bio
-
 KAIST Vaccine for Tick-Borne Disease ‘SFTS’ Protects Against Lethal Infection
A KAIST research team reported the development of a DNA vaccine for Severe Fever with Thrombocytopenia Syndrome Virus (SFTSV) which completely protects against lethal infection in ferrets. The team confirmed that ferrets immunized with DNA vaccines encoding all SFTSV proteins showed 100% survival rate without detectable viremia and did not develop any clinical symptoms. This study was published in Nature Communications on August 23.
Severe Fever with Thrombocytopenia Syndrome (SFTS) is a newly emerging tick-borne infectious disease. The disease causes fever, severe thrombocytopenia, leukocytopenia as well as vomiting and diarrhea. Severe cases end up with organ system failure often accompanied by hemorrhages, and its mortality rate stands at 10–20%.
The viral disease has been endemic to East Asia but the spread of the tick vector to North America increases the likelihood of potential outbreak beyond the Far East Asia. The World Health Organization (WHO) has also put SFTSV into the priority pathogen requiring urgent attention category. Currently, no vaccine has been available to prevent SFTS.
The research team led by Professor Su-Hyung Park noted that DNA vaccines induce broader immunity to multiple antigens than traditional ones. Moreover, DNA vaccines stimulate both T cell and antibody immunity, which make them suitable for vaccine development.
They constructed DNA vaccines that encode full-length Gn, Gc, N, NS, and RNA polymerase genes based on common sequences of 31 SFTSV strains isolated from patients. Their vaccine candidates induced both neutralizing antibody response and multifunctional SFTSV-specific T cell response in mice and ferrets.
To investigate the vaccine’s efficacy in vivo, the research team applied a recently developed ferret model that recapitulates fatal clinical symptoms in SFTSV infection in humans. Vaccinated ferrets were completely protected from lethal SFTSV challenge without SFTSV detection in their blood, whereas all control ferrets died within 10 days’ post-infection.
The KAIST team found that anti-envelope antibodies play an important role in protective immunity, suggesting that envelope glycoproteins of SFTSV may be the most effective antigens for inducing protective immunity. Moreover, the study revealed that T cell responses specific to non-envelope proteins of SFTSV also can contribute to protection against SFTSV infection.
Professor Park said, “This is the first study demonstrating complete protection against lethal SFTSV challenge using an immunocompetent, middle-sized animal model with clinical manifestations of SFTSV infection. We believe this study provides valuable insights into designing preventive vaccines for SFTSV.”
2020.01.31 View 3471
KAIST Vaccine for Tick-Borne Disease ‘SFTS’ Protects Against Lethal Infection
A KAIST research team reported the development of a DNA vaccine for Severe Fever with Thrombocytopenia Syndrome Virus (SFTSV) which completely protects against lethal infection in ferrets. The team confirmed that ferrets immunized with DNA vaccines encoding all SFTSV proteins showed 100% survival rate without detectable viremia and did not develop any clinical symptoms. This study was published in Nature Communications on August 23.
Severe Fever with Thrombocytopenia Syndrome (SFTS) is a newly emerging tick-borne infectious disease. The disease causes fever, severe thrombocytopenia, leukocytopenia as well as vomiting and diarrhea. Severe cases end up with organ system failure often accompanied by hemorrhages, and its mortality rate stands at 10–20%.
The viral disease has been endemic to East Asia but the spread of the tick vector to North America increases the likelihood of potential outbreak beyond the Far East Asia. The World Health Organization (WHO) has also put SFTSV into the priority pathogen requiring urgent attention category. Currently, no vaccine has been available to prevent SFTS.
The research team led by Professor Su-Hyung Park noted that DNA vaccines induce broader immunity to multiple antigens than traditional ones. Moreover, DNA vaccines stimulate both T cell and antibody immunity, which make them suitable for vaccine development.
They constructed DNA vaccines that encode full-length Gn, Gc, N, NS, and RNA polymerase genes based on common sequences of 31 SFTSV strains isolated from patients. Their vaccine candidates induced both neutralizing antibody response and multifunctional SFTSV-specific T cell response in mice and ferrets.
To investigate the vaccine’s efficacy in vivo, the research team applied a recently developed ferret model that recapitulates fatal clinical symptoms in SFTSV infection in humans. Vaccinated ferrets were completely protected from lethal SFTSV challenge without SFTSV detection in their blood, whereas all control ferrets died within 10 days’ post-infection.
The KAIST team found that anti-envelope antibodies play an important role in protective immunity, suggesting that envelope glycoproteins of SFTSV may be the most effective antigens for inducing protective immunity. Moreover, the study revealed that T cell responses specific to non-envelope proteins of SFTSV also can contribute to protection against SFTSV infection.
Professor Park said, “This is the first study demonstrating complete protection against lethal SFTSV challenge using an immunocompetent, middle-sized animal model with clinical manifestations of SFTSV infection. We believe this study provides valuable insights into designing preventive vaccines for SFTSV.”
