according to plan.
10. Quantum computing
Quantum computers’ almost limitless potential has only ever been matched by the difficulty and cost of their construction. This explains why today the small ones that have been built have not yet managed to exceed the power of supercomputers. But progress is being made and in 2016 the technology firm IBM provided public access to the first quantum computer in the cloud.
2017.06.28 View 10331
Top 10 Emerging Technologies of 2017
The World Economic Forum’s Expert Network and Global Future Councils in collaboration with Scientific American and its Board of Advisors announced the top 10 emerging technologies of 2017 on June 26 in Dalian, China where the 2017 Summer Davos Forum is being held. Each technology was chosen for its potential to improve lives, transform industries, and safeguard the planet.
The KAIST delegation, headed by President Sung-Chul Shin, is participating in the forum’s diverse activities including IdeasLab and GULF (Global University Leaders Forum). KAIST is the only Korean representative participating in the IdeasLab. KAIST Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering, director of KAIST Institute, has served as a committee member of the Global Agenda Council on Emerging Technologies since 2012 and Global Future Council on the Fourth Industrial Revolution. He also chairs the Global Future Council on Biotechnologies.
Professor Lee said, “Very diverse technological breakthroughs were proposed for the final list of candidates. We made the final selections through very in-depth discussion with experts in each field.
We focused on the technologies which have a level of maturity that will enable them to be adopted widely within three to five years."
The top 10 emerging technologies are
(courtesy from https://
www.weforum.org/agenda/2017/06/these-are-the-top-10-emerging-technologies-of-2017):
2017 10대 기술.
1. Liquid biopsies
Liquid biopsies mark a step forward in the fight against cancer. First, they are an alternative where traditional tissue-based biopsies are not possible. Second, they provide a full spectrum of information compared to tissue samples, which only reflect the information available in the sample. Lastly, by homing in on circulating-tumor DNA (ctDNA), genetic material that routinely finds its way from cancer cells into the bloodstream, disease progression or resistance to treatment can be spotted much faster than otherwise relying on symptoms or imaging.
2. Harvesting clean water from air
The ability to extract clean water from air is not new, however existing techniques require high moisture levels and a lot of electricity. This is changing. A team from MIT and University of California, Berkeley has successfully tested a process using porous crystals that convert the water using no energy at all.
3. Deep learning for visual tasks
Computers are beginning to recognize images better than humans. Thanks to deep learning, an emerging field of artificial intelligence, computer-vision technologies are increasingly being used in applications as diverse as driving autonomous vehicles, medical diagnostics, damage assessment for insurance claims, and monitoring water levels and crop yield.
4. Liquid fuels from sunshine
Can we mimic the humble leaf to create artificial photosynthesis to generate and store energy? The prospects are looking increasingly positive. The answer lies in using sunlight-activated catalysts to split water molecules into water and hydrogen, and then using the same hydrogen to convert CO2 into hydrocarbons.
5. The Human Cell Atlas
An international collaboration aimed at deciphering the human body, called the Human Cell Atlas, was launched in October 2016. The project aims to identify every cell type in every tissue; learn exactly which genes, proteins, and other molecules are active in each type, and the processes which control that activity.
6. Precision farming
The Fourth Industrial Revolution is providing farmers with a new set of tools to boost crop yield and quality while reducing water and chemical use. Sensors, robots, GPS, mapping tools, and data-analytics software are all being used to customize the care that plants need.
7. Affordable catalysts for green vehicles
Progress is being made on a promising zero-emission technology, the hydrogen-fed fuel cell. Progress to date has been stymied by the high price of catalysts which contain platinum. However, much progress has been made in reducing reliance on this rare and expensive metal, and the latest developments involve catalysts that include no platinum, or in some cases no metal at all.
8. Genomic vaccines
Vaccines based on genes are superior to more conventional ones in a number of ways. They are faster to manufacture, which is crucial during violent outbreaks. Compared to manufacturing proteins in cell cultures or eggs, producing genetic material should also be simpler and less expensive.
9. Sustainable design of communities
Applying green construction to multiple buildings at once has the potential to revolutionize the amount of energy and water we consume. Sending locally-generated solar power to a smart microgrid could reduce electricity consumption by half and reduce carbon emissions to zero if a project currently under development at the University of California at Berkeley goes according to plan.
10. Quantum computing
Quantum computers’ almost limitless potential has only ever been matched by the difficulty and cost of their construction. This explains why today the small ones that have been built have not yet managed to exceed the power of supercomputers. But progress is being made and in 2016 the technology firm IBM provided public access to the first quantum computer in the cloud.
2017.06.28 View 10331 -
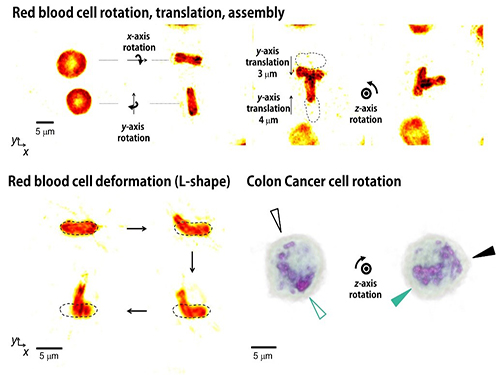 Controlling 3D Behavior of Biological Cells Using Laser Holographic Techniques
A research team led by Professor YongKeun Park of the Physics Department at KAIST has developed an optical manipulation technique that can freely control the position, orientation, and shape of microscopic samples having complex shapes. The study has been published online in Nature Communications on May 22.
Conventional optical manipulation techniques called “optical tweezers,” have been used as an invaluable tool for exerting micro-scale force on microscopic particles and manipulating three-dimensional (3-D) positions of particles. Optical tweezers employ a tightly-focused laser whose beam diameter is smaller than one micrometer (1/100 of hair thickness), which can generate attractive force on neighboring microscopic particles moving toward the beam focus. Controlling the positions of the beam focus enabled researchers to hold the particles and move them freely to other locations so they coined the name “optical tweezers,” and have been widely used in various fields of physical and biological studies.
So far, most experiments using optical tweezers have been conducted for trapping spherical particles because physical principles can easily predict optical forces and the responding motion of microspheres. For trapping objects having complicated shapes, however, conventional optical tweezers induce unstable motion of such particles, and controllable orientation of such objects is limited, which hinder controlling the 3-D motion of microscopic objects having complex shapes such as living cells.
The research team has developed a new optical manipulation technique that can trap complex objects of arbitrary shapes. This technique first measures 3-D structures of an object in real time using a 3-D holographic microscope, which shares the same physical principle of X-Ray CT imaging. Based on the measured 3-D shape of the object, the researchers precisely calculates the shape of light that can stably control the object. When the shape of light is the same as the shape of the object, the energy of the object is minimized, which provides the stable trapping of the object having the complicated shape.
Moreover, by controlling the shape of light to have various positions, directions, and shapes of objects, it is possible to freely control the 3-D motion of the object and make the object have a desired shape. This process resembles the generation of a mold for casting a statue having desired shape so the researchers coined the name of the present technique “tomographic mold for optical trapping (TOMOTRAP).” The team succeeded in trapping individual human red blood cells stably, rotating them with desired orientations, folding them in an L-shape, and assembling two red blood cells together to form a new structure. In addition, colon cancer cells having a complex structure could be stably trapped and rotated at desired orientations. All of which have been difficult to be realized by the conventional optical techniques.
