protein
-
 Mechanism in regulation of cancer-related key enzyme, ATM, for DNA damage and repair revealed
Professor Kwang-Wook Choi
A research team led by Professor Kwang-Wook Choi and Dr. Seong-Tae Hong from the Department of Biological Sciences at KAIST has successfully investigated the operational mechanism of the protein Ataxia Telangiectasia Mutated (ATM), an essential protein to the function of a crucial key enzyme that repairs the damaged DNA which stores biometric information. The results were published on December 19th Nature Communications online edition.
All organisms, including humans, constantly strive to protect the information within their DNA from damages posed by a number of factors, such as carbonized materials in our daily food intake, radioactive materials such as radon emitting from the cement of buildings or ultraviolet of the sunlight, which could be a trigger for cancer.
In order to keep the DNA information safe, the organisms are always carrying out complex and sophisticated DNA repair work, which involves the crucial DNA damage repair protein ATM. Consequently, a faulty ATM leads to higher risks of cancer.
Until now, academia predicted that the Translationally Controlled Tumor Protein (TCTP) will play an important role in regulating the function of ATM. However, since most of main research regarding TCTP has only been conducted in cultured cells, it was unable to identify exactly what mechanisms TCTP employs to control ATM.
The KAIST research team identified that TCTP can combine with ATM or increase the enzymatic activity of ATM. In addition, Drosophilia, one of the most widely used model organisms for molecular genetics, has been used to identify that TCTP and ATM play a very important role in repairing the DNA damaged by radiation. This information has allowed the researchers to establish TCTP’s essential function in maintaining the DNA information in cell cultures and even in higher organisms, and to provide specific and important clues to the regulation of ATM by TCTP.
Professor Kwang-Wook Choi said, “Our research is a good example that basic research using Drosophilia can make important contributions to understanding the process of diseases, such as cancer, and to developing adequate treatment.”
The research has been funded by the Ministry of Science, ICT and Future Planning, Republic of Korea, and the National Research Foundation of Korea.
Figure 1. When the amount of TCTP protein is reduced, cells of the Drosophila's eye are abnormally deformed by radiation. Scale bars = 200mm
Figure 2. When the amount of TCTP protein is reduced, the chromosomes of Drosophilia are easily broken by radiation. Scale bars = 10 mm.
Figure 3. When gene expressions of TCTP and ATM are reduced, large defects occur in the normal development of the eye. (Left: normal Drosophilia's eye, right: development-deficient eye)
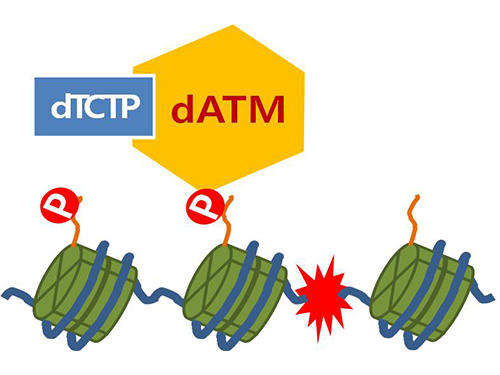
Figure 4. ATM marks the position of the broken DNA, with TCTP helping to facilitate this reaction. DNA (blue line) within the cell nucleus is coiled around the histone protein (green cylinder). When DNA is broken, ATM protein attaches a phosphate group (P). Multiple DNA repair protein recognizes the phosphate as a signal that requires repair and gathers at the site.
2014.01.07 View 10075
Mechanism in regulation of cancer-related key enzyme, ATM, for DNA damage and repair revealed
Professor Kwang-Wook Choi
A research team led by Professor Kwang-Wook Choi and Dr. Seong-Tae Hong from the Department of Biological Sciences at KAIST has successfully investigated the operational mechanism of the protein Ataxia Telangiectasia Mutated (ATM), an essential protein to the function of a crucial key enzyme that repairs the damaged DNA which stores biometric information. The results were published on December 19th Nature Communications online edition.
All organisms, including humans, constantly strive to protect the information within their DNA from damages posed by a number of factors, such as carbonized materials in our daily food intake, radioactive materials such as radon emitting from the cement of buildings or ultraviolet of the sunlight, which could be a trigger for cancer.
In order to keep the DNA information safe, the organisms are always carrying out complex and sophisticated DNA repair work, which involves the crucial DNA damage repair protein ATM. Consequently, a faulty ATM leads to higher risks of cancer.
Until now, academia predicted that the Translationally Controlled Tumor Protein (TCTP) will play an important role in regulating the function of ATM. However, since most of main research regarding TCTP has only been conducted in cultured cells, it was unable to identify exactly what mechanisms TCTP employs to control ATM.
The KAIST research team identified that TCTP can combine with ATM or increase the enzymatic activity of ATM. In addition, Drosophilia, one of the most widely used model organisms for molecular genetics, has been used to identify that TCTP and ATM play a very important role in repairing the DNA damaged by radiation. This information has allowed the researchers to establish TCTP’s essential function in maintaining the DNA information in cell cultures and even in higher organisms, and to provide specific and important clues to the regulation of ATM by TCTP.
Professor Kwang-Wook Choi said, “Our research is a good example that basic research using Drosophilia can make important contributions to understanding the process of diseases, such as cancer, and to developing adequate treatment.”
The research has been funded by the Ministry of Science, ICT and Future Planning, Republic of Korea, and the National Research Foundation of Korea.
Figure 1. When the amount of TCTP protein is reduced, cells of the Drosophila's eye are abnormally deformed by radiation. Scale bars = 200mm
Figure 2. When the amount of TCTP protein is reduced, the chromosomes of Drosophilia are easily broken by radiation. Scale bars = 10 mm.
Figure 3. When gene expressions of TCTP and ATM are reduced, large defects occur in the normal development of the eye. (Left: normal Drosophilia's eye, right: development-deficient eye)
Figure 4. ATM marks the position of the broken DNA, with TCTP helping to facilitate this reaction. DNA (blue line) within the cell nucleus is coiled around the histone protein (green cylinder). When DNA is broken, ATM protein attaches a phosphate group (P). Multiple DNA repair protein recognizes the phosphate as a signal that requires repair and gathers at the site.
2014.01.07 View 10075 -
 Therapy developed to induce Angiogenesis of Retina
- Junyeop Lee, Graduate School of Medical Sciences and Engineering
- Research results expected to be applied for treatment of diabetic retinopathy
A major clue to treatment of retinovascular disease, which causes blindness, has been found. The key to protection of the retinal nerve is the angiogenic protein that promotes healthy retinal vessel growth around the retina, which usually does not receive blood supply readily. This research offers a beginning to the possible improvement of therapy for diabetic retinopathy1 and retinopathy of prematurity2. Also important to the research is the fact that the ophthalmology specialist researcher, currently undergoing professional training, provided the results.
KAIST Graduate School of Medical Sciences and Engineering’s Junyeop Lee is the opthalmology specialist, who carried out the research under supervision by academic advisers Gyuyeong Go and Wookjun Yoo. The Ministry of Science, ICT and Future Planning as well as the National Research Foundation of Korea have funded his research. The research results have been published as a cover paper on ‘Science Translational Medicine’ on 18th August. This journal is a sister publication of Science, which is prestigious in the field of translational medicine that ties the basic science with clinical medicine. (Thesis title: Angiopoietin-1 Guides Directional Angiogenesis Through Integrin αvβ5 Signaling for Recovery of Ischemic Retinopathy)
The traditional treatment of diabetic retinopathy includes laser photocoagulation to destroy the retinal tissues or antibody therapeutics, which prevents vessel proliferation and blood leaking. The advantage of antibody therapeutics3 is that it retains the retinal nerves, however, it is not the fundamental solution but merely a temporary one, which requires repeated treatments.