2020.01.31 View 3471 -
 Scientists Discover the Mechanism of DNA High-Order Structure Formation
(Molecular structures of Abo1 in different energy states (left), Demonstration of an Abo1-assisted histone loading onto DNA by the DNA curtain assay. )
The genetic material of our cells—DNA—exists in a high-order structure called “chromatin”. Chromatin consists of DNA wrapped around histone proteins and efficiently packs DNA into a small volume. Moreover, using a spool and thread analogy, chromatin allows DNA to be locally wound or unwound, thus enabling genes to be enclosed or exposed. The misregulation of chromatin structures results in aberrant gene expression and can ultimately lead to developmental disorders or cancers. Despite the importance of DNA high-order structures, the complexity of the underlying machinery has circumvented molecular dissection.
For the first time, molecular biologists have uncovered how one particular mechanism uses energy to ensure proper histone placement onto DNA to form chromatin. They published their results on Dec. 17 in Nature Communications.
The study focused on proteins called histone chaperones. Histone chaperones are responsible for adding and removing specific histones at specific times during the DNA packaging process. The wrong histone at the wrong time and place could result in the misregulation of gene expression or aberrant DNA replication. Thus, histone chaperones are key players in the assembly and disassembly of chromatin.
“In order to carefully control the assembly and disassembly of chromatin units, histone chaperones act as molecular escorts that prevent histone aggregation and undesired interactions,” said Professor Ji-Joon Song in the Department of Biological Sciences at KAIST. “We set out to understand how a unique histone chaperone uses chemical energy to assemble or disassemble chromatin.”
Song and his team looked to Abo1, the only known histone chaperone that utilizes cellular energy (ATP). While Abo1 is found in yeast, it has an analogous partner in other organisms, including humans, called ATAD2. Both use ATP, which is produced through a cellular process where enzymes break down a molecule’s phosphate bond. ATP energy is typically used to power other cellular processes, but it is a rare partner for histone chaperones.
“This was an interesting problem in the field because all other histone chaperones studied to date do not use ATP,” Song said.
By imaging Abo1 with a single-molecule fluorescence imaging technique known as the DNA curtain assay, the researchers could examine the protein interactions at the single-molecule level. The technique allows scientists to arrange the DNA molecules and proteins on a single layer of a microfluidic chamber and examine the layer with fluorescence microscopy.
The researchers found through real-time observation that Abo1 is ring-shaped and changes its structure to accommodate a specific histone and deposit it on DNA. Moreover, they found that the accommodating structural changes are powered by ADP.
“We discovered a mechanism by which Abo1 accommodates histone substrates, ultimately allowing it to function as a unique energy-dependent histone chaperone,” Song said. “We also found that despite looking like a protein disassembly machine, Abo1 actually loads histone substrates onto DNA to facilitate chromatin assembly.”
The researchers plan to continue exploring how energy-dependent histone chaperones bind and release histones, with the ultimate goal of developing therapeutics that can target cancer-causing misbehavior by Abo1’s analogous human counterpart, ATAD2.
-Profile
Professor Ji-Joon Song
Department of Biological Sciences KI for the BioCentury (https://kis.kaist.ac.kr/index.php?mid=KIB_O) KAIST
2020.01.07 View 6731
Scientists Discover the Mechanism of DNA High-Order Structure Formation
(Molecular structures of Abo1 in different energy states (left), Demonstration of an Abo1-assisted histone loading onto DNA by the DNA curtain assay. )
The genetic material of our cells—DNA—exists in a high-order structure called “chromatin”. Chromatin consists of DNA wrapped around histone proteins and efficiently packs DNA into a small volume. Moreover, using a spool and thread analogy, chromatin allows DNA to be locally wound or unwound, thus enabling genes to be enclosed or exposed. The misregulation of chromatin structures results in aberrant gene expression and can ultimately lead to developmental disorders or cancers. Despite the importance of DNA high-order structures, the complexity of the underlying machinery has circumvented molecular dissection.
For the first time, molecular biologists have uncovered how one particular mechanism uses energy to ensure proper histone placement onto DNA to form chromatin. They published their results on Dec. 17 in Nature Communications.
The study focused on proteins called histone chaperones. Histone chaperones are responsible for adding and removing specific histones at specific times during the DNA packaging process. The wrong histone at the wrong time and place could result in the misregulation of gene expression or aberrant DNA replication. Thus, histone chaperones are key players in the assembly and disassembly of chromatin.
“In order to carefully control the assembly and disassembly of chromatin units, histone chaperones act as molecular escorts that prevent histone aggregation and undesired interactions,” said Professor Ji-Joon Song in the Department of Biological Sciences at KAIST. “We set out to understand how a unique histone chaperone uses chemical energy to assemble or disassemble chromatin.”
Song and his team looked to Abo1, the only known histone chaperone that utilizes cellular energy (ATP). While Abo1 is found in yeast, it has an analogous partner in other organisms, including humans, called ATAD2. Both use ATP, which is produced through a cellular process where enzymes break down a molecule’s phosphate bond. ATP energy is typically used to power other cellular processes, but it is a rare partner for histone chaperones.