Professor Park said, “Our technique has the advantage of controlling the 3-D motion of complex shaped objects without knowing prior information about their shape and optical characteristics, and can be applied in various fields including physics, optics, nanotechnology, and medical science.”
Dr. Kyoohyun Kim, the lead author of this paper, noted that this technique can induce controlled deformation of biological cells with desired shapes. “This approach can be also applied to real-time monitoring of surgical prognosis of cellular-level surgeries for capturing and deforming cells as well as subcellular organelles,” added Kim.
Figure 1. Concept of optical manipulation techniques
Figure 2. Experimental setup
Figure 3. Research results
2017.05.25 View 7237
Controlling 3D Behavior of Biological Cells Using Laser Holographic Techniques
A research team led by Professor YongKeun Park of the Physics Department at KAIST has developed an optical manipulation technique that can freely control the position, orientation, and shape of microscopic samples having complex shapes. The study has been published online in Nature Communications on May 22.
Conventional optical manipulation techniques called “optical tweezers,” have been used as an invaluable tool for exerting micro-scale force on microscopic particles and manipulating three-dimensional (3-D) positions of particles. Optical tweezers employ a tightly-focused laser whose beam diameter is smaller than one micrometer (1/100 of hair thickness), which can generate attractive force on neighboring microscopic particles moving toward the beam focus. Controlling the positions of the beam focus enabled researchers to hold the particles and move them freely to other locations so they coined the name “optical tweezers,” and have been widely used in various fields of physical and biological studies.
So far, most experiments using optical tweezers have been conducted for trapping spherical particles because physical principles can easily predict optical forces and the responding motion of microspheres. For trapping objects having complicated shapes, however, conventional optical tweezers induce unstable motion of such particles, and controllable orientation of such objects is limited, which hinder controlling the 3-D motion of microscopic objects having complex shapes such as living cells.
The research team has developed a new optical manipulation technique that can trap complex objects of arbitrary shapes. This technique first measures 3-D structures of an object in real time using a 3-D holographic microscope, which shares the same physical principle of X-Ray CT imaging. Based on the measured 3-D shape of the object, the researchers precisely calculates the shape of light that can stably control the object. When the shape of light is the same as the shape of the object, the energy of the object is minimized, which provides the stable trapping of the object having the complicated shape.
Moreover, by controlling the shape of light to have various positions, directions, and shapes of objects, it is possible to freely control the 3-D motion of the object and make the object have a desired shape. This process resembles the generation of a mold for casting a statue having desired shape so the researchers coined the name of the present technique “tomographic mold for optical trapping (TOMOTRAP).” The team succeeded in trapping individual human red blood cells stably, rotating them with desired orientations, folding them in an L-shape, and assembling two red blood cells together to form a new structure. In addition, colon cancer cells having a complex structure could be stably trapped and rotated at desired orientations. All of which have been difficult to be realized by the conventional optical techniques.
Professor Park said, “Our technique has the advantage of controlling the 3-D motion of complex shaped objects without knowing prior information about their shape and optical characteristics, and can be applied in various fields including physics, optics, nanotechnology, and medical science.”
Dr. Kyoohyun Kim, the lead author of this paper, noted that this technique can induce controlled deformation of biological cells with desired shapes. “This approach can be also applied to real-time monitoring of surgical prognosis of cellular-level surgeries for capturing and deforming cells as well as subcellular organelles,” added Kim.
Figure 1. Concept of optical manipulation techniques
Figure 2. Experimental setup
Figure 3. Research results
2017.05.25 View 7237 -
 Distinguished Professor Lee Elected to the NAS
Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering was elected as a foreign associate to the US National Academy of Sciences (NAS) on May 2. The National Academy of Sciences elected 84 new members and 21 foreign associates in recognition of their distinguished and continuing achievements in their original research. Election to the Academy is widely regarded as one of the highest honors that a scientist can receive.
Professor Lee was also elected in 2010 as a member of the US National Academy of Engineering (NAE) for his leadership in microbial biotechnology and metabolic engineering, including the development of fermentation processes for biodegradable polymers and organic acids. Until 2016, there are only 12 people worldwide who are foreign associates of both NAS and NAE.
He is the first Korean elected to both prestigious academies, the NAS and the NAE in the US. Professor Lee is currently the dean of KAIST Institutes, the world leading institute for multi-and interdisciplinary research. He is also serving as co-chair of the Global Council on Biotechnology and member of the Global Future Council on the Fourth Industrial Revolution, the World Economic Forum.
2017.05.16 View 6837
Distinguished Professor Lee Elected to the NAS
Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering was elected as a foreign associate to the US National Academy of Sciences (NAS) on May 2. The National Academy of Sciences elected 84 new members and 21 foreign associates in recognition of their distinguished and continuing achievements in their original research. Election to the Academy is widely regarded as one of the highest honors that a scientist can receive.
Professor Lee was also elected in 2010 as a member of the US National Academy of Engineering (NAE) for his leadership in microbial biotechnology and metabolic engineering, including the development of fermentation processes for biodegradable polymers and organic acids. Until 2016, there are only 12 people worldwide who are foreign associates of both NAS and NAE.
He is the first Korean elected to both prestigious academies, the NAS and the NAE in the US. Professor Lee is currently the dean of KAIST Institutes, the world leading institute for multi-and interdisciplinary research. He is also serving as co-chair of the Global Council on Biotechnology and member of the Global Future Council on the Fourth Industrial Revolution, the World Economic Forum.
2017.05.16 View 6837 -
 ANSYS Korea Donates Engineering Simulation Software
ANSYS Korea made an in-kind donation of engineering simulation software, Multiphysics Campus Solution, to KAIST on March 24. ANSYS Korea donated 10,000 copies for education and 1,000 copies for research valued at about 4 billion KRW (about 200 billion KRW commercially).
The ANSYS software will benefit the engineering simulation work in nine departments and 60 labs for three years, including the departments of mechanical engineering, aerospace engineering, electrical engineering, civil and environmental engineering, nuclear and quantum engineering, chemical and bimolecular engineering, bio and brain engineering, materials science and engineering, and the Cho Chun Shik Graduate School of Green Transportation.
ANSYS is a global engineering simulation company. It provides ANSYS CAE (Computer Aided Engineering) software products in various industries in the world as well as various support, training, and consulting services. Deemed an exemplary model of university-industry R&D collaboration especially in the Industry 4.0 era, their donation will help create the best engineering education environment possible at KAIST.
ANSYS's multi-physics campus solution is a comprehensive software suite that spans the entire range of physics, providing access to virtually any field of engineering simulation that a design process requires. It expands the fields of fluids, structures, electromagnetics, and semiconductors. Undergraduates use it to learn physics principles and gain hands-on, real-world experience that can lead to a deeper understanding of engineering concepts. Postgraduate researchers apply simulation tools to solve complex engineering problems and produce data for their theses.
"Engineering simulations are playing a stronger role in science and engineering. ANSYS software will help our undergraduates and our researchers learn the principles of physics and deepen their understanding of engineering concepts. We hope this will serve as an instrumental tool for multidisciplinary studies, critical to fostering our students," said President Sung-Chul Shin.
ANSYS Korea CEO Yong-Won Cho added, "We sincerely hope our software will help KAIST students and researchers experience the best engineering education and achieve significant research results."
(Photo caption: President Shin (left) poses with ANSYS Korea CEO Yong-Won Cho at the donation ceremony on March 24 at KAIST)
2017.03.24 View 6549
ANSYS Korea Donates Engineering Simulation Software
ANSYS Korea made an in-kind donation of engineering simulation software, Multiphysics Campus Solution, to KAIST on March 24. ANSYS Korea donated 10,000 copies for education and 1,000 copies for research valued at about 4 billion KRW (about 200 billion KRW commercially).