The research team identified that Angiopoietin-14 protein, known as essential for growth and stabilization of vessels, also plays an important role in retinal vessel growth. The protein protects the retinal nerves, as well as provides improvement for retinal ischemia5 that is the root cause of vision loss due to retinal hemorrhages. It is expected to become a key to finding fundamental treatment method – by providing sufficient blood supply to the retina, thereby preserving the retinal nerve functions.
The results show that administration of Angiopoietin-1 to retinopathy mouse model promotes growth of healthy vessel growth, further preventing abnormal vessel growth, retinal hemorrhage and vision loss due to retinal ischemia.
Junyeop Lee said, “This research has identified that Angiopoietin-1 is an important factor in retinal vessel generation and stabilization. The paradigm will shift from traditional treatment method, which prevents vessel growth, to a new method that generates healthy vessels and strengthens vessel functions.”
1 Diabetic retinopathy: This retinovascular disease is a diabetic complication caused by insufficient blood supply. It is the major causes of blindness in adults.
2 Retinopathy of prematurity: The retinal vascular disease that occurs in premature infants with incomplete retinal vascular development. It is also the most common cause of blindness in children.
3 Antibody Therapeutics: Antibody developed to selectively inhibit abnormal blood vessel growth and leakage. Typical antibody therapeutics is Avastin and Lucentis, which hinder vascular endothelial growth factor (VEGF).
4 Angiopoietin-1: A critical growth factor that induces the production of healthy blood vessels and maintains the stability of the created vessel.
5 Retinal ischemia: State of ailment where retinal tissue blood supply is not sufficient.
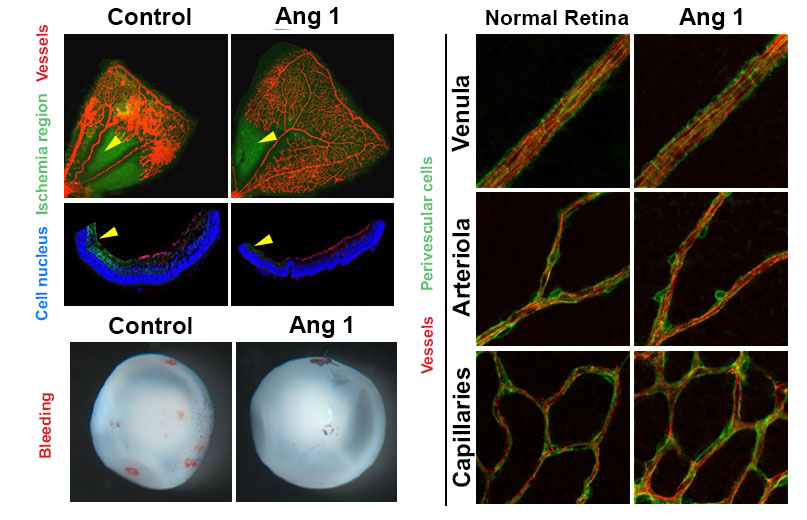
Figure 1. Retinopathy mouse models show that, in comparison to the control group, the VEGF-Trap treatment and Angiopoietin-1 (Ang1) treatment groups significantly suppresses the pathological vascular proliferation. In addition, the Ang 1 group show vessel growth toward the central avascular area (region of retinal ischemia), which is not observed in VEGF-Trap treatment.
Figure 2. Reduced retinal ischemia, retinal bleeding and blood vessel normalization by Angiopoietin-1. Retinal ischemic region (arrow) and retinal bleeding significantly reduced in the Angiopoietin-1 (Ang1) treatment model in comparison to control group (left). The newly generated vessels in Ang 1 model are structurally supported by perivascular cells as normal retinal vessels do (right).
2013.10.12 View 9011
Therapy developed to induce Angiogenesis of Retina
- Junyeop Lee, Graduate School of Medical Sciences and Engineering
- Research results expected to be applied for treatment of diabetic retinopathy
A major clue to treatment of retinovascular disease, which causes blindness, has been found. The key to protection of the retinal nerve is the angiogenic protein that promotes healthy retinal vessel growth around the retina, which usually does not receive blood supply readily. This research offers a beginning to the possible improvement of therapy for diabetic retinopathy1 and retinopathy of prematurity2. Also important to the research is the fact that the ophthalmology specialist researcher, currently undergoing professional training, provided the results.
KAIST Graduate School of Medical Sciences and Engineering’s Junyeop Lee is the opthalmology specialist, who carried out the research under supervision by academic advisers Gyuyeong Go and Wookjun Yoo. The Ministry of Science, ICT and Future Planning as well as the National Research Foundation of Korea have funded his research. The research results have been published as a cover paper on ‘Science Translational Medicine’ on 18th August. This journal is a sister publication of Science, which is prestigious in the field of translational medicine that ties the basic science with clinical medicine. (Thesis title: Angiopoietin-1 Guides Directional Angiogenesis Through Integrin αvβ5 Signaling for Recovery of Ischemic Retinopathy)
The traditional treatment of diabetic retinopathy includes laser photocoagulation to destroy the retinal tissues or antibody therapeutics, which prevents vessel proliferation and blood leaking. The advantage of antibody therapeutics3 is that it retains the retinal nerves, however, it is not the fundamental solution but merely a temporary one, which requires repeated treatments.
The research team identified that Angiopoietin-14 protein, known as essential for growth and stabilization of vessels, also plays an important role in retinal vessel growth. The protein protects the retinal nerves, as well as provides improvement for retinal ischemia5 that is the root cause of vision loss due to retinal hemorrhages. It is expected to become a key to finding fundamental treatment method – by providing sufficient blood supply to the retina, thereby preserving the retinal nerve functions.
The results show that administration of Angiopoietin-1 to retinopathy mouse model promotes growth of healthy vessel growth, further preventing abnormal vessel growth, retinal hemorrhage and vision loss due to retinal ischemia.
Junyeop Lee said, “This research has identified that Angiopoietin-1 is an important factor in retinal vessel generation and stabilization. The paradigm will shift from traditional treatment method, which prevents vessel growth, to a new method that generates healthy vessels and strengthens vessel functions.”
1 Diabetic retinopathy: This retinovascular disease is a diabetic complication caused by insufficient blood supply. It is the major causes of blindness in adults.
2 Retinopathy of prematurity: The retinal vascular disease that occurs in premature infants with incomplete retinal vascular development. It is also the most common cause of blindness in children.
3 Antibody Therapeutics: Antibody developed to selectively inhibit abnormal blood vessel growth and leakage. Typical antibody therapeutics is Avastin and Lucentis, which hinder vascular endothelial growth factor (VEGF).
4 Angiopoietin-1: A critical growth factor that induces the production of healthy blood vessels and maintains the stability of the created vessel.
5 Retinal ischemia: State of ailment where retinal tissue blood supply is not sufficient.
Figure 1. Retinopathy mouse models show that, in comparison to the control group, the VEGF-Trap treatment and Angiopoietin-1 (Ang1) treatment groups significantly suppresses the pathological vascular proliferation. In addition, the Ang 1 group show vessel growth toward the central avascular area (region of retinal ischemia), which is not observed in VEGF-Trap treatment.
Figure 2. Reduced retinal ischemia, retinal bleeding and blood vessel normalization by Angiopoietin-1. Retinal ischemic region (arrow) and retinal bleeding significantly reduced in the Angiopoietin-1 (Ang1) treatment model in comparison to control group (left). The newly generated vessels in Ang 1 model are structurally supported by perivascular cells as normal retinal vessels do (right).