“This was an interesting problem in the field because all other histone chaperones studied to date do not use ATP,” Song said.
By imaging Abo1 with a single-molecule fluorescence imaging technique known as the DNA curtain assay, the researchers could examine the protein interactions at the single-molecule level. The technique allows scientists to arrange the DNA molecules and proteins on a single layer of a microfluidic chamber and examine the layer with fluorescence microscopy.
The researchers found through real-time observation that Abo1 is ring-shaped and changes its structure to accommodate a specific histone and deposit it on DNA. Moreover, they found that the accommodating structural changes are powered by ADP.
“We discovered a mechanism by which Abo1 accommodates histone substrates, ultimately allowing it to function as a unique energy-dependent histone chaperone,” Song said. “We also found that despite looking like a protein disassembly machine, Abo1 actually loads histone substrates onto DNA to facilitate chromatin assembly.”
The researchers plan to continue exploring how energy-dependent histone chaperones bind and release histones, with the ultimate goal of developing therapeutics that can target cancer-causing misbehavior by Abo1’s analogous human counterpart, ATAD2.
-Profile
Professor Ji-Joon Song
Department of Biological Sciences KI for the BioCentury (https://kis.kaist.ac.kr/index.php?mid=KIB_O) KAIST
2020.01.07 View 6731 -
 Professor Shin-Hyun Kim Receives the Young Scientist Award
Professor Shin-Hyun Kim from the Department of Chemical and Biomolecular Engineering received the Young Scientist Award from the Korean Academy of Science and Technology.
The Young Scientist Award is presented to a promising young Korean scientist under the age of 40 who shows significant potential, passion, and remarkable achievement.
Professor Kim was lauded for his research of intelligent soft materials. By applying his research, he developed a capsule sensor material that can not only be used for sensors, but also for displays, color aesthetics, anti-counterfeit technology, residual drug detection, and more.
The award ceremony took place on December 14 at the Gwacheon National Science Museum.
The Korean minister of Science and ICT delivered words of encouragement, reminding everyone that “the driving force behind creative performance of scientists is the provision of continuous support.” He added, “Researchers of Korea deserve greater public attention and support.”
(END)
2019.12.21 View 4940
Professor Shin-Hyun Kim Receives the Young Scientist Award
Professor Shin-Hyun Kim from the Department of Chemical and Biomolecular Engineering received the Young Scientist Award from the Korean Academy of Science and Technology.
The Young Scientist Award is presented to a promising young Korean scientist under the age of 40 who shows significant potential, passion, and remarkable achievement.
Professor Kim was lauded for his research of intelligent soft materials. By applying his research, he developed a capsule sensor material that can not only be used for sensors, but also for displays, color aesthetics, anti-counterfeit technology, residual drug detection, and more.
The award ceremony took place on December 14 at the Gwacheon National Science Museum.
The Korean minister of Science and ICT delivered words of encouragement, reminding everyone that “the driving force behind creative performance of scientists is the provision of continuous support.” He added, “Researchers of Korea deserve greater public attention and support.”
(END)
2019.12.21 View 4940 -
 New IEEE Fellow, Professor Jong Chul Ye
Professor Jong Chul Ye from the Department of Bio and Brain Engineering was named a new fellow of the Institute of Electrical and Electronics Engineers (IEEE). IEEE announced this on December 1 in recognition of Professor Ye’s contributions to the development of signal processing and artificial intelligence (AI) technology in the field of biomedical imaging.
As the world’s largest society in the electrical and electronics field, IEEE names the top 0.1% of their members as fellows based on their research achievements.Professor Ye has published more than 100 research papers in world-leading journals in the biomedical imaging field, including those affiliated with IEEE.
He also gave a keynote talk at the yearly conference of the International Society for Magnetic Resonance Imaging (ISMRM) on medical AI technology. In addition, Professor Ye has been appointed to serve as the next chair of the Computational Imaging Technical Committee of the IEEE Signal Processing Society, and the chair of the IEEE Symposium on Biomedical Imaging (ISBI) 2020 to be held in April in Iowa, USA.
Professor Ye said, “The importance of AI technology is developing in the biomedical imaging field. I feel proud that my contributions have been internationally recognized and allowed me to be named an IEEE fellow.”
2019.12.18 View 5882
New IEEE Fellow, Professor Jong Chul Ye
Professor Jong Chul Ye from the Department of Bio and Brain Engineering was named a new fellow of the Institute of Electrical and Electronics Engineers (IEEE). IEEE announced this on December 1 in recognition of Professor Ye’s contributions to the development of signal processing and artificial intelligence (AI) technology in the field of biomedical imaging.
As the world’s largest society in the electrical and electronics field, IEEE names the top 0.1% of their members as fellows based on their research achievements.Professor Ye has published more than 100 research papers in world-leading journals in the biomedical imaging field, including those affiliated with IEEE.