The ANSYS software will benefit the engineering simulation work in nine departments and 60 labs for three years, including the departments of mechanical engineering, aerospace engineering, electrical engineering, civil and environmental engineering, nuclear and quantum engineering, chemical and bimolecular engineering, bio and brain engineering, materials science and engineering, and the Cho Chun Shik Graduate School of Green Transportation.
ANSYS is a global engineering simulation company. It provides ANSYS CAE (Computer Aided Engineering) software products in various industries in the world as well as various support, training, and consulting services. Deemed an exemplary model of university-industry R&D collaboration especially in the Industry 4.0 era, their donation will help create the best engineering education environment possible at KAIST.
ANSYS's multi-physics campus solution is a comprehensive software suite that spans the entire range of physics, providing access to virtually any field of engineering simulation that a design process requires. It expands the fields of fluids, structures, electromagnetics, and semiconductors. Undergraduates use it to learn physics principles and gain hands-on, real-world experience that can lead to a deeper understanding of engineering concepts. Postgraduate researchers apply simulation tools to solve complex engineering problems and produce data for their theses.
"Engineering simulations are playing a stronger role in science and engineering. ANSYS software will help our undergraduates and our researchers learn the principles of physics and deepen their understanding of engineering concepts. We hope this will serve as an instrumental tool for multidisciplinary studies, critical to fostering our students," said President Sung-Chul Shin.
ANSYS Korea CEO Yong-Won Cho added, "We sincerely hope our software will help KAIST students and researchers experience the best engineering education and achieve significant research results."
(Photo caption: President Shin (left) poses with ANSYS Korea CEO Yong-Won Cho at the donation ceremony on March 24 at KAIST)
2017.03.24 View 6549 -
 Professor Jae Kyoung Kim Receives the 2017 HSFP Award
The Human Frontier Science Program (HSFP), one of the most competitive research grants in life sciences, has funded researchers worldwide across and beyond the field since 1990. Each year, the program selects a handful of recipients who push the envelope of basic research in biology to bring breakthroughs from novel approaches. Among its 7,000 recipients thus far, 26 scientists have received the Nobel Prize. For that reason, HSFP grants are often referred to as “Nobel Prize Grants.”
Professor Jae Kyoung Kim of the Mathematical Sciences Department at KAIST and his international collaborators, Professor Robert Havekes from the University of Groningen, the Netherlands, Professor Sara Aton from the University of Michigan in Ann Arbor, the United States, and Professor Matias Zurbriggen from the University of Düsseldorf, Germany, won the Young Investigator Grants of the 2017 HSFP.
The 30 winning teams of the 2017 competition (in 9 Young Investigator Grants and 21 Program Grants) went through a rigorous year-long review process from a total of 1,073 applications submitted from more than 60 countries around the world. Each winning team will receive financial support averaging 110,000-125,000 USD per year for three years.
Although Professor Kim was trained as a mathematician, he has extended his research focus into biological sciences and attempted to solve some of the most difficult problems in biology by employing mathematical theories and applications including nonlinear dynamics, stochastic process, singular perturbation, and parameter estimation.
The project that won the Young Investigator Grants was a study on how a molecular circadian clock may affect sleep-regulated neurophysiology in mammals. Physiological and metabolic processes such as sleep, blood pressure, and hormone secretion exhibit circadian rhythms in mammals. Professor Kim used mathematical modeling and analysis to explain that the mammalian circadian clock is a hierarchical system, in which the master clock in the superchiasmatic nucleus, a tiny region in the brain that controls circadian rhythms, functions as a pacemaker and synchronizer of peripheral clocks to generate coherent systematic rhythms throughout the body.
Professor Kim said, “The mechanisms of our neuronal and hormonal activities regulating many of our bodily functions over a 24-hour cycle are not yet fully known. We go to sleep every night, but do not really know how it affects our brain functions. I hope my experience in mathematics, along with insights from biologists, can find meaningful answers to some of today’s puzzling problems in biological sciences, for example, revealing the complexities of our brains and showing how they work.”
“In the meantime, I hope collaborations between the fields of mathematics and biology, as yet a rare phenomenon in the Korean scientific community, will become more popular in the near future.”
Professor Kim received his doctoral degree in Applied and Interdisciplinary Mathematics in 2013 from the University of Michigan and joined KAIST in 2015. He has published numerous articles in reputable science journals such as Science, Molecular Cell, Proceedings of the National Academy of Sciences, and Nature Communications.
Both the Program Grants and Young Investigator Grants support international teams with members from at least two countries for innovative and creative research. This year, the Program Grants were awarded to research topics ranging from the evolution of counting and the role of extracellular vesicles in breast cancer bone metastasis to the examination of obesity from a mechanobiological point of view.
The Young Investigator Grants are limited to teams that established their independent research within the last five years and received their doctoral degrees within the last decade. Besides Professor Kim’s study, such topics as the use of infrasound for navigation by seabirds and protein formation in photochemistry and photophysics were awarded in 2017.
Full lists of the 2017 HFSP winners are available at: http://www.hfsp.org/awardees/newly-awarded.
About the Human Frontier Science Program (HFSP):
The HFSP is a research funding program implemented by the International Human Frontier Science Program (HFSPO) based in Strasbourg, France. It promotes intercontinental collaboration and training in cutting-edge, interdisciplinary research specializing in life sciences. Founded in 1989, the HFSPO consists of the European Union and 14 other countries including the G7 nations and South Korea.
2017.03.21 View 7807
Professor Jae Kyoung Kim Receives the 2017 HSFP Award
The Human Frontier Science Program (HSFP), one of the most competitive research grants in life sciences, has funded researchers worldwide across and beyond the field since 1990. Each year, the program selects a handful of recipients who push the envelope of basic research in biology to bring breakthroughs from novel approaches. Among its 7,000 recipients thus far, 26 scientists have received the Nobel Prize. For that reason, HSFP grants are often referred to as “Nobel Prize Grants.”
Professor Jae Kyoung Kim of the Mathematical Sciences Department at KAIST and his international collaborators, Professor Robert Havekes from the University of Groningen, the Netherlands, Professor Sara Aton from the University of Michigan in Ann Arbor, the United States, and Professor Matias Zurbriggen from the University of Düsseldorf, Germany, won the Young Investigator Grants of the 2017 HSFP.
The 30 winning teams of the 2017 competition (in 9 Young Investigator Grants and 21 Program Grants) went through a rigorous year-long review process from a total of 1,073 applications submitted from more than 60 countries around the world. Each winning team will receive financial support averaging 110,000-125,000 USD per year for three years.
Although Professor Kim was trained as a mathematician, he has extended his research focus into biological sciences and attempted to solve some of the most difficult problems in biology by employing mathematical theories and applications including nonlinear dynamics, stochastic process, singular perturbation, and parameter estimation.
The project that won the Young Investigator Grants was a study on how a molecular circadian clock may affect sleep-regulated neurophysiology in mammals. Physiological and metabolic processes such as sleep, blood pressure, and hormone secretion exhibit circadian rhythms in mammals. Professor Kim used mathematical modeling and analysis to explain that the mammalian circadian clock is a hierarchical system, in which the master clock in the superchiasmatic nucleus, a tiny region in the brain that controls circadian rhythms, functions as a pacemaker and synchronizer of peripheral clocks to generate coherent systematic rhythms throughout the body.