2013.10.12 View 9011 -
 The new era of personalized cancer diagnosis and treatment
Professor Tae-Young Yoon
- Succeeded in observing carcinogenic protein at the molecular level
- “Paved the way to customized cancer treatment through accurate analysis of carcinogenic protein”
The joint KAIST research team of Professor Tae Young Yoon of the Department of Physics and Professor Won Do Huh of the Department of Biological Sciences have developed the technology to monitor characteristics of carcinogenic protein in cancer tissue – for the first time in the world.
The technology makes it possible to analyse the mechanism of cancer development through a small amount of carcinogenic protein from a cancer patient. Therefore, a personalised approach to diagnosis and treatment using the knowledge of the specific mechanism of cancer development in the patient may be possible in the future.
Until recently, modern medicine could only speculate on the cause of cancer through statistics. Although developed countries, such as the United States, are known to use a large sequencing technology that analyses the patient’s DNA, identification of the interactions between proteins responsible for causing cancer remained an unanswered question for a long time in medicine.
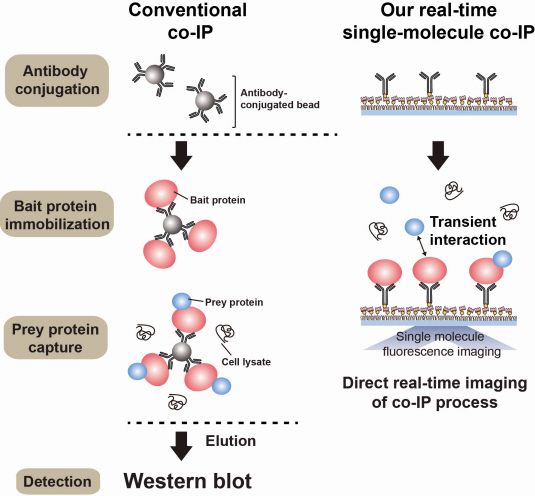
Firstly, Professor Yoon’s research team has developed a fluorescent microscope that can observe even a single molecule. Then, the “Immunoprecipitation method”, a technology to extract a specific protein exploiting the high affinity between antigens and antibodies was developed. Using this technology and the microscope, “Real-Time Single Molecule co-Immunoprecipitation Method” was created. In this way, the team succeeded in observing the interactions between carcinogenic and other proteins at a molecular level, in real time.
To validate the developed technology, the team investigated Ras, a carcinogenic protein; its mutation statistically is known to cause around 30% of cancers.
The experimental results confirmed that 30-50% of Ras protein was expressed in mouse tumour and human cancer cells. In normal cells, less than 5% of Ras protein was expressed. Thus, the experiment showed that unusual increase in activation of Ras protein induces cancer.
The increase in the ratio of active Ras protein can be inferred from existing research data but the measurement of specific numerical data has never been done before.
The team suggested a new molecular level diagnosis technique of identifying the progress of cancer in patients through measuring the percentage of activated carcinogenic protein in cancer tissue.
Professor Yoon Tae-young said, “This newly developed technology does not require a separate procedure of protein expression or refining, hence the existing proteins in real biological tissues or cancer cells can be observed directly.” He also said, “Since carcinogenic protein can be analyzed accurately, it has opened up the path to customized cancer treatment in the future.”
“Since the observation is possible on a molecular level, the technology confers the advantage that researchers can carry out various examinations on a small sample of the cancer patient.” He added, “The clinical trial will start in December 2012 and in a few years customized cancer diagnosis and treatment will be possible.”
Meanwhile, the research has been published in Nature Communications (February 19). Many researchers from various fields have participated, regardless of the differences in their speciality, and successfully produced interdisciplinary research. Professor Tae Young Yoon of the Department of Physics and Professors Dae Sik Lim and Won Do Huh of Biological Sciences at KAIST, and Professor Chang Bong Hyun of Computational Science of KIAS contributed to developing the technique.
Figure 1: Schematic diagram of observed interactions at the molecular level in real time using fluorescent microscope. The carcinogenic protein from a mouse tumour is fixed on the microchip, and its molecular characteristics are observed live.
Figure 2: Molecular interaction data using a molecular level fluorescent microscope. A signal in the form of spike is shown when two proteins combine. This is monitored live using an Electron Multiplying Charge Coupled Device (EMCCD). It shows signal results in bright dots.
An organism has an immune system as a defence mechanism to foreign intruders. The immune system is activated when unwanted pathogens or foreign protein are in the body. Antibodies form in recognition of the specific antigen to protect itself. Organisms evolved to form antibodies with high specificity to a certain antigen. Antibodies only react to its complementary antigens. The field of molecular biology uses the affinity between antigens and antibodies to extract specific proteins; a technology called immunoprecipitation. Even in a mixture of many proteins, the protein sought can be extracted using antibodies. Thus immunoprecipitation is widely used to detect pathogens or to extract specific proteins.
Technology co-IP is a well-known example that uses immunoprecipitation. The research on interactions between proteins uses co-IP in general. The basis of fixing the antigen on the antibody to extract antigen protein is the same as immunoprecipitation. Then, researchers inject and observe its reaction with the partner protein to observe the interactions and precipitate the antibodies. If the reaction occurs, the partner protein will be found with the antibodies in the precipitations. If not, then the partner protein will not be found. This shows that the two proteins interact.
However, the traditional co-IP can be used to infer the interactions between the two proteins although the information of the dynamics on how the reaction occurs is lost. To overcome these shortcomings, the Real-Time Single Molecule co-IP Method enables observation on individual protein level in real time. Therefore, the significance of the new technique is in making observation of interactions more direct and quantitative.
Additional Figure 1: Comparison between Conventional co-IP and Real-Time Single Molecule co-IP
2013.04.01 View 14835
The new era of personalized cancer diagnosis and treatment
Professor Tae-Young Yoon
- Succeeded in observing carcinogenic protein at the molecular level
- “Paved the way to customized cancer treatment through accurate analysis of carcinogenic protein”
The joint KAIST research team of Professor Tae Young Yoon of the Department of Physics and Professor Won Do Huh of the Department of Biological Sciences have developed the technology to monitor characteristics of carcinogenic protein in cancer tissue – for the first time in the world.
The technology makes it possible to analyse the mechanism of cancer development through a small amount of carcinogenic protein from a cancer patient. Therefore, a personalised approach to diagnosis and treatment using the knowledge of the specific mechanism of cancer development in the patient may be possible in the future.
Until recently, modern medicine could only speculate on the cause of cancer through statistics. Although developed countries, such as the United States, are known to use a large sequencing technology that analyses the patient’s DNA, identification of the interactions between proteins responsible for causing cancer remained an unanswered question for a long time in medicine.
Firstly, Professor Yoon’s research team has developed a fluorescent microscope that can observe even a single molecule. Then, the “Immunoprecipitation method”, a technology to extract a specific protein exploiting the high affinity between antigens and antibodies was developed. Using this technology and the microscope, “Real-Time Single Molecule co-Immunoprecipitation Method” was created. In this way, the team succeeded in observing the interactions between carcinogenic and other proteins at a molecular level, in real time.
To validate the developed technology, the team investigated Ras, a carcinogenic protein; its mutation statistically is known to cause around 30% of cancers.
The experimental results confirmed that 30-50% of Ras protein was expressed in mouse tumour and human cancer cells. In normal cells, less than 5% of Ras protein was expressed. Thus, the experiment showed that unusual increase in activation of Ras protein induces cancer.