He also gave a keynote talk at the yearly conference of the International Society for Magnetic Resonance Imaging (ISMRM) on medical AI technology. In addition, Professor Ye has been appointed to serve as the next chair of the Computational Imaging Technical Committee of the IEEE Signal Processing Society, and the chair of the IEEE Symposium on Biomedical Imaging (ISBI) 2020 to be held in April in Iowa, USA.
Professor Ye said, “The importance of AI technology is developing in the biomedical imaging field. I feel proud that my contributions have been internationally recognized and allowed me to be named an IEEE fellow.”
2019.12.18 View 5882 -
 Tungsten Suboxide Improves the Efficiency of Platinum in Hydrogen Production
< PhD Candidate Jinkyu Park and Professor Jinwoo Lee >
Researchers presented a new strategy for enhancing catalytic activity using tungsten suboxide as a single-atom catalyst (SAC). This strategy, which significantly improves hydrogen evolution reaction (HER) in metal platinum (pt) by 16.3 times, sheds light on the development of new electrochemical catalyst technologies.
Hydrogen has been touted as a promising alternative to fossil fuels. However, most of the conventional industrial hydrogen production methods come with environmental issues, releasing significant amounts of carbon dioxide and greenhouse gases.
Electrochemical water splitting is considered a potential approach for clean hydrogen production. Pt is one of the most commonly used catalysts to improve HER performance in electrochemical water splitting, but the high cost and scarcity of Pt remain key obstacles to mass commercial applications.
SACs, where all metal species are individually dispersed on a desired support material, have been identified as one way to reduce the amount of Pt usage, as they offer the maximum number of surface exposed Pt atoms.
Inspired by earlier studies, which mainly focused on SACs supported by carbon-based materials, a KAIST research team led by Professor Jinwoo Lee from the Department of Chemical and Biomolecular Engineering investigated the influence of support materials on the performance of SACs.
Professor Lee and his researchers suggested mesoporous tungsten suboxide as a new support material for atomically dispersed Pt, as this was expected to provide high electronic conductivity and have a synergetic effect with Pt.
They compared the performance of single-atom Pt supported by carbon and tungsten suboxide respectively. The results revealed that the support effect occurred with tungsten suboxide, in which the mass activity of a single-atom Pt supported by tungsten suboxide was 2.1 times greater than that of single-atom Pt supported by carbon, and 16.3 times higher than that of Pt nanoparticles supported by carbon.
The team indicated a change in the electronic structure of Pt via charge transfer from tungsten suboxide to Pt. This phenomenon was reported as a result of strong metal-support interaction between Pt and tungsten suboxide.
HER performance can be improved not only by changing the electronic structure of the supported metal, but also by inducing another support effect, the spillover effect, the research group reported. Hydrogen spillover is a phenomenon where adsorbed hydrogen migrates from one surface to another, and it occurs more easily as the Pt size becomes smaller.
The researchers compared the performance of single-atom Pt and Pt nanoparticles supported by tungsten suboxide. The single-atom Pt supported by tungsten suboxide exhibited a higher degree of hydrogen spillover phenomenon, which enhanced the Pt mass activity for hydrogen evolution up to 10.7 times compared to Pt nanoparticles supported by tungsten suboxide.
Professor Lee said, “Choosing the right support material is important for improving electrocatalysis in hydrogen production. The tungsten suboxide catalyst we used to support Pt in our study implies that interactions between the well-matched metal and support can drastically enhance the efficiency of the process.”
This research was supported by the Ministry of Science and ICT and introduced in the International Edition of the German journal Angewandte Chemie.
Figure. Schematic representation of hydrogen evolution reaction (HER) of pseudo single-atom Pt supported by tungsten suboxide
-Publication
Jinkyu Park, Dr. Seonggyu Lee, Hee-Eun Kim, Ara Cho, Seongbeen Kim, Dr. Youngjin Ye, Prof. Jeong Woo Han, Prof. Hyunjoo Lee, Dr. Jong Hyun Jang, and Prof. Jinwoo Lee. 2019. Investigation of the Support Effect in Atomically Dispersed Pt on WO3−x for Utilization of Pt in the Hydrogen Evolution Reaction. International Edition of Angewandte Chemie. Volume No. 58. Issue No. 45. 6 pages. https://doi.org/10.1002/anie.201908122
-ProfileProfessor Jinwoo LeeConvergence of Energy and Nano Science Laboratoryhttp://cens.kaist.ac.kr
Department of Chemical and Biomolecular EngineeringKAIST
2019.10.28 View 16020
Tungsten Suboxide Improves the Efficiency of Platinum in Hydrogen Production
< PhD Candidate Jinkyu Park and Professor Jinwoo Lee >
Researchers presented a new strategy for enhancing catalytic activity using tungsten suboxide as a single-atom catalyst (SAC). This strategy, which significantly improves hydrogen evolution reaction (HER) in metal platinum (pt) by 16.3 times, sheds light on the development of new electrochemical catalyst technologies.
Hydrogen has been touted as a promising alternative to fossil fuels. However, most of the conventional industrial hydrogen production methods come with environmental issues, releasing significant amounts of carbon dioxide and greenhouse gases.
