research
Professor Gou Young Koh in the Graduate School of Medical Science and Engineering and his team have identified a new mechanism involved in the development and progression of glaucoma, and found a potential therapeutic option to treat it. Glaucoma is the second cause of irreversible blindness, after cataracts. It affects about 3.5% of the world population aged 40 to 80.
Professor Koh also serves as the director of the Center for Vascular Research at the Institute for Basic Science. The IBS said the study, published in the Journal of Clinical Investigation, is expected to help the development of therapies to treat primary open-angle glaucoma (POAG), which counts for three quarters of all glaucoma patients.
One of the most important risk factors for glaucoma is the increased pressure inside the eye. A liquid called aqueous humor is constantly produced and drained out from the eye. It transports nutrients and inflates the eye giving it a roughly spherical shape. However, if this fluid cannot flow out of the eye chambers freely, an increase in intraocular pressure can damage the optic nerve, leading to vision loss. The precise mechanism of elevated resistance to aqueous humor outflow remains unclear, and although the current treatments for glaucoma tackle the production and outflow of aqueous humor, their outcomes are still poor.
A component of the eye that plays a fundamental role in draining out the aqueous humor is Schlemm's canal. It collects the aqueous humor and mediates its transfer from the eye chambers to blood circulation. The cells on the walls of the canal, endothelial cells, ship the liquid from the inner to the outer side in “packages”, called vacuoles. As the shape and number of the vacuoles reflects the outflow performance, several giant vacuoles are expected in the normal outflow process.
The team explained how imbalances in Schlemm's canal significantly increase the risk of glaucoma. They showed that an important regulator for canal functionality is the angiopoietin-Tie2 system. Angiopoietins, such as Ang1 and Ang2, are proteins important for the growth of new blood vessels and Tie2 is the receptor that binds them. It is known that the angiopoietin-Tie2 system plays a role in Schlemm’s canal formation, as Tie2 mutations or angiopoietin absence result in congenital glaucoma. However, this study clarified that it is also critically important during adulthood.
The researchers reported that adult mice deficient in Tie2 suffer from an elevated intraocular pressure, retinal neuronal damage and partial visual impairment. Moreover, they had a markedly decreased number of giant vacuoles inside Schlemm’s canal endothelial cells, which indicate a poor aqueous humor drainage.
The scientists also investigated if and how this process changes in older mice, as aging is a major risk factor for glaucoma, and showed that aged mice experience reduced levels of giant vacuoles, Tie2, Ang1, and Ang2, as well as other proteins connected with the angiopoietin-Tie2 pathway, like Prox1.
To test whether Tie2 activation could shift the situation, the researchers tested the antibody ABTAA (Ang2-binding and Tie2-activating antibody). They injected it in one eye of mice, while the other eye of the same mice functioned as the negative control. After one week, levels of Tie2 and Prox1, number and diameter of giant vacuoles in Schlemm’s canals increased in the ABTAA-treated eyes compared to control eyes. The researchers observed a similar outcome with decreased intraocular pressure when ABTAA was injected to the eyes of mice suffering from POAG with regressed Schlemm’s canals, indicating that this antibody might be considered as a therapeutic option.
"Slow development of glaucoma treatments is partly due to the poor understanding of the underlying pathogenesis," said Professor Koh, the corresponding author of the study. "We hope that identifying the critical role of the angiopoietin-Tie2 system in adult Schlemm’s canals will bring a significant boost in the development of therapeutics."

Figure 1: Schlemm's canal position inside the eye.
Schlemm's canal (green) plays a fundamental role in draining the aqueous humor (white arrows) from the anterior chamber of the eye to blood circulation. If the aqueous humor is not able to flow out freely, elevated intraocular pressure damages the optical nerve causing glaucoma and eventually blindness.
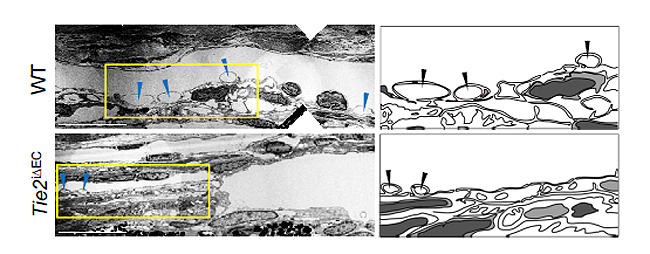
Figure 2: Electron microscope images reveal how the aqueous humor is packaged in vacuoles (arrowheads) inside the cells forming the walls of Schlemm's canal.
Aging and glaucoma cause the number and size of giant vacuoles to decrease, meaning that the aqueous humor outflow is compromised. The images compare the giant vacuoles in Schlemm's canals of a healthy mouse (top) and a mouse lacking Tie2 (bottom)

Figure 3: The Ang2-binding and Tie2-activating antibody (ABTAA) rejuvenates the eye of aged mice and rescues them from glaucoma.
Aging causes a reduction of the protein Tie2, a risk factor for increased intraocular pressure and glaucoma. In this experiment, one eye of mice lacking Ang1 and Ang2 was injected with the premixed ABTAA and Ang2, while the other eye was used as negative control. The researchers observed an increase in the area of Schlemm’s canal, together with higher levels of Tie2 (red) and lower intraocular pressure, suggesting that ABTAA restores the canal's functionality. The image includes the transcription factor Prox1 (green) and CD144 (blue), a protein present at the junctions between cells that form the wall of the canal. The angiopoietin-Tie2 system and Prox1 are linked by a vicious circle: the less Tie2 and Ang2, the less Prox1, leading to Schlemm's canal damage, increase in intraocular pressure, and acceleration of glaucoma progression.