The increase in the ratio of active Ras protein can be inferred from existing research data but the measurement of specific numerical data has never been done before.
The team suggested a new molecular level diagnosis technique of identifying the progress of cancer in patients through measuring the percentage of activated carcinogenic protein in cancer tissue.
Professor Yoon Tae-young said, “This newly developed technology does not require a separate procedure of protein expression or refining, hence the existing proteins in real biological tissues or cancer cells can be observed directly.” He also said, “Since carcinogenic protein can be analyzed accurately, it has opened up the path to customized cancer treatment in the future.”
“Since the observation is possible on a molecular level, the technology confers the advantage that researchers can carry out various examinations on a small sample of the cancer patient.” He added, “The clinical trial will start in December 2012 and in a few years customized cancer diagnosis and treatment will be possible.”
Meanwhile, the research has been published in Nature Communications (February 19). Many researchers from various fields have participated, regardless of the differences in their speciality, and successfully produced interdisciplinary research. Professor Tae Young Yoon of the Department of Physics and Professors Dae Sik Lim and Won Do Huh of Biological Sciences at KAIST, and Professor Chang Bong Hyun of Computational Science of KIAS contributed to developing the technique.
Figure 1: Schematic diagram of observed interactions at the molecular level in real time using fluorescent microscope. The carcinogenic protein from a mouse tumour is fixed on the microchip, and its molecular characteristics are observed live.
Figure 2: Molecular interaction data using a molecular level fluorescent microscope. A signal in the form of spike is shown when two proteins combine. This is monitored live using an Electron Multiplying Charge Coupled Device (EMCCD). It shows signal results in bright dots.
An organism has an immune system as a defence mechanism to foreign intruders. The immune system is activated when unwanted pathogens or foreign protein are in the body. Antibodies form in recognition of the specific antigen to protect itself. Organisms evolved to form antibodies with high specificity to a certain antigen. Antibodies only react to its complementary antigens. The field of molecular biology uses the affinity between antigens and antibodies to extract specific proteins; a technology called immunoprecipitation. Even in a mixture of many proteins, the protein sought can be extracted using antibodies. Thus immunoprecipitation is widely used to detect pathogens or to extract specific proteins.
Technology co-IP is a well-known example that uses immunoprecipitation. The research on interactions between proteins uses co-IP in general. The basis of fixing the antigen on the antibody to extract antigen protein is the same as immunoprecipitation. Then, researchers inject and observe its reaction with the partner protein to observe the interactions and precipitate the antibodies. If the reaction occurs, the partner protein will be found with the antibodies in the precipitations. If not, then the partner protein will not be found. This shows that the two proteins interact.
However, the traditional co-IP can be used to infer the interactions between the two proteins although the information of the dynamics on how the reaction occurs is lost. To overcome these shortcomings, the Real-Time Single Molecule co-IP Method enables observation on individual protein level in real time. Therefore, the significance of the new technique is in making observation of interactions more direct and quantitative.
Additional Figure 1: Comparison between Conventional co-IP and Real-Time Single Molecule co-IP
2013.04.01 View 14835 -
 Ligand Recognition Mechanism of Protein Identified
Professor Hak-Sung Kim
-“Solved the 50 year old mystery of how protein recognises and binds to ligands”
- Exciting potential for understanding life phenomena and the further development of highly effective therapeutic agent development
KAIST’s Biological Science Department’s Professor Hak-Sung Kim, working in collaboration with Professor Sung-Chul Hong of Department of Physics, Seoul National University, has identified the mechanism of how the protein recognizes and binds to ligands within the human body.
The research findings were published in the online edition of Nature Chemical Biology (March 18), which is the most prestigious journal in the field of life science.
Since the research identified the mechanism, of which protein recognises and binds to ligands, it will take an essential role in understanding complex life phenomenon by understanding regulatory function of protein.
Also, ligand recognition of proteins is closely related to the cause of various diseases. Therefore the research team hopes to contribute to the development of highly effective treatments.
Ligands, well-known examples include nucleic acid and proteins, form the structure of an organism or are essential constituents with special functions such as information signalling.
In particular, the most important role of protein is recognising and binding to a particular ligand and hence regulating and maintaining life phenomena. The abnormal occurrence of an error in recognition of ligands may lead to various diseases.
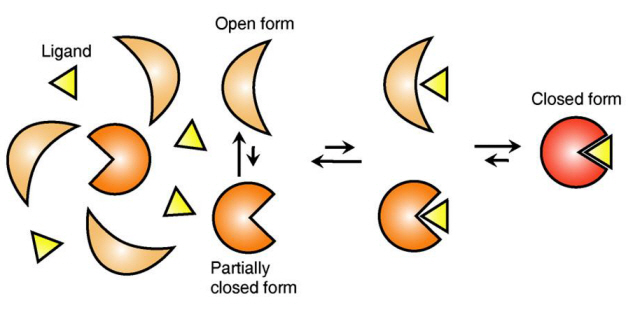
The research team focused on the repetition of change in protein structure from the most stable “open form” to a relatively unstable “partially closed form”.
Professor Kim’s team analysed the change in protein structure when binding to a ligand on a molecular level in real time to explain the ligand recognition mechanism.
The research findings showed that ligands prefer the most stable protein structure. The team was the first in the world to identify that ligands alter protein structure to the most stable, the lowest energy level, when it binds to the protein.
In addition, the team found that ligands bind to unstable partially-closed forms to change protein structure.
The existing models to explain ligand recognition mechanism of protein are “Induced Custom Model”, which involves change in protein structure in binding to ligands, and the “Structure Selection Model”, which argues that ligands select and recognise only the best protein structure out of many. The academic world considers that the team’s research findings have perfectly proved the models through experiments for the first time in the world.
Professor Kim explained, “In the presence of ligands, there exists a phenomenon where the speed of altering protein structure is changed. This phenomenon is analysed on a molecular level to prove ligand recognition mechanism of protein for the first time”. He also said, “The 50-year old mystery, that existed only as a hypothesis on biology textbooks and was thought never to be solved, has been confirmed through experiments for the first time.”
Figure 1: Proteins, with open and partially open form, recognising and binding to ligands.
Figure 2: Ligands temporarily bind to a stable protein structure, open form, which changes into the most stable structure, closed form. In addition, binding to partially closed form also changes protein structure to closed form.
2013.04.01 View 8875
Ligand Recognition Mechanism of Protein Identified
Professor Hak-Sung Kim
-“Solved the 50 year old mystery of how protein recognises and binds to ligands”
- Exciting potential for understanding life phenomena and the further development of highly effective therapeutic agent development
KAIST’s Biological Science Department’s Professor Hak-Sung Kim, working in collaboration with Professor Sung-Chul Hong of Department of Physics, Seoul National University, has identified the mechanism of how the protein recognizes and binds to ligands within the human body.
The research findings were published in the online edition of Nature Chemical Biology (March 18), which is the most prestigious journal in the field of life science.
Since the research identified the mechanism, of which protein recognises and binds to ligands, it will take an essential role in understanding complex life phenomenon by understanding regulatory function of protein.
Also, ligand recognition of proteins is closely related to the cause of various diseases. Therefore the research team hopes to contribute to the development of highly effective treatments.
Ligands, well-known examples include nucleic acid and proteins, form the structure of an organism or are essential constituents with special functions such as information signalling.
In particular, the most important role of protein is recognising and binding to a particular ligand and hence regulating and maintaining life phenomena. The abnormal occurrence of an error in recognition of ligands may lead to various diseases.
The research team focused on the repetition of change in protein structure from the most stable “open form” to a relatively unstable “partially closed form”.