Electrochemical water splitting is considered a potential approach for clean hydrogen production. Pt is one of the most commonly used catalysts to improve HER performance in electrochemical water splitting, but the high cost and scarcity of Pt remain key obstacles to mass commercial applications.
SACs, where all metal species are individually dispersed on a desired support material, have been identified as one way to reduce the amount of Pt usage, as they offer the maximum number of surface exposed Pt atoms.
Inspired by earlier studies, which mainly focused on SACs supported by carbon-based materials, a KAIST research team led by Professor Jinwoo Lee from the Department of Chemical and Biomolecular Engineering investigated the influence of support materials on the performance of SACs.
Professor Lee and his researchers suggested mesoporous tungsten suboxide as a new support material for atomically dispersed Pt, as this was expected to provide high electronic conductivity and have a synergetic effect with Pt.
They compared the performance of single-atom Pt supported by carbon and tungsten suboxide respectively. The results revealed that the support effect occurred with tungsten suboxide, in which the mass activity of a single-atom Pt supported by tungsten suboxide was 2.1 times greater than that of single-atom Pt supported by carbon, and 16.3 times higher than that of Pt nanoparticles supported by carbon.
The team indicated a change in the electronic structure of Pt via charge transfer from tungsten suboxide to Pt. This phenomenon was reported as a result of strong metal-support interaction between Pt and tungsten suboxide.
HER performance can be improved not only by changing the electronic structure of the supported metal, but also by inducing another support effect, the spillover effect, the research group reported. Hydrogen spillover is a phenomenon where adsorbed hydrogen migrates from one surface to another, and it occurs more easily as the Pt size becomes smaller.
The researchers compared the performance of single-atom Pt and Pt nanoparticles supported by tungsten suboxide. The single-atom Pt supported by tungsten suboxide exhibited a higher degree of hydrogen spillover phenomenon, which enhanced the Pt mass activity for hydrogen evolution up to 10.7 times compared to Pt nanoparticles supported by tungsten suboxide.
Professor Lee said, “Choosing the right support material is important for improving electrocatalysis in hydrogen production. The tungsten suboxide catalyst we used to support Pt in our study implies that interactions between the well-matched metal and support can drastically enhance the efficiency of the process.”
This research was supported by the Ministry of Science and ICT and introduced in the International Edition of the German journal Angewandte Chemie.
Figure. Schematic representation of hydrogen evolution reaction (HER) of pseudo single-atom Pt supported by tungsten suboxide
-Publication
Jinkyu Park, Dr. Seonggyu Lee, Hee-Eun Kim, Ara Cho, Seongbeen Kim, Dr. Youngjin Ye, Prof. Jeong Woo Han, Prof. Hyunjoo Lee, Dr. Jong Hyun Jang, and Prof. Jinwoo Lee. 2019. Investigation of the Support Effect in Atomically Dispersed Pt on WO3−x for Utilization of Pt in the Hydrogen Evolution Reaction. International Edition of Angewandte Chemie. Volume No. 58. Issue No. 45. 6 pages. https://doi.org/10.1002/anie.201908122
-ProfileProfessor Jinwoo LeeConvergence of Energy and Nano Science Laboratoryhttp://cens.kaist.ac.kr
Department of Chemical and Biomolecular EngineeringKAIST
2019.10.28 View 16020 -
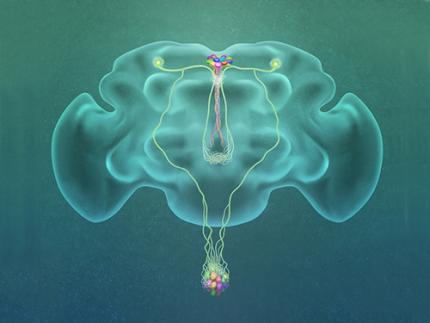 A Single, Master Switch for Sugar Levels?
When a fly eats sugar, a single brain cell sends simultaneous messages to stimulate one hormone and inhibit another to control glucose levels in the body. Further research into this control system with remarkable precision could shed light on the neural mechanisms of diabetes and obesity in humans .
A single neuron appears to monitor and control sugar levels in the fly body, according to research published this week in Nature. This new insight into the mechanisms in the fly brain that maintain a balance of two key hormones controlling glucose levels, insulin and glucagon, can provide a framework for understanding diabetes and obesity in humans.
Neurons that sense and respond to glucose were identified more than 50 years ago, but what they do in our body has remained unclear. Researchers at the Korea Advanced Institute of Science and Technology (KAIST) and New York University School of Medicine have now found a single “glucose-sensing neuron” that appears to be the master controller in Drosophila, the vinegar fly, for maintaining an ideal glucose balance, called homeostasis.
Professor Greg Seong-Bae Suh, Dr. Yangkyun Oh and colleagues identified a key neuron that is excited by glucose, which they called CN neuron. This CN neuron has a unique shape – it has an axon (which is used to transmit information to downstream cells) that is bifurcated. One branch projects to insulin-producing cells, and sends a signal triggering the secretion of the insulin equivalent in flies. The other branch projects to glucagon-producing cells and sends a signal inhibiting the secretion of the glucagon equivalent.