Professor Kim said, “The mechanisms of our neuronal and hormonal activities regulating many of our bodily functions over a 24-hour cycle are not yet fully known. We go to sleep every night, but do not really know how it affects our brain functions. I hope my experience in mathematics, along with insights from biologists, can find meaningful answers to some of today’s puzzling problems in biological sciences, for example, revealing the complexities of our brains and showing how they work.”
“In the meantime, I hope collaborations between the fields of mathematics and biology, as yet a rare phenomenon in the Korean scientific community, will become more popular in the near future.”
Professor Kim received his doctoral degree in Applied and Interdisciplinary Mathematics in 2013 from the University of Michigan and joined KAIST in 2015. He has published numerous articles in reputable science journals such as Science, Molecular Cell, Proceedings of the National Academy of Sciences, and Nature Communications.
Both the Program Grants and Young Investigator Grants support international teams with members from at least two countries for innovative and creative research. This year, the Program Grants were awarded to research topics ranging from the evolution of counting and the role of extracellular vesicles in breast cancer bone metastasis to the examination of obesity from a mechanobiological point of view.
The Young Investigator Grants are limited to teams that established their independent research within the last five years and received their doctoral degrees within the last decade. Besides Professor Kim’s study, such topics as the use of infrasound for navigation by seabirds and protein formation in photochemistry and photophysics were awarded in 2017.
Full lists of the 2017 HFSP winners are available at: http://www.hfsp.org/awardees/newly-awarded.
About the Human Frontier Science Program (HFSP):
The HFSP is a research funding program implemented by the International Human Frontier Science Program (HFSPO) based in Strasbourg, France. It promotes intercontinental collaboration and training in cutting-edge, interdisciplinary research specializing in life sciences. Founded in 1989, the HFSPO consists of the European Union and 14 other countries including the G7 nations and South Korea.
2017.03.21 View 7807 -
 13 KAIST Faculty Named as Inaugural Members of Y-KAST
The Korean Academy of Science and Technology (KAST) launched the Young Korean Academy of Science and Technology (Y-KAST) and selected 73 scientists as its inaugural members on February 24. Among them, 13 KAIST faculty were recognized as the inaugural members of Y-KAST.
Y-KAIST, made up of distinguished mid-career scientists under the age of 45, will take the leading role in international collaboration as well as innovative agenda-making in science and technology.
The inaugural members include Professor Hyotcherl Ihee of the Department of Chemistry and Dr. Sung-Jin Oh of the Center for Mathematical Challenges at the Korea Institute for Advanced Study (KIAS), affiliated with KAIST. Professor Ihee is gaining wide acclaim in the fields of physics and chemistry, and in 2016, Dr. Oh was the youngest ever awardee of the Presidential Award of Young Scientist.
The other Y-KAIST members are as follows: Professors Haeshin Lee of the Department of Chemistry; Mi Young Kim, Byung-Kwan Cho, and Ji-Joon Song of the Department of Biological Sciences; Song-Yong Kim of the Department of Mechanical Engineering; Sang-il Oum of the Department of Mathematical Sciences; Jung Kyoon Choi of the Department of Bio and Brain Engineering; Seokwoo Jeon, Sang Ouk Kim, and Il-Doo Kim of the Department of Materials Science and Engineering; Jang Wook Choi of the Graduate School of EEWS (Energy, Environment, Water and Sustainability); and Jeong Ho Lee of the Graduate School of Medical Science and Engineering.
The leading countries of the Academy of Science, which include Germany, Sweden, Belgium, Canada, and Japan, have established the Young Academy of Science since 2010 in order to encourage the research activities of their young scientists and to establish a global platform for collaborative research projects through their active networking at home and abroad.
President Myung-Chul Lee of KAST said, “We will spare no effort to connect these outstanding mid-career researchers for their future collaboration. Their networking will make significant impacts toward their own research activities as well as the global stature of Korea’s science and technology R&D.
(Photo caption: Members of Y-KAST pose at the inaugural ceremony of Y-KAST on February 24.)
2017.03.02 View 13767
13 KAIST Faculty Named as Inaugural Members of Y-KAST
The Korean Academy of Science and Technology (KAST) launched the Young Korean Academy of Science and Technology (Y-KAST) and selected 73 scientists as its inaugural members on February 24. Among them, 13 KAIST faculty were recognized as the inaugural members of Y-KAST.
Y-KAIST, made up of distinguished mid-career scientists under the age of 45, will take the leading role in international collaboration as well as innovative agenda-making in science and technology.
The inaugural members include Professor Hyotcherl Ihee of the Department of Chemistry and Dr. Sung-Jin Oh of the Center for Mathematical Challenges at the Korea Institute for Advanced Study (KIAS), affiliated with KAIST. Professor Ihee is gaining wide acclaim in the fields of physics and chemistry, and in 2016, Dr. Oh was the youngest ever awardee of the Presidential Award of Young Scientist.
The other Y-KAIST members are as follows: Professors Haeshin Lee of the Department of Chemistry; Mi Young Kim, Byung-Kwan Cho, and Ji-Joon Song of the Department of Biological Sciences; Song-Yong Kim of the Department of Mechanical Engineering; Sang-il Oum of the Department of Mathematical Sciences; Jung Kyoon Choi of the Department of Bio and Brain Engineering; Seokwoo Jeon, Sang Ouk Kim, and Il-Doo Kim of the Department of Materials Science and Engineering; Jang Wook Choi of the Graduate School of EEWS (Energy, Environment, Water and Sustainability); and Jeong Ho Lee of the Graduate School of Medical Science and Engineering.
The leading countries of the Academy of Science, which include Germany, Sweden, Belgium, Canada, and Japan, have established the Young Academy of Science since 2010 in order to encourage the research activities of their young scientists and to establish a global platform for collaborative research projects through their active networking at home and abroad.
President Myung-Chul Lee of KAST said, “We will spare no effort to connect these outstanding mid-career researchers for their future collaboration. Their networking will make significant impacts toward their own research activities as well as the global stature of Korea’s science and technology R&D.
(Photo caption: Members of Y-KAST pose at the inaugural ceremony of Y-KAST on February 24.)
2017.03.02 View 13767 -
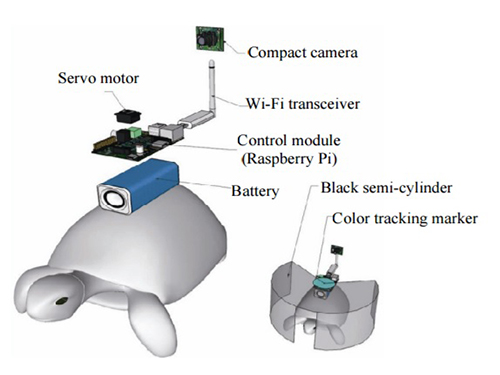 Controlling Turtle Motion with Human Thought
KAIST researchers have developed a technology that can remotely control an animal’s movement with human thought.
In the 2009 blockbuster “Avatar,” a human remotely controls the body of an alien. It does so by injecting human intelligence into a remotely located, biological body. Although still in the realm of science fiction, researchers are nevertheless developing so-called ‘brain-computer interfaces’ (BCIs) following recent advances in electronics and computing. These technologies can ‘read’ and use human thought to control machines, for example, humanoid robots.
New research has demonstrated the possibility of combining a BCI with a device that transmits information from a computer to a brain, or known as a ‘computer-to-brain interface’ (CBI). The combination of these devices could be used to establish a functional link between the brains of different species. Now, researchers from the Korea Advanced Institute of Science and Technology (KAIST) have developed a human-turtle interaction system in which a signal originating from a human brain can affect where a turtle moves.