Professor Kim’s team analysed the change in protein structure when binding to a ligand on a molecular level in real time to explain the ligand recognition mechanism.
The research findings showed that ligands prefer the most stable protein structure. The team was the first in the world to identify that ligands alter protein structure to the most stable, the lowest energy level, when it binds to the protein.
In addition, the team found that ligands bind to unstable partially-closed forms to change protein structure.
The existing models to explain ligand recognition mechanism of protein are “Induced Custom Model”, which involves change in protein structure in binding to ligands, and the “Structure Selection Model”, which argues that ligands select and recognise only the best protein structure out of many. The academic world considers that the team’s research findings have perfectly proved the models through experiments for the first time in the world.
Professor Kim explained, “In the presence of ligands, there exists a phenomenon where the speed of altering protein structure is changed. This phenomenon is analysed on a molecular level to prove ligand recognition mechanism of protein for the first time”. He also said, “The 50-year old mystery, that existed only as a hypothesis on biology textbooks and was thought never to be solved, has been confirmed through experiments for the first time.”
Figure 1: Proteins, with open and partially open form, recognising and binding to ligands.
Figure 2: Ligands temporarily bind to a stable protein structure, open form, which changes into the most stable structure, closed form. In addition, binding to partially closed form also changes protein structure to closed form.
2013.04.01 View 8875 -
 Genetic Cause of ADHD (Attention Deficit Hyperactivity Disorder) Found
The cooperative research team consisting research teams under Professor Kim Eun Joon and Professor Kang Chang Won of the department of Biological Sciences discovered that ADHD arises from the deficiency of GIT1 protein in the brain’s neural synapses.
ADHD (Attention Deficit Hyperactivity Disorder) is found in around 5% of children around the world and is a disorder where the child becomes unable to concentrate, show over the top responses, and display impulsive behavior.
The research team found that the difference between children with ADHD and those without it is one base in the GIT1 gene. The difference of a single base causes the underproduction of this protein, and those children with low levels of the protein had a higher probability to develop ADHD.
In addition, further evidence was provided when the research team conducted mice experiments. Those mice with low levels of GIT1 exhibited impulsive and exaggerated reactions like humans with ADHD, had learning disabilities, and produced abnormal brain waves. And upon injecting these mice with cure for ADHD, the symptoms of ADHD disappeared.
The impulsive behavior of ADHD children disappears as the child enters adulthood and a similar pattern was found in mice. A mice with low levels of GIT1 showed impulsive behaviors when 2 months old, but these behaviors disappeared as it got older to around 7 months old (equivalent to 20~30 years old for humans).
Professor Kim Eun Joon commented that there has to be equilibrium between mechanisms that excite the neurons and mechanisms that calm the neurons, but the lack of GIT1 leads to the decrease in the mechanisms that calm the neurons which causes the impulsive behavior of ADHD patients.
In addition, Professor Kang Chang Won commented that the results of the experiment has been receiving rave reviews and is being seen as the new method in the production of the cure for ADHD.
The result of the experiment was published in the online edition of Nature Medicine magazine.
2011.04.30 View 8661
Genetic Cause of ADHD (Attention Deficit Hyperactivity Disorder) Found
The cooperative research team consisting research teams under Professor Kim Eun Joon and Professor Kang Chang Won of the department of Biological Sciences discovered that ADHD arises from the deficiency of GIT1 protein in the brain’s neural synapses.
ADHD (Attention Deficit Hyperactivity Disorder) is found in around 5% of children around the world and is a disorder where the child becomes unable to concentrate, show over the top responses, and display impulsive behavior.
The research team found that the difference between children with ADHD and those without it is one base in the GIT1 gene. The difference of a single base causes the underproduction of this protein, and those children with low levels of the protein had a higher probability to develop ADHD.
In addition, further evidence was provided when the research team conducted mice experiments. Those mice with low levels of GIT1 exhibited impulsive and exaggerated reactions like humans with ADHD, had learning disabilities, and produced abnormal brain waves. And upon injecting these mice with cure for ADHD, the symptoms of ADHD disappeared.
The impulsive behavior of ADHD children disappears as the child enters adulthood and a similar pattern was found in mice. A mice with low levels of GIT1 showed impulsive behaviors when 2 months old, but these behaviors disappeared as it got older to around 7 months old (equivalent to 20~30 years old for humans).
Professor Kim Eun Joon commented that there has to be equilibrium between mechanisms that excite the neurons and mechanisms that calm the neurons, but the lack of GIT1 leads to the decrease in the mechanisms that calm the neurons which causes the impulsive behavior of ADHD patients.
In addition, Professor Kang Chang Won commented that the results of the experiment has been receiving rave reviews and is being seen as the new method in the production of the cure for ADHD.
The result of the experiment was published in the online edition of Nature Medicine magazine.
2011.04.30 View 8661 -
 Professor Ko Kyu Young Appointed as a Distinguished Professor at KAIST
Professor Ko Kyu Young of the Graduate School of Medical Sciences was appointed as the Distinguished Professor at KAIST.
Professor Ko is famous internationally for his work on the catalyst for blood vessel growth COMP-ANG1, and also for his research on blood vessel growth and lymph duct growth control.
Professor Ko developed the Double Anti-Angiogenic Protein (DAAP) which effectively restricts the blood vessels from growing, opening a new approach to curing caner. The paper was published in ‘Cancer Cell’ as the cover paper (2010 August 17th edition) and is widely recognized as the marker that sums up the new paradigm of cure for cancer.
In addition, his work on explaining how the new antigen interacts with the T-lymphocyte during a vaccination lead to the possibility of the increase of the efficiency of vaccination. The result of the research was published as the cover paper in ‘Immunity’ magazine.
As is obvious to see his work with blood vessel growth and lymph duct growth and control is being published in major scientific journals. In addition he is continuously invited to international conferences as guest speakers and leader, effectively leading the field. As a result, he was appointed as the editor of ‘Blood’ magazine, the world’s best journal in the field of hematology and received ‘2010 KAISTian of the Year’ Award.
The title Distinguished Professor is appointed to those who have made world-class research results and educational results and actively lead their respective field. They are provided with extra incentives and can even continue on with the professorship after retirement.
It is only limited to 3% of the professors at KAIST and has to be someone recommended by the President, Vice-President, and the Deans of department and their worthiness is scrutinized by a foreign expert.
2011.03.25 View 9165
Professor Ko Kyu Young Appointed as a Distinguished Professor at KAIST
Professor Ko Kyu Young of the Graduate School of Medical Sciences was appointed as the Distinguished Professor at KAIST.
Professor Ko is famous internationally for his work on the catalyst for blood vessel growth COMP-ANG1, and also for his research on blood vessel growth and lymph duct growth control.
Professor Ko developed the Double Anti-Angiogenic Protein (DAAP) which effectively restricts the blood vessels from growing, opening a new approach to curing caner. The paper was published in ‘Cancer Cell’ as the cover paper (2010 August 17th edition) and is widely recognized as the marker that sums up the new paradigm of cure for cancer.
In addition, his work on explaining how the new antigen interacts with the T-lymphocyte during a vaccination lead to the possibility of the increase of the efficiency of vaccination. The result of the research was published as the cover paper in ‘Immunity’ magazine.
As is obvious to see his work with blood vessel growth and lymph duct growth and control is being published in major scientific journals. In addition he is continuously invited to international conferences as guest speakers and leader, effectively leading the field. As a result, he was appointed as the editor of ‘Blood’ magazine, the world’s best journal in the field of hematology and received ‘2010 KAISTian of the Year’ Award.