When flies consume food, the levels of glucose in their body increase; this excites the CN neuron, which fires the simultaneous signals to stimulate insulin and inhibit glucagon secretion, thereby maintaining the appropriate balance between the hormones and sugar in the blood. The researchers were able to see this happening in the brain in real time by using a combination of cutting-edge fluorescent calcium imaging technology, as well as measuring hormone and sugar levels and applying highly sophisticated molecular genetic techniques.
When flies were not fed, however, the researchers observed a reduction in the activity of CN neuron, a reduction in insulin secretion and an increase in glucagon secretion. These findings indicate that these key hormones are under the direct control of the glucose-sensing neuron. Furthermore, when they silenced the CN neuron rendering dysfunctional CN neuron in flies, these animals experienced an imbalance, resulting in hyperglycemia – high levels of sugars in the blood, similar to what is observed in diabetes in humans. This further suggests that the CN neuron is critical to maintaining glucose homeostasis in animals.
While further research is required to investigate this process in humans, Suh notes this is a significant step forward in the fields of both neurobiology and endocrinology.
“This work lays the foundation for translational research to better understand how this delicate regulatory process is affected by diabetes, obesity, excessive nutrition and diets high in sugar,” Suh said.
Profile: Greg Seong-Bae Suh
seongbaesuh@kaist.ac.kr
Professor Department of Biological Sciences
KAIST
(Figure: A single glucose-excited CN neuron extends bifurcated axonal branches,
one of which innervates insulin producing cells and stimulates their activity an the other axonal branch projects to glucagon producing cells and inhibits their activity.)
2019.10.24 View 14359
A Single, Master Switch for Sugar Levels?
When a fly eats sugar, a single brain cell sends simultaneous messages to stimulate one hormone and inhibit another to control glucose levels in the body. Further research into this control system with remarkable precision could shed light on the neural mechanisms of diabetes and obesity in humans .
A single neuron appears to monitor and control sugar levels in the fly body, according to research published this week in Nature. This new insight into the mechanisms in the fly brain that maintain a balance of two key hormones controlling glucose levels, insulin and glucagon, can provide a framework for understanding diabetes and obesity in humans.
Neurons that sense and respond to glucose were identified more than 50 years ago, but what they do in our body has remained unclear. Researchers at the Korea Advanced Institute of Science and Technology (KAIST) and New York University School of Medicine have now found a single “glucose-sensing neuron” that appears to be the master controller in Drosophila, the vinegar fly, for maintaining an ideal glucose balance, called homeostasis.
Professor Greg Seong-Bae Suh, Dr. Yangkyun Oh and colleagues identified a key neuron that is excited by glucose, which they called CN neuron. This CN neuron has a unique shape – it has an axon (which is used to transmit information to downstream cells) that is bifurcated. One branch projects to insulin-producing cells, and sends a signal triggering the secretion of the insulin equivalent in flies. The other branch projects to glucagon-producing cells and sends a signal inhibiting the secretion of the glucagon equivalent.
When flies consume food, the levels of glucose in their body increase; this excites the CN neuron, which fires the simultaneous signals to stimulate insulin and inhibit glucagon secretion, thereby maintaining the appropriate balance between the hormones and sugar in the blood. The researchers were able to see this happening in the brain in real time by using a combination of cutting-edge fluorescent calcium imaging technology, as well as measuring hormone and sugar levels and applying highly sophisticated molecular genetic techniques.
When flies were not fed, however, the researchers observed a reduction in the activity of CN neuron, a reduction in insulin secretion and an increase in glucagon secretion. These findings indicate that these key hormones are under the direct control of the glucose-sensing neuron. Furthermore, when they silenced the CN neuron rendering dysfunctional CN neuron in flies, these animals experienced an imbalance, resulting in hyperglycemia – high levels of sugars in the blood, similar to what is observed in diabetes in humans. This further suggests that the CN neuron is critical to maintaining glucose homeostasis in animals.
While further research is required to investigate this process in humans, Suh notes this is a significant step forward in the fields of both neurobiology and endocrinology.
“This work lays the foundation for translational research to better understand how this delicate regulatory process is affected by diabetes, obesity, excessive nutrition and diets high in sugar,” Suh said.
Profile: Greg Seong-Bae Suh
seongbaesuh@kaist.ac.kr
Professor Department of Biological Sciences
KAIST
(Figure: A single glucose-excited CN neuron extends bifurcated axonal branches,
one of which innervates insulin producing cells and stimulates their activity an the other axonal branch projects to glucagon producing cells and inhibits their activity.)
2019.10.24 View 14359 -
 Professor Hyun Gyu Park Appointed as Associate Editor for Biosensors and Bioelectronics
Professor Hyun Gyu Park from the Department of Chemical and Biomolecular Engineering was appointed as an associate editor for Biosensors and Bioelectronics, an international journal published by Elsevier.