Unlike previous research that has tried to control animal movement by applying invasive methods, most notably in insects, Professors Phill-Seung Lee of the Mechanical Engineering Department and Sungho Jo of the Computing School propose a conceptual system that can guide an animal’s moving path by controlling its instinctive escape behavior. They chose a turtle because of its cognitive abilities as well as its ability to distinguish different wavelengths of light. Specifically, turtles can recognize a white light source as an open space and so move toward it. They also show specific avoidance behavior to things that might obstruct their view. Turtles also move toward and away from obstacles in their environment in a predictable manner. It was this instinctive, predictable behavior that the researchers induced using the BCI.
The entire human-turtle setup is as follows: A head-mounted display (HMD) is combined with a BCI to immerse the human user in the turtle’s environment. The human operator wears the BCI-HMD system, while the turtle has a 'cyborg system'—consisting of a camera, Wi-Fi transceiver, computer control module, and battery—all mounted on the turtle’s upper shell. Also included on the turtle’s shell is a black semi-cylinder with a slit, which forms the ‘stimulation device.’ This can be turned ±36 degrees via the BCI.
The entire process works like this: the human operator receives images from the camera mounted on the turtle. These real-time video images allow the human operator to decide where the turtle should move. The human provides thought commands that are recognized by the wearable BCI system as electroencephalography (EEG) signals. The BCI can distinguish between three mental states: left, right, and idle. The left and right commands activate the turtle’s stimulation device via Wi-Fi, turning it so that it obstructs the turtle’s view. This invokes its natural instinct to move toward light and change its direction. Finally, the human acquires updated visual feedback from the camera mounted on the shell and in this way continues to remotely navigate the turtle’s trajectory.
The research demonstrates that the animal guiding scheme via BCI can be used in a variety of environments with turtles moving indoors and outdoors on many different surfaces, like gravel and grass, and tackling a range of obstacles, such as shallow water and trees. This technology could be developed to integrate positioning systems and improved augmented and virtual reality techniques, enabling various applications, including devices for military reconnaissance and surveillance.
***
Reference: “Remote Navigation of Turtle by Controlling Instinct Behavior via Human Brain-computer Interface,” Journal of Bionic Engineering, July 2016 (DOI: 10.1016/S1672-6529(16)60322-0)
Depiction of Cyborg System
A human controller influences the turtle’s escape behavior by sending left and right signals via Wi-Fi to a control system on the back of the turtle.
2017.02.21 View 12913
Controlling Turtle Motion with Human Thought
KAIST researchers have developed a technology that can remotely control an animal’s movement with human thought.
In the 2009 blockbuster “Avatar,” a human remotely controls the body of an alien. It does so by injecting human intelligence into a remotely located, biological body. Although still in the realm of science fiction, researchers are nevertheless developing so-called ‘brain-computer interfaces’ (BCIs) following recent advances in electronics and computing. These technologies can ‘read’ and use human thought to control machines, for example, humanoid robots.
New research has demonstrated the possibility of combining a BCI with a device that transmits information from a computer to a brain, or known as a ‘computer-to-brain interface’ (CBI). The combination of these devices could be used to establish a functional link between the brains of different species. Now, researchers from the Korea Advanced Institute of Science and Technology (KAIST) have developed a human-turtle interaction system in which a signal originating from a human brain can affect where a turtle moves.
Unlike previous research that has tried to control animal movement by applying invasive methods, most notably in insects, Professors Phill-Seung Lee of the Mechanical Engineering Department and Sungho Jo of the Computing School propose a conceptual system that can guide an animal’s moving path by controlling its instinctive escape behavior. They chose a turtle because of its cognitive abilities as well as its ability to distinguish different wavelengths of light. Specifically, turtles can recognize a white light source as an open space and so move toward it. They also show specific avoidance behavior to things that might obstruct their view. Turtles also move toward and away from obstacles in their environment in a predictable manner. It was this instinctive, predictable behavior that the researchers induced using the BCI.
The entire human-turtle setup is as follows: A head-mounted display (HMD) is combined with a BCI to immerse the human user in the turtle’s environment. The human operator wears the BCI-HMD system, while the turtle has a 'cyborg system'—consisting of a camera, Wi-Fi transceiver, computer control module, and battery—all mounted on the turtle’s upper shell. Also included on the turtle’s shell is a black semi-cylinder with a slit, which forms the ‘stimulation device.’ This can be turned ±36 degrees via the BCI.
The entire process works like this: the human operator receives images from the camera mounted on the turtle. These real-time video images allow the human operator to decide where the turtle should move. The human provides thought commands that are recognized by the wearable BCI system as electroencephalography (EEG) signals. The BCI can distinguish between three mental states: left, right, and idle. The left and right commands activate the turtle’s stimulation device via Wi-Fi, turning it so that it obstructs the turtle’s view. This invokes its natural instinct to move toward light and change its direction. Finally, the human acquires updated visual feedback from the camera mounted on the shell and in this way continues to remotely navigate the turtle’s trajectory.
The research demonstrates that the animal guiding scheme via BCI can be used in a variety of environments with turtles moving indoors and outdoors on many different surfaces, like gravel and grass, and tackling a range of obstacles, such as shallow water and trees. This technology could be developed to integrate positioning systems and improved augmented and virtual reality techniques, enabling various applications, including devices for military reconnaissance and surveillance.
***
Reference: “Remote Navigation of Turtle by Controlling Instinct Behavior via Human Brain-computer Interface,” Journal of Bionic Engineering, July 2016 (DOI: 10.1016/S1672-6529(16)60322-0)
Depiction of Cyborg System
A human controller influences the turtle’s escape behavior by sending left and right signals via Wi-Fi to a control system on the back of the turtle.
2017.02.21 View 12913 -
 Mystery of Biological Plastic Synthesis Machinery Unveiled
Plastics and other polymers are used every day. These polymers are mostly made from fossil resources by refining petrochemicals. On the other hand, many microorganisms naturally synthesize polyesters known as polyhydroxyalkanoates (PHAs) as distinct granules inside cells.
PHAs are a family of microbial polyesters that have attracted much attention as biodegradable and biocompatible plastics and elastomers that can substitute petrochemical counterparts. There have been numerous papers and patents on gene cloning and metabolic engineering of PHA biosynthetic machineries, biochemical studies, and production of PHAs; simple Google search with “polyhydroxyalkanoates” yielded returns of 223,000 document pages. PHAs have always been considered amazing examples of biological polymer synthesis. It is astounding to see PHAs of 500 kDa to sometimes as high as 10,000 kDa can be synthesized in vivo by PHA synthase, the key polymerizing enzyme in PHA biosynthesis. They have attracted great interest in determining the crystal structure of PHA synthase over the last 30 years, but unfortunately without success. Thus, the characteristics and molecular mechanisms of PHA synthase were under a dark veil.
In two papers published back-to-back in Biotechnology Journal online on November 30, 2016, a Korean research team led by Professor Kyung-Jin Kim at Kyungpook National University and Distinguished Professor Sang Yup Lee at the Korea Advanced Institute of Science and Technology (KAIST) described the crystal structure of PHA synthase from Ralstonia eutropha, the best studied bacterium for PHA production, and reported the structural basis for the detailed molecular mechanisms of PHA biosynthesis. The crystal structure has been deposited to Protein Data Bank in February 2016. After deciphering the crystal structure of the catalytic domain of PHA synthase, in addition to other structural studies on whole enzyme and related proteins, the research team also performed experiments to elucidate the mechanisms of the enzyme reaction, validating detailed structures, enzyme engineering, and also N-terminal domain studies among others.