The title Distinguished Professor is appointed to those who have made world-class research results and educational results and actively lead their respective field. They are provided with extra incentives and can even continue on with the professorship after retirement.
It is only limited to 3% of the professors at KAIST and has to be someone recommended by the President, Vice-President, and the Deans of department and their worthiness is scrutinized by a foreign expert.
2011.03.25 View 9165 -
 New Bio-Clock gene and its function found
The Ministry of Education, Science and Technology announced that a Korean research team has found a new gene responsible for maintaining the bio-clock (twenty-four) and its mechanism.
Twnety-four was led by Professor Choi Joon Ho and Dr. Lee Jong Bin of KAIST (department of Biology) and was a joint operation with Professor Ravi Allada and Dr.Lim Jeong Hoon of Northwestern University (department of neurobiology) and the result was published in ‘Nature’ magazine.
The research team experimented with transformed small fruit flies for 4 years and found that there was an undiscovered gene that deals with the bio rhythm in the brain which they named ‘twenty-four’.
The understanding with genes prior to twenty-four was that these genes regulate biorhythm in the transcription phase (DNA to mRNA). Twenty-four operates in the step after transcription when the ribosome creates proteins. Especially twenty-four has a great effect on the ‘period protein’ which acts as a sub-atomic clock that regulates the rhythm and life of each cell.
The experiment was innovational in that it was able to scientifically prove the function of the protein produced by the gene.
The result is expected to help solve the problems associated with sleep disorders, jetlags, eating rhythms, bio rhythms, etc.
The name twenty-four was the fact that a day, a cycle, is 24 hours long and the gene’s serial numbers CG4857 adds up to twenty four.
2011.02.23 View 9505
New Bio-Clock gene and its function found
The Ministry of Education, Science and Technology announced that a Korean research team has found a new gene responsible for maintaining the bio-clock (twenty-four) and its mechanism.
Twnety-four was led by Professor Choi Joon Ho and Dr. Lee Jong Bin of KAIST (department of Biology) and was a joint operation with Professor Ravi Allada and Dr.Lim Jeong Hoon of Northwestern University (department of neurobiology) and the result was published in ‘Nature’ magazine.
The research team experimented with transformed small fruit flies for 4 years and found that there was an undiscovered gene that deals with the bio rhythm in the brain which they named ‘twenty-four’.
The understanding with genes prior to twenty-four was that these genes regulate biorhythm in the transcription phase (DNA to mRNA). Twenty-four operates in the step after transcription when the ribosome creates proteins. Especially twenty-four has a great effect on the ‘period protein’ which acts as a sub-atomic clock that regulates the rhythm and life of each cell.
The experiment was innovational in that it was able to scientifically prove the function of the protein produced by the gene.
The result is expected to help solve the problems associated with sleep disorders, jetlags, eating rhythms, bio rhythms, etc.
The name twenty-four was the fact that a day, a cycle, is 24 hours long and the gene’s serial numbers CG4857 adds up to twenty four.
2011.02.23 View 9505 -
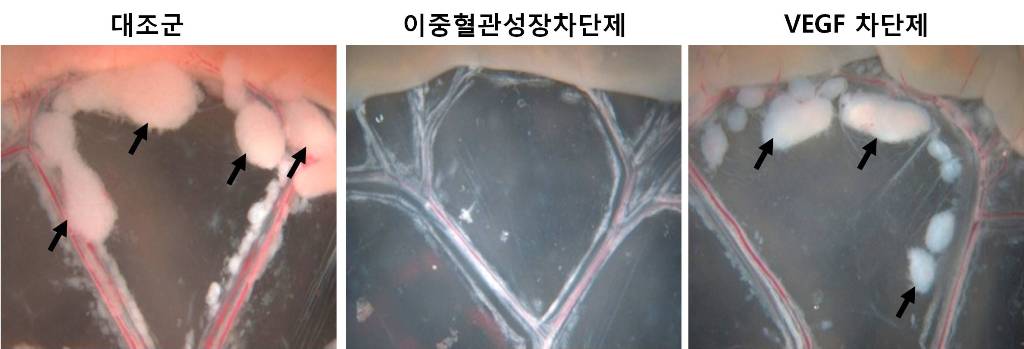 An internationally renowned academic journal published the research result produced by a KAST research team on its cover.
Fc DAAP VEGF-Trap
Photograph showing the gross features of tumor growth along the mesentery-intestinal border. T: tumor. Scale bars represent 5 mm.
Professor Gou-Young Koh of the Biological Sciences Department, KAIST, and his research team published their research result in Cancer Cell, a peer-review scientific journal, as a cover article dated August 17, 2010. It is the first time for the journal to pick up a paper written by a Korean research team and publish it as the cover.
It has been known that a vascular growth factor (VEGF) is closely related to the growth of a tumor. The research team recently discovered that in addition to VEGF, another growth factor, angiopoietin-2 (Ang2), is also engaged with the increase of tumors.
Professor Koh said, “VEGF and the angiopoietins play critical roles in tumor progression and metastasis, and a single inhibitor targeting both factors have not been available.”
The team led by Professor Koh has developed a double anti-angiogenic protein (DAAP) that can simultaneously bind VEGF-A and the angiopoietins and block their actions.
Professor Koh said in his paper, “DAAP is a highly effective molecule for regressing tumor angiogenesis and metastasis in implanted and spontaneous solid tumor; it can also effectively reduce ascites formation and vascular leakage in an ovarian carcinoma model. Thus, simultaneous blockade of VEGF-A and angiopoietins with DAAP is an effective therapeutic strategy for blocking tumor angiogenesis, metastasis, and vascular leakage.”
So far, cancer patients have received Avastin, anticancer drug, to inhibit VEGF, but the drug has not successfully restrained the growth of cancer tumors and brought to some of the patients with serious side effects instead.
Professor Koh said, “DAAP will be very effective to control the expansion of tumor growth factors, which will open up a new possibility for the development of more helpful cancer medicine with low side effects.”
2010.08.20 View 9933
An internationally renowned academic journal published the research result produced by a KAST research team on its cover.
Fc DAAP VEGF-Trap
Photograph showing the gross features of tumor growth along the mesentery-intestinal border. T: tumor. Scale bars represent 5 mm.
Professor Gou-Young Koh of the Biological Sciences Department, KAIST, and his research team published their research result in Cancer Cell, a peer-review scientific journal, as a cover article dated August 17, 2010. It is the first time for the journal to pick up a paper written by a Korean research team and publish it as the cover.
It has been known that a vascular growth factor (VEGF) is closely related to the growth of a tumor. The research team recently discovered that in addition to VEGF, another growth factor, angiopoietin-2 (Ang2), is also engaged with the increase of tumors.
Professor Koh said, “VEGF and the angiopoietins play critical roles in tumor progression and metastasis, and a single inhibitor targeting both factors have not been available.”
The team led by Professor Koh has developed a double anti-angiogenic protein (DAAP) that can simultaneously bind VEGF-A and the angiopoietins and block their actions.
Professor Koh said in his paper, “DAAP is a highly effective molecule for regressing tumor angiogenesis and metastasis in implanted and spontaneous solid tumor; it can also effectively reduce ascites formation and vascular leakage in an ovarian carcinoma model. Thus, simultaneous blockade of VEGF-A and angiopoietins with DAAP is an effective therapeutic strategy for blocking tumor angiogenesis, metastasis, and vascular leakage.”
So far, cancer patients have received Avastin, anticancer drug, to inhibit VEGF, but the drug has not successfully restrained the growth of cancer tumors and brought to some of the patients with serious side effects instead.