Biosensors and Bioelectronics is one of the top SCI journals in the fields of chemistry and analytical science (IF 9.518 as of 2018). Professor Park was recognized and appointed as the associate editor for this journal due to his outstanding research achievements in the fields of nucleic acid engineering, biosensors, and nanobiotechnology.
Professor Park will serve as the associate editor from this October until December 2021.
(END)
2019.10.01 View 4532
Professor Hyun Gyu Park Appointed as Associate Editor for Biosensors and Bioelectronics
Professor Hyun Gyu Park from the Department of Chemical and Biomolecular Engineering was appointed as an associate editor for Biosensors and Bioelectronics, an international journal published by Elsevier.
Biosensors and Bioelectronics is one of the top SCI journals in the fields of chemistry and analytical science (IF 9.518 as of 2018). Professor Park was recognized and appointed as the associate editor for this journal due to his outstanding research achievements in the fields of nucleic acid engineering, biosensors, and nanobiotechnology.
Professor Park will serve as the associate editor from this October until December 2021.
(END)
2019.10.01 View 4532 -
 Professor Ki-Jun Yoon selected as the 2019 SUHF Young Investigator
< Professor Ki-Jun Yoon >
Professor Ki-Jun Yoon from the Department of Biological Sciences was named one of four recipients of the 2019 Suh Kyung-Bae Science Foundation (SUHF) Young Investigator Awards.
The SUHF is a non-profit organization established in 2016 and funded by a personal donation of 300 billion KRW in shares from Chairman and CEO Kyung-Bae Suh of the Amorepacific Group. The primary purpose of the foundation is to serve as a platform to nurture and provide comprehensive long-term support for creative and passionate young Korean scientists committed to pursuing research in the field of life sciences. The SUHF selects three to five scientists through an open recruiting process every year, and grants each scientist a maximum of 2.5 billion KRW over a period of up to five years.
Since January this year, the foundation received 83 research proposals from scientists across the nation, especially from those who had less than five years of experience as professors, and selected the four recipients, including Professor Yoon.
Professor Yoon was recognized for his contributions to the advancement of research on how post-transcriptional mechanisms may modulate stem cell properties. His research project involves deciphering the molecular mechanisms controlling RNA metabolism in neural stem cells during normal development, and how alterations in RNA regulatory programs lead to human brain disorders.
< (From left) Professor Joo-Hong Park, Professor Yuree Lee, Chairman and CEO Kyung-Bae Suh, Professor Eunjung Lee, Professor Ki-Jun Yoon, ⓒ Amorepacific Group >
The other awards were given to Professor Joo-Hong Park and Professor Yuree Lee of Seoul National University, and Professor Eunjung Lee of Boston Children's Hospital and Harvard Medical School.
The awards ceremony was held on September 18 at the Amorepacific Headquarters in Seoul.
With these four new awardees, a total of 14 scientists have been named as SUHF Young Investigators to date.
(END)
2019.09.23 View 6015
Professor Ki-Jun Yoon selected as the 2019 SUHF Young Investigator
< Professor Ki-Jun Yoon >
Professor Ki-Jun Yoon from the Department of Biological Sciences was named one of four recipients of the 2019 Suh Kyung-Bae Science Foundation (SUHF) Young Investigator Awards.
The SUHF is a non-profit organization established in 2016 and funded by a personal donation of 300 billion KRW in shares from Chairman and CEO Kyung-Bae Suh of the Amorepacific Group. The primary purpose of the foundation is to serve as a platform to nurture and provide comprehensive long-term support for creative and passionate young Korean scientists committed to pursuing research in the field of life sciences. The SUHF selects three to five scientists through an open recruiting process every year, and grants each scientist a maximum of 2.5 billion KRW over a period of up to five years.
Since January this year, the foundation received 83 research proposals from scientists across the nation, especially from those who had less than five years of experience as professors, and selected the four recipients, including Professor Yoon.
Professor Yoon was recognized for his contributions to the advancement of research on how post-transcriptional mechanisms may modulate stem cell properties. His research project involves deciphering the molecular mechanisms controlling RNA metabolism in neural stem cells during normal development, and how alterations in RNA regulatory programs lead to human brain disorders.
< (From left) Professor Joo-Hong Park, Professor Yuree Lee, Chairman and CEO Kyung-Bae Suh, Professor Eunjung Lee, Professor Ki-Jun Yoon, ⓒ Amorepacific Group >
The other awards were given to Professor Joo-Hong Park and Professor Yuree Lee of Seoul National University, and Professor Eunjung Lee of Boston Children's Hospital and Harvard Medical School.
The awards ceremony was held on September 18 at the Amorepacific Headquarters in Seoul.
With these four new awardees, a total of 14 scientists have been named as SUHF Young Investigators to date.