Through several biochemical studies based on crystal structure, the authors show that PHA synthase exists as a dimer and is divided into two distinct domains, the N-terminal domain (RePhaC1ND) and the C-terminal domain (RePhaC1CD). The RePhaC1CD catalyzes the polymerization reaction via a non-processive ping-pong mechanism using a Cys-His-Asp catalytic triad. The two catalytic sites of the RePhaC1CD dimer are positioned 33.4 Å apart, suggesting that the polymerization reaction occurs independently at each site. This study also presents the structure-based mechanisms for substrate specificities of various PHA synthases from different classes.
Professor Sang Yup Lee, who has worked on this topic for more than 20 years, said,
“The results and information presented in these two papers have long been awaited not only in the PHA community, but also metabolic engineering, bacteriology/microbiology, and in general biological sciences communities. The structural information on PHA synthase together with the recently deciphered reaction mechanisms will be valuable for understanding the detailed mechanisms of biosynthesizing this important energy/redox storage material, and also for the rational engineering of PHA synthases to produce designer bioplastics from various monomers more efficiently.”
Indeed, these two papers published in Biotechnology Journal finally reveal the 30-year mystery of machinery of biological polyester synthesis, and will serve as the essential compass in creating designer and more efficient bioplastic machineries.
References:
Jieun Kim, Yeo-Jin Kim, So Young Choi, Sang Yup Lee and Kyung-Jin Kim. “Crystal structure of Ralstonia eutropha polyhydroxyalkanoate synthase C-terminal domain and reaction mechanisms” Biotechnology Journal DOI: 10.1002/biot.201600648
http://onlinelibrary.wiley.com/doi/10.1002/biot.201600648/abstract
Yeo-Jin Kim, So Young Choi, Jieun Kim, Kyeong Sik Jin, Sang Yup Lee and Kyung-Jin Kim. “Structure and function of the N-terminal domain of Ralstonia eutropha polyhydroxyalkanoate synthase, and the proposed structure and mechanisms of the whole enzyme” Biotechnology Journal DOI: 10.1002/biot.201600649
http://onlinelibrary.wiley.com/doi/10.1002/biot.201600649/abstract
2016.12.02 View 8172
Mystery of Biological Plastic Synthesis Machinery Unveiled
Plastics and other polymers are used every day. These polymers are mostly made from fossil resources by refining petrochemicals. On the other hand, many microorganisms naturally synthesize polyesters known as polyhydroxyalkanoates (PHAs) as distinct granules inside cells.
PHAs are a family of microbial polyesters that have attracted much attention as biodegradable and biocompatible plastics and elastomers that can substitute petrochemical counterparts. There have been numerous papers and patents on gene cloning and metabolic engineering of PHA biosynthetic machineries, biochemical studies, and production of PHAs; simple Google search with “polyhydroxyalkanoates” yielded returns of 223,000 document pages. PHAs have always been considered amazing examples of biological polymer synthesis. It is astounding to see PHAs of 500 kDa to sometimes as high as 10,000 kDa can be synthesized in vivo by PHA synthase, the key polymerizing enzyme in PHA biosynthesis. They have attracted great interest in determining the crystal structure of PHA synthase over the last 30 years, but unfortunately without success. Thus, the characteristics and molecular mechanisms of PHA synthase were under a dark veil.
In two papers published back-to-back in Biotechnology Journal online on November 30, 2016, a Korean research team led by Professor Kyung-Jin Kim at Kyungpook National University and Distinguished Professor Sang Yup Lee at the Korea Advanced Institute of Science and Technology (KAIST) described the crystal structure of PHA synthase from Ralstonia eutropha, the best studied bacterium for PHA production, and reported the structural basis for the detailed molecular mechanisms of PHA biosynthesis. The crystal structure has been deposited to Protein Data Bank in February 2016. After deciphering the crystal structure of the catalytic domain of PHA synthase, in addition to other structural studies on whole enzyme and related proteins, the research team also performed experiments to elucidate the mechanisms of the enzyme reaction, validating detailed structures, enzyme engineering, and also N-terminal domain studies among others.
Through several biochemical studies based on crystal structure, the authors show that PHA synthase exists as a dimer and is divided into two distinct domains, the N-terminal domain (RePhaC1ND) and the C-terminal domain (RePhaC1CD). The RePhaC1CD catalyzes the polymerization reaction via a non-processive ping-pong mechanism using a Cys-His-Asp catalytic triad. The two catalytic sites of the RePhaC1CD dimer are positioned 33.4 Å apart, suggesting that the polymerization reaction occurs independently at each site. This study also presents the structure-based mechanisms for substrate specificities of various PHA synthases from different classes.
Professor Sang Yup Lee, who has worked on this topic for more than 20 years, said,
“The results and information presented in these two papers have long been awaited not only in the PHA community, but also metabolic engineering, bacteriology/microbiology, and in general biological sciences communities. The structural information on PHA synthase together with the recently deciphered reaction mechanisms will be valuable for understanding the detailed mechanisms of biosynthesizing this important energy/redox storage material, and also for the rational engineering of PHA synthases to produce designer bioplastics from various monomers more efficiently.”
Indeed, these two papers published in Biotechnology Journal finally reveal the 30-year mystery of machinery of biological polyester synthesis, and will serve as the essential compass in creating designer and more efficient bioplastic machineries.
References:
Jieun Kim, Yeo-Jin Kim, So Young Choi, Sang Yup Lee and Kyung-Jin Kim. “Crystal structure of Ralstonia eutropha polyhydroxyalkanoate synthase C-terminal domain and reaction mechanisms” Biotechnology Journal DOI: 10.1002/biot.201600648
http://onlinelibrary.wiley.com/doi/10.1002/biot.201600648/abstract
Yeo-Jin Kim, So Young Choi, Jieun Kim, Kyeong Sik Jin, Sang Yup Lee and Kyung-Jin Kim. “Structure and function of the N-terminal domain of Ralstonia eutropha polyhydroxyalkanoate synthase, and the proposed structure and mechanisms of the whole enzyme” Biotechnology Journal DOI: 10.1002/biot.201600649
http://onlinelibrary.wiley.com/doi/10.1002/biot.201600649/abstract
2016.12.02 View 8172 -
 Professor Lee Co-chairs the Global Future Councils on Biotechnology of the WEF
The World Economic Forum (WEF) established a new global network of the world’s leading experts, “The Annual Meeting of the Global Future Councils,” to explore innovative solutions for the most pressing global challenges. The Councils’ first meeting took place on November 13-14, 2016, in Dubai, the United Arab Emirates (UAE). Some 25 nations joined as member states. The Councils have 35 committees.
Over 700 global leaders in business, government, civil society and academia gathered at the inaugural meeting to “develop ideas and strategies to prepare the world for the Fourth Industrial Revolution, with topics including smart cities, robotics, and the future of mobility,” according to a statement issued by the WEF.
Distinguished Professor Sang Yup Lee of Chemical and Biomolecular Engineering at KAIST was appointed to co-chair one of the Councils' committees, The Annual Meeting of the Global Future Councils on Biotechnology, for two years. The other chairperson is Dr. Feng Zhang, a professor of Biomedical Engineering at the Massachusetts Institute of Technology (MIT), who played a critical role in the development of optogenetics and CRISPR technologies.