Professor Koh said, “DAAP will be very effective to control the expansion of tumor growth factors, which will open up a new possibility for the development of more helpful cancer medicine with low side effects.”
2010.08.20 View 9933 -
 Native-like Spider Silk Produced in Metabolically Engineered Bacterium
Microscopic picture of 285 kilodalton recombinant spider silk fiber
Researchers have long envied spiders’ ability to manufacture silk that is light-weighted while as strong and tough as steel or Kevlar. Indeed, finer than human hair, five times stronger by weight than steel, and three times tougher than the top quality man-made fiber Kevlar, spider dragline silk is an ideal material for numerous applications. Suggested industrial applications have ranged from parachute cords and protective clothing to composite materials in aircrafts. Also, many biomedical applications are envisioned due to its biocompatibility and biodegradability.
Unfortunately, natural dragline silk cannot be conveniently obtained by farming spiders because they are highly territorial and aggressive. To develop a more sustainable process, can scientists mass-produce artificial silk while maintaining the amazing properties of native silk? That is something Sang Yup Lee at the Korea Advanced Institute of Science and Technology (KAIST) in Daejeon, the Republic of Korea, and his collaborators, Professor Young Hwan Park at Seoul National University and Professor David Kaplan at Tufts University, wanted to figure out. Their method is very similar to what spiders essentially do: first, expression of recombinant silk proteins; second, making the soluble silk proteins into water-insoluble fibers through spinning.
For the successful expression of high molecular weight spider silk protein, Professor Lee and his colleagues pieced together the silk gene from chemically synthesized oligonucleotides, and then inserted it into the expression host (in this case, an industrially safe bacterium Escherichia coli which is normally found in our gut). Initially, the bacterium refused to the challenging task of producing high molecular weight spider silk protein due to the unique characteristics of the protein, such as extremely large size, repetitive nature of the protein structure, and biased abundance of a particular amino acid glycine. “To make E. coli synthesize this ultra high molecular weight (as big as 285 kilodalton) spider silk protein having highly repetitive amino acid sequence, we helped E. coli overcome the difficulties by systems metabolic engineering,” says Sang Yup Lee, Distinguished Professor of KAIST, who led this project. His team boosted the pool of glycyl-tRNA, the major building block of spider silk protein synthesis. “We could obtain appreciable expression of the 285 kilodalton spider silk protein, which is the largest recombinant silk protein ever produced in E. coli. That was really incredible.” says Dr. Xia.
But this was only step one. The KAIST team performed high-cell-density cultures for mass production of the recombinant spider silk protein. Then, the team developed a simple, easy to scale-up purification process for the recombinant spider silk protein. The purified spider silk protein could be spun into beautiful silk fiber. To study the mechanical properties of the artificial spider silk, the researchers determined tenacity, elongation, and Young’s modulus, the three critical mechanical parameters that represent a fiber’s strength, extensibility, and stiffness. Importantly, the artificial fiber displayed the tenacity, elongation, and Young’s modulus of 508 MPa, 15%, and 21 GPa, respectively, which are comparable to those of the native spider silk.
“We have offered an overall platform for mass production of native-like spider dragline silk. This platform would enable us to have broader industrial and biomedical applications for spider silk. Moreover, many other silk-like biomaterials such as elastin, collagen, byssus, resilin, and other repetitive proteins have similar features to spider silk protein. Thus, our platform should also be useful for their efficient bio-based production and applications,” concludes Professor Lee.
This work is published on July 26 in the Proceedings of the National Academy of Sciences (PNAS) online.
2010.07.28 View 14735
Native-like Spider Silk Produced in Metabolically Engineered Bacterium
Microscopic picture of 285 kilodalton recombinant spider silk fiber
Researchers have long envied spiders’ ability to manufacture silk that is light-weighted while as strong and tough as steel or Kevlar. Indeed, finer than human hair, five times stronger by weight than steel, and three times tougher than the top quality man-made fiber Kevlar, spider dragline silk is an ideal material for numerous applications. Suggested industrial applications have ranged from parachute cords and protective clothing to composite materials in aircrafts. Also, many biomedical applications are envisioned due to its biocompatibility and biodegradability.
Unfortunately, natural dragline silk cannot be conveniently obtained by farming spiders because they are highly territorial and aggressive. To develop a more sustainable process, can scientists mass-produce artificial silk while maintaining the amazing properties of native silk? That is something Sang Yup Lee at the Korea Advanced Institute of Science and Technology (KAIST) in Daejeon, the Republic of Korea, and his collaborators, Professor Young Hwan Park at Seoul National University and Professor David Kaplan at Tufts University, wanted to figure out. Their method is very similar to what spiders essentially do: first, expression of recombinant silk proteins; second, making the soluble silk proteins into water-insoluble fibers through spinning.
For the successful expression of high molecular weight spider silk protein, Professor Lee and his colleagues pieced together the silk gene from chemically synthesized oligonucleotides, and then inserted it into the expression host (in this case, an industrially safe bacterium Escherichia coli which is normally found in our gut). Initially, the bacterium refused to the challenging task of producing high molecular weight spider silk protein due to the unique characteristics of the protein, such as extremely large size, repetitive nature of the protein structure, and biased abundance of a particular amino acid glycine. “To make E. coli synthesize this ultra high molecular weight (as big as 285 kilodalton) spider silk protein having highly repetitive amino acid sequence, we helped E. coli overcome the difficulties by systems metabolic engineering,” says Sang Yup Lee, Distinguished Professor of KAIST, who led this project. His team boosted the pool of glycyl-tRNA, the major building block of spider silk protein synthesis. “We could obtain appreciable expression of the 285 kilodalton spider silk protein, which is the largest recombinant silk protein ever produced in E. coli. That was really incredible.” says Dr. Xia.
But this was only step one. The KAIST team performed high-cell-density cultures for mass production of the recombinant spider silk protein. Then, the team developed a simple, easy to scale-up purification process for the recombinant spider silk protein. The purified spider silk protein could be spun into beautiful silk fiber. To study the mechanical properties of the artificial spider silk, the researchers determined tenacity, elongation, and Young’s modulus, the three critical mechanical parameters that represent a fiber’s strength, extensibility, and stiffness. Importantly, the artificial fiber displayed the tenacity, elongation, and Young’s modulus of 508 MPa, 15%, and 21 GPa, respectively, which are comparable to those of the native spider silk.
“We have offered an overall platform for mass production of native-like spider dragline silk. This platform would enable us to have broader industrial and biomedical applications for spider silk. Moreover, many other silk-like biomaterials such as elastin, collagen, byssus, resilin, and other repetitive proteins have similar features to spider silk protein. Thus, our platform should also be useful for their efficient bio-based production and applications,” concludes Professor Lee.
This work is published on July 26 in the Proceedings of the National Academy of Sciences (PNAS) online.
2010.07.28 View 14735 -
 The thermal fluctuation and elasticity of cell membranes, lipid vesicles, interacting with pore-forming peptides were reported by a research team at KAIST.
A research team from KAIST, consisted of Sung-Min Choi, Professor of Nuclear and Quantum Engineering Department, and Ji-Hwan Lee, a doctoral student in the Department, published a paper on the “thermal fluctuation and elasticity of lipid vesicles interacting with pore-forming peptides.” The paper was carried by Physical Review Letters, an internationally renowned peer-review journal on physics on July 16, 2010.
Cell membranes, which consist of lipid bilayers, play important roles in cells as barriers to maintain concentrations and matrices to host membrane proteins. During cellular processes such as cell fission and fusion, the cell membranes undergo various morphological changes governed by the interplay between protein and lipid membranes. There have been many theoretical and experimental approaches to understand cellular processes driven by protein-lipid membrane interactions. However, it is not fully established how the membrane elastic properties, which play an important role in membrane deformation, are affected by the protein-membrane interactions.