The Biotechnology Committee consists of 24 globally recognized professionals in life sciences, law, ethics and policy including Thomas Connelly, the executive director of the American Chemical Society, Tina Fano, the executive vice president of Novozymes, and Mostafa Ronaghi, the chief technology officer of Illumina.
Professor Lee also serves as a committee member of The Annual Meeting of the Global Future Councils on the Fourth Industrial Revolution.
“Life sciences and engineering will receive more attention as a key element of the Fourth Industrial Revolution that the global society as a whole has been experiencing now. Together with thought leaders gathered worldwide, I will join the international community’s concerted efforts to address issues of importance that impact greatly on the future of humanity,” Professor Lee said.
In addition, Professor Lee received the James E. Bailey Award 2016 from The Society for Biological Engineering on November 15, 2016. He is the first Asian researcher to be recognized for his contributions to the field of biotechnology.
2016.11.15 View 6944
Professor Lee Co-chairs the Global Future Councils on Biotechnology of the WEF
The World Economic Forum (WEF) established a new global network of the world’s leading experts, “The Annual Meeting of the Global Future Councils,” to explore innovative solutions for the most pressing global challenges. The Councils’ first meeting took place on November 13-14, 2016, in Dubai, the United Arab Emirates (UAE). Some 25 nations joined as member states. The Councils have 35 committees.
Over 700 global leaders in business, government, civil society and academia gathered at the inaugural meeting to “develop ideas and strategies to prepare the world for the Fourth Industrial Revolution, with topics including smart cities, robotics, and the future of mobility,” according to a statement issued by the WEF.
Distinguished Professor Sang Yup Lee of Chemical and Biomolecular Engineering at KAIST was appointed to co-chair one of the Councils' committees, The Annual Meeting of the Global Future Councils on Biotechnology, for two years. The other chairperson is Dr. Feng Zhang, a professor of Biomedical Engineering at the Massachusetts Institute of Technology (MIT), who played a critical role in the development of optogenetics and CRISPR technologies.
The Biotechnology Committee consists of 24 globally recognized professionals in life sciences, law, ethics and policy including Thomas Connelly, the executive director of the American Chemical Society, Tina Fano, the executive vice president of Novozymes, and Mostafa Ronaghi, the chief technology officer of Illumina.
Professor Lee also serves as a committee member of The Annual Meeting of the Global Future Councils on the Fourth Industrial Revolution.
“Life sciences and engineering will receive more attention as a key element of the Fourth Industrial Revolution that the global society as a whole has been experiencing now. Together with thought leaders gathered worldwide, I will join the international community’s concerted efforts to address issues of importance that impact greatly on the future of humanity,” Professor Lee said.
In addition, Professor Lee received the James E. Bailey Award 2016 from The Society for Biological Engineering on November 15, 2016. He is the first Asian researcher to be recognized for his contributions to the field of biotechnology.
2016.11.15 View 6944 -
 Doctoral Student Receives the Best Paper Award from the International Metabolic Engineering Conference 2016
So Young Choi, a Ph.D. candidate at the Department of Chemical and Biomolecular Engineering at KAIST, received the Student and Young Investigator Poster Award at the 11th International Metabolic Engineering Conference held in Awaji, Japan on June 26-30.
Choi received the award for her research on one-step fermentative production of Poly(lactate-co-glycolate) (PLGA) from carbohydrates in Escherichia coli, which was published in the April 2016 issue of Nature Biotechnology.
In her paper, she presented a novel technology to synthesize PLGA, a non-natural copolymer, through a biological production process. Because of its biodegradability, non-toxicity, and biocompatibility, PLGA is widely used in biomedical and therapeutic applications, including surgical sutures, prosthetic devices, drug delivery, and tissue engineering.
Employing a metabolic engineering approach, Choi manipulated the metabolic pathway of an Escherichia coli bacterium to convert glucose and xylose into the biosynthesis of PLGA within the cell. Previously, PLGA could be obtained only through chemical synthesis.
Choi said, “I’m thrilled to receive an award from a flagship conference of my research field. Mindful of this recognition, I will continue my research to produce meaningful results, thereby contributing to the development of science and technology in Korea.”
The International Metabolic Engineering Conference is a leading professional gathering where state-of-the-art developments and achievements made in the field of metabolic engineering are shared. With the participation of about 400 professionals from all around the world, the conference participants discussed this year’s theme of “Design, Synthesis and System Integration for Metabolic Engineering.”
2016.07.07 View 9267
Doctoral Student Receives the Best Paper Award from the International Metabolic Engineering Conference 2016
So Young Choi, a Ph.D. candidate at the Department of Chemical and Biomolecular Engineering at KAIST, received the Student and Young Investigator Poster Award at the 11th International Metabolic Engineering Conference held in Awaji, Japan on June 26-30.
Choi received the award for her research on one-step fermentative production of Poly(lactate-co-glycolate) (PLGA) from carbohydrates in Escherichia coli, which was published in the April 2016 issue of Nature Biotechnology.
In her paper, she presented a novel technology to synthesize PLGA, a non-natural copolymer, through a biological production process. Because of its biodegradability, non-toxicity, and biocompatibility, PLGA is widely used in biomedical and therapeutic applications, including surgical sutures, prosthetic devices, drug delivery, and tissue engineering.
Employing a metabolic engineering approach, Choi manipulated the metabolic pathway of an Escherichia coli bacterium to convert glucose and xylose into the biosynthesis of PLGA within the cell. Previously, PLGA could be obtained only through chemical synthesis.
Choi said, “I’m thrilled to receive an award from a flagship conference of my research field. Mindful of this recognition, I will continue my research to produce meaningful results, thereby contributing to the development of science and technology in Korea.”
The International Metabolic Engineering Conference is a leading professional gathering where state-of-the-art developments and achievements made in the field of metabolic engineering are shared. With the participation of about 400 professionals from all around the world, the conference participants discussed this year’s theme of “Design, Synthesis and System Integration for Metabolic Engineering.”
2016.07.07 View 9267 -
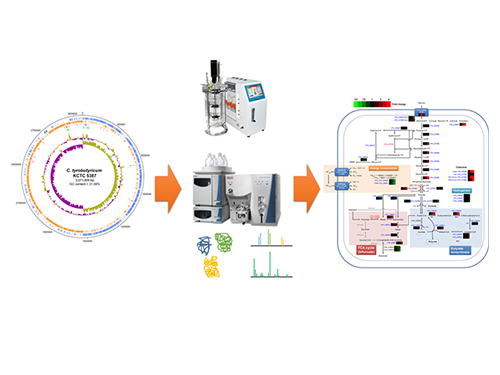 Unveiling the Distinctive Features of Industrial Microorganism
KAIST researchers have sequenced the whole genome of Clostridium tyrobutyricum, which has a higher tolerance to toxic chemicals, such as 1-butanol, compared to other clostridial bacterial strains.
Clostridium tyrobutyricum, a Gram-positive, anaerobic spore-forming bacterium, is considered a promising industrial host strain for the production of various chemicals including butyric acid which has many applications in different industries such as a precursor to biofuels. Despite such potential, C. tyrobutyricum has received little attention, mainly due to a limited understanding of its genotypic and metabolic characteristics at the genome level.
A Korean research team headed by Distinguished Professor Sang Yup Lee of the Chemical and Biomolecular Engineering Department at the Korea Advanced Institute of Science and Technology (KAIST) deciphered the genome sequence of C. tyrobutyricum and its proteome profiles during the course of batch fermentation. As a result, the research team learned that the bacterium is not only capable of producing a large amount of butyric acid but also can tolerate toxic compounds such as 1-butanol. The research results were published in mBio on June 14, 2016.
The team adopted a genoproteomic approach, combining genomics and proteomics, to investigate the metabolic features of C. tyrobutyricum. Unlike Clostridium acetobutylicum, the most widely used organism for 1-butanol production, C. tyrobutyricum has a novel butyrate-producing pathway and various mechanisms for energy conservation under anaerobic conditions. The expression of various metabolic genes, including those involved in butyrate formation, was analyzed using the “shotgun” proteome approach.
To date, the bio-based production of 1-butanol, a next-generation biofuel, has relied on several clostridial hosts including C. acetobutylicum and C. beijerinckii. However, these organisms have a low tolerance against 1-butanol even though they are naturally capable of producing it. C. tyrobutyricum cannot produce 1-butanol itself, but has a higher 1-butanol-tolerance and rapid uptake of monosaccharides, compared to those two species.
The team identified most of the genes involved in the central metabolism of C. tyrobutyricum from the whole-genome and shotgun proteome data, and this study will accelerate the bacterium’s engineering to produce useful chemicals including butyric acid and 1-butanol, replacing traditional bacterial hosts.
Professor Lee said,
“The unique metabolic features and energy conservation mechanisms of C. tyrobutyricum can be employed in the various microbial hosts we have previously developed to further improve their productivity and yield. Moreover, findings on C. tyrobutyricum revealed by this study will be the first step to directly engineer this bacterium.”
Director Jin-Woo Kim at the Platform Technology Division of the Ministry of Science, ICT and Future Planning of Korea, who oversees the Technology Development Program to Solve Climate Change, said,
“Over the years, Professor Lee’s team has researched the development of a bio-refinery system to produce natural and non-natural chemicals with the systems metabolic engineering of microorganisms. They were able to design strategies for the development of diverse industrial microbial strains to produce useful chemicals from inedible biomass-based carbon dioxide fixation. We believe the efficient production of butyric acid using a metabolic engineering approach will play an important role in the establishment of a bioprocess for chemical production.”
The title of the research paper is “Deciphering Clostridium tyrobutyricum Metabolism Based on the Who-Genome Sequence and Proteome Analyses.” (DOI: 10.1128/mBio.00743-16)
The lead authors are Joungmin Lee, a post-doctoral fellow in the BioProcess Research Center at KAIST, currently working in CJ CheilJedang Research Institute; Yu-Sin Jang, a research fellow in the BioProcess Research Center at KAIST, currently working at Gyeongsang National University as an assistant professor; and Mee-Jung Han, an assistant professor in the Environmental Engineering and Energy Department at Dongyang University. Jin Young Kim, a senior researcher at the Korea Basic Science Institute, also participated in the research.
This research was supported by the Technology Development Program to Solve Climate Change’s research project entitled “Systems Metabolic Engineering for Biorefineries” from the Ministry of Science, ICT and Future Planning through the National Research Foundation of Korea (NRF-2012M1A2A2026556).
Schematic Diagram of C. tyrobutyricum’s Genome Sequence and Its Proteome Profiles
The picture below shows the complete genome sequence, global protein expression profiles, and the genome-based metabolic characteristics during batch fermentation of C. tyrobutyricum.
2016.06.20 View 8568
Unveiling the Distinctive Features of Industrial Microorganism
KAIST researchers have sequenced the whole genome of Clostridium tyrobutyricum, which has a higher tolerance to toxic chemicals, such as 1-butanol, compared to other clostridial bacterial strains.
Clostridium tyrobutyricum, a Gram-positive, anaerobic spore-forming bacterium, is considered a promising industrial host strain for the production of various chemicals including butyric acid which has many applications in different industries such as a precursor to biofuels. Despite such potential, C. tyrobutyricum has received little attention, mainly due to a limited understanding of its genotypic and metabolic characteristics at the genome level.
A Korean research team headed by Distinguished Professor Sang Yup Lee of the Chemical and Biomolecular Engineering Department at the Korea Advanced Institute of Science and Technology (KAIST) deciphered the genome sequence of C. tyrobutyricum and its proteome profiles during the course of batch fermentation. As a result, the research team learned that the bacterium is not only capable of producing a large amount of butyric acid but also can tolerate toxic compounds such as 1-butanol. The research results were published in mBio on June 14, 2016.
The team adopted a genoproteomic approach, combining genomics and proteomics, to investigate the metabolic features of C. tyrobutyricum. Unlike Clostridium acetobutylicum, the most widely used organism for 1-butanol production, C. tyrobutyricum has a novel butyrate-producing pathway and various mechanisms for energy conservation under anaerobic conditions. The expression of various metabolic genes, including those involved in butyrate formation, was analyzed using the “shotgun” proteome approach.
To date, the bio-based production of 1-butanol, a next-generation biofuel, has relied on several clostridial hosts including C. acetobutylicum and C. beijerinckii. However, these organisms have a low tolerance against 1-butanol even though they are naturally capable of producing it. C. tyrobutyricum cannot produce 1-butanol itself, but has a higher 1-butanol-tolerance and rapid uptake of monosaccharides, compared to those two species.
The team identified most of the genes involved in the central metabolism of C. tyrobutyricum from the whole-genome and shotgun proteome data, and this study will accelerate the bacterium’s engineering to produce useful chemicals including butyric acid and 1-butanol, replacing traditional bacterial hosts.
Professor Lee said,
“The unique metabolic features and energy conservation mechanisms of C. tyrobutyricum can be employed in the various microbial hosts we have previously developed to further improve their productivity and yield. Moreover, findings on C. tyrobutyricum revealed by this study will be the first step to directly engineer this bacterium.”
Director Jin-Woo Kim at the Platform Technology Division of the Ministry of Science, ICT and Future Planning of Korea, who oversees the Technology Development Program to Solve Climate Change, said,
“Over the years, Professor Lee’s team has researched the development of a bio-refinery system to produce natural and non-natural chemicals with the systems metabolic engineering of microorganisms. They were able to design strategies for the development of diverse industrial microbial strains to produce useful chemicals from inedible biomass-based carbon dioxide fixation. We believe the efficient production of butyric acid using a metabolic engineering approach will play an important role in the establishment of a bioprocess for chemical production.”
The title of the research paper is “Deciphering Clostridium tyrobutyricum Metabolism Based on the Who-Genome Sequence and Proteome Analyses.” (DOI: 10.1128/mBio.00743-16)
The lead authors are Joungmin Lee, a post-doctoral fellow in the BioProcess Research Center at KAIST, currently working in CJ CheilJedang Research Institute; Yu-Sin Jang, a research fellow in the BioProcess Research Center at KAIST, currently working at Gyeongsang National University as an assistant professor; and Mee-Jung Han, an assistant professor in the Environmental Engineering and Energy Department at Dongyang University. Jin Young Kim, a senior researcher at the Korea Basic Science Institute, also participated in the research.
This research was supported by the Technology Development Program to Solve Climate Change’s research project entitled “Systems Metabolic Engineering for Biorefineries” from the Ministry of Science, ICT and Future Planning through the National Research Foundation of Korea (NRF-2012M1A2A2026556).
Schematic Diagram of C. tyrobutyricum’s Genome Sequence and Its Proteome Profiles
The picture below shows the complete genome sequence, global protein expression profiles, and the genome-based metabolic characteristics during batch fermentation of C. tyrobutyricum.
2016.06.20 View 8568