Antimicrobial peptides are one of the most common examples of proteins that modify membrane morphology. While the pore-forming mechanisms of antimicrobial peptides in lipid bilayers have been widely investigated, there have been only a few attempts to understand the mechanisms in terms of membrane elastic properties. In particular, the effects of pore formation on the membrane fluctuation and elastic properties, which provide key information to understand the mechanism of antimicrobial peptide activity, have not been reported yet. The research team reports the thermal fluctuation and elasticity of lipid vesicles interacting with pore-forming peptides, which were measured by neutron spin-echo spectroscopy.
The results of this study are expected to pay an important role in understanding the elastic behavior and morphological changes of cell membranes induced by protein-membrane interactions, and may provide new insights for developing new theoretical models for membrane fluctuations which include the membrane mediated interaction between protein patches.
(a) (b)
Figure
(a) Schematics for bound melittin and pores in lipid bilayers
(b) P NMR signal ratio (with/without Mn2+) of DOPC LUV-melittin vs P/L at 30˚C. The dashed line is a guide for eyes.
2010.07.23 View 9666
The thermal fluctuation and elasticity of cell membranes, lipid vesicles, interacting with pore-forming peptides were reported by a research team at KAIST.
A research team from KAIST, consisted of Sung-Min Choi, Professor of Nuclear and Quantum Engineering Department, and Ji-Hwan Lee, a doctoral student in the Department, published a paper on the “thermal fluctuation and elasticity of lipid vesicles interacting with pore-forming peptides.” The paper was carried by Physical Review Letters, an internationally renowned peer-review journal on physics on July 16, 2010.
Cell membranes, which consist of lipid bilayers, play important roles in cells as barriers to maintain concentrations and matrices to host membrane proteins. During cellular processes such as cell fission and fusion, the cell membranes undergo various morphological changes governed by the interplay between protein and lipid membranes. There have been many theoretical and experimental approaches to understand cellular processes driven by protein-lipid membrane interactions. However, it is not fully established how the membrane elastic properties, which play an important role in membrane deformation, are affected by the protein-membrane interactions.
Antimicrobial peptides are one of the most common examples of proteins that modify membrane morphology. While the pore-forming mechanisms of antimicrobial peptides in lipid bilayers have been widely investigated, there have been only a few attempts to understand the mechanisms in terms of membrane elastic properties. In particular, the effects of pore formation on the membrane fluctuation and elastic properties, which provide key information to understand the mechanism of antimicrobial peptide activity, have not been reported yet. The research team reports the thermal fluctuation and elasticity of lipid vesicles interacting with pore-forming peptides, which were measured by neutron spin-echo spectroscopy.
The results of this study are expected to pay an important role in understanding the elastic behavior and morphological changes of cell membranes induced by protein-membrane interactions, and may provide new insights for developing new theoretical models for membrane fluctuations which include the membrane mediated interaction between protein patches.
(a) (b)
Figure
(a) Schematics for bound melittin and pores in lipid bilayers
(b) P NMR signal ratio (with/without Mn2+) of DOPC LUV-melittin vs P/L at 30˚C. The dashed line is a guide for eyes.
2010.07.23 View 9666 -
 KAIST Professor Exposes Structural Dynamics of Protein in Solution
-- Dr. Hyot-Cherl Ihee"s 3-Year Research Is Valuable in Pharmaceutical Application
Prof. Hyot-Cherl Ihee and his team at the Department of Chemistry, KAIST, has successfully unveiled the structural dynamics of protein in solution as a result of more than three years" research work.
Nature Methods, a sister publication of the authoritative science magazine Nature, published the treatise, titled "Tracking the structural dynamics of proteins in solution using time-resolved wide-angle X-ray scattering" in its Sept. 22 online edition. The research paper will be carried in the magazine"s printed version in its October edition, according to Dr. Lee who is its correspondence author.
In May 2005, Prof. Ihee successfully photographed the structural dynamics of protein in solid state and his findings were published in the Proceedings of National Academy of Science of the United States. As protein normally exists in human body in solution, not in solid state, he directed his research to developing the technology to capture protein"s dynamics in resolved state.
In July that year, Prof. Ihee succeeded in measuring the structural changes of simple organic molecules in real time. He further developed the technology to uncover the structural dynamics of hemoglobin, myoglobin and cytochrome C.
Prof. Ihee"s research, helped with the Education-Science-Technology Ministry"s Creative Research Promotion Fund, can be applied to new pharmaceutical development projects as well as nanotechnology development, according to KAIST officials.
Prof. Ihee who earned his doctorate at California Institute of Technology in 1994 began teaching at KAIST in 2003. He won the Young Scientist Award given by the Korean government in 2006.
2008.09.22 View 11071
KAIST Professor Exposes Structural Dynamics of Protein in Solution
-- Dr. Hyot-Cherl Ihee"s 3-Year Research Is Valuable in Pharmaceutical Application
Prof. Hyot-Cherl Ihee and his team at the Department of Chemistry, KAIST, has successfully unveiled the structural dynamics of protein in solution as a result of more than three years" research work.
Nature Methods, a sister publication of the authoritative science magazine Nature, published the treatise, titled "Tracking the structural dynamics of proteins in solution using time-resolved wide-angle X-ray scattering" in its Sept. 22 online edition. The research paper will be carried in the magazine"s printed version in its October edition, according to Dr. Lee who is its correspondence author.
In May 2005, Prof. Ihee successfully photographed the structural dynamics of protein in solid state and his findings were published in the Proceedings of National Academy of Science of the United States. As protein normally exists in human body in solution, not in solid state, he directed his research to developing the technology to capture protein"s dynamics in resolved state.
In July that year, Prof. Ihee succeeded in measuring the structural changes of simple organic molecules in real time. He further developed the technology to uncover the structural dynamics of hemoglobin, myoglobin and cytochrome C.
Prof. Ihee"s research, helped with the Education-Science-Technology Ministry"s Creative Research Promotion Fund, can be applied to new pharmaceutical development projects as well as nanotechnology development, according to KAIST officials.
Prof. Ihee who earned his doctorate at California Institute of Technology in 1994 began teaching at KAIST in 2003. He won the Young Scientist Award given by the Korean government in 2006.
2008.09.22 View 11071 -
 Prof. Chung Named Winner of 2008 KAIST Scientific Award
Professor Chung Jong-Kyeong of the Department of Biological Sciences was named the winner of the 2008 KAIST Scientific Award.
The prize was awarded by KAIST President Suh Nam-Pyo during the 37th KAIST anniversary ceremony on Feb. 16.
Chung was cited for disclosing the new anti-cancer aspect of adenosine monophosphate-activated protein kinase (AMPK). His papers, published in the science magazine Nature in 2006 and again in 2007, revealed that the protein could be used to treat certain forms of cancer, as well as prevent malignant growths.
2008.02.28 View 10844
Prof. Chung Named Winner of 2008 KAIST Scientific Award
Professor Chung Jong-Kyeong of the Department of Biological Sciences was named the winner of the 2008 KAIST Scientific Award.
The prize was awarded by KAIST President Suh Nam-Pyo during the 37th KAIST anniversary ceremony on Feb. 16.
Chung was cited for disclosing the new anti-cancer aspect of adenosine monophosphate-activated protein kinase (AMPK). His papers, published in the science magazine Nature in 2006 and again in 2007, revealed that the protein could be used to treat certain forms of cancer, as well as prevent malignant growths.
2008.02.28 View 10844