(END)
2019.09.23 View 6015 -
 Two More Cross-generation Collaborative Labs Open
< President Sung-Chul Shin (sixth from the left) and Professor Sun Chang Kim (seventh from the left) at the signboard ceremony of KAIST BioDesigneering Laboratory >
KAIST opened two more cross-generation collaborative labs last month. KAIST BioDesigneering Laboratory headed by Professor Sun Chang Kim from the Department of Biological Sciences and Nanophotonics Laboratory led by Professor Yong-Hee Lee from the Department of Physics have been selected to receive 500 million KRW funding for five years.
A four-member selection committee including the former President of ETH Zürich Professor Emeritus Ralph Eichler and Professor Kwang-Soo Kim of Harvard Medical School conducted a three-month review and evaluation for this selection to be made. With these two new labs onboard, a total of six cross-generation collaborative labs will be operated on campus.
The operation of cross-generation collaborative labs has been in trial since March last year, as one of the KAIST’s Vision 2031 research innovation initiatives. This novel approach is to pair up senior and junior faculty members for sustaining research and academic achievements even after the senior researcher retires, so that the spectrum of knowledge and research competitiveness can be extended to future generations. The selected labs will be funded for five years, and the funding will be extended if necessary. KAIST will continue to select new labs every year.
One of this year’s selectees Professor Sun Chang Kim will be teamed up with Professor Byung-Kwan Cho from the same department and Professor Jung Kyoon Choi from the Department of Bio and Brain Engineering to collaborate in the fields of synthetic biology, systems biology, and genetic engineering. This group mainly aims at designing and synthesizing optimal genomes that can efficiently manufacture protein drug and biomedical active materials. They will also strive to secure large amounts of high-functioning natural active substances, new adhesive antibacterial peptides, and eco-friendly ecological restoration materials. It is expected that collaboration between these three multigenerational professors will help innovate their bio-convergence technology and further strengthen their international competitiveness in the global bio-market.
Another world-renowned scholar Professor Yong-Hee Lee of photonic crystal laser study will be joined by Professor Minkyo Seo from the same department and Professor Hansuek Lee from the Graduate School of Nanoscience and Technology. They will explore the extreme limits of light-material interaction based on optical micro/nano resonators, with the goal of developing future nonlinear optoelectronic and quantum optical devices. The knowledge and technology newly gained from the research are expected to provide an important platform for a diverse range of fields from quantum communications to biophysics.
(END)
2019.09.06 View 6649
Two More Cross-generation Collaborative Labs Open
< President Sung-Chul Shin (sixth from the left) and Professor Sun Chang Kim (seventh from the left) at the signboard ceremony of KAIST BioDesigneering Laboratory >
KAIST opened two more cross-generation collaborative labs last month. KAIST BioDesigneering Laboratory headed by Professor Sun Chang Kim from the Department of Biological Sciences and Nanophotonics Laboratory led by Professor Yong-Hee Lee from the Department of Physics have been selected to receive 500 million KRW funding for five years.
A four-member selection committee including the former President of ETH Zürich Professor Emeritus Ralph Eichler and Professor Kwang-Soo Kim of Harvard Medical School conducted a three-month review and evaluation for this selection to be made. With these two new labs onboard, a total of six cross-generation collaborative labs will be operated on campus.
The operation of cross-generation collaborative labs has been in trial since March last year, as one of the KAIST’s Vision 2031 research innovation initiatives. This novel approach is to pair up senior and junior faculty members for sustaining research and academic achievements even after the senior researcher retires, so that the spectrum of knowledge and research competitiveness can be extended to future generations. The selected labs will be funded for five years, and the funding will be extended if necessary. KAIST will continue to select new labs every year.
One of this year’s selectees Professor Sun Chang Kim will be teamed up with Professor Byung-Kwan Cho from the same department and Professor Jung Kyoon Choi from the Department of Bio and Brain Engineering to collaborate in the fields of synthetic biology, systems biology, and genetic engineering. This group mainly aims at designing and synthesizing optimal genomes that can efficiently manufacture protein drug and biomedical active materials. They will also strive to secure large amounts of high-functioning natural active substances, new adhesive antibacterial peptides, and eco-friendly ecological restoration materials. It is expected that collaboration between these three multigenerational professors will help innovate their bio-convergence technology and further strengthen their international competitiveness in the global bio-market.
Another world-renowned scholar Professor Yong-Hee Lee of photonic crystal laser study will be joined by Professor Minkyo Seo from the same department and Professor Hansuek Lee from the Graduate School of Nanoscience and Technology. They will explore the extreme limits of light-material interaction based on optical micro/nano resonators, with the goal of developing future nonlinear optoelectronic and quantum optical devices. The knowledge and technology newly gained from the research are expected to provide an important platform for a diverse range of fields from quantum communications to biophysics.
(END)
2019.09.06 View 6649 -
 Professor Sang Gyu Kim Receives Yeochon Award for Ecology
Professor Sang-Gyu Kim from the Department of Biological Sciences was selected as the winner of the 12th Yeochon Award for Ecology presented by the Yeochon Association for Ecological Research.
The award was conferred on August 13 in Jeju at the annual conference co-hosted by the Ecological Society of Korea and the Yeochon Association for Ecological Research. Professor Kim received 10 million KRW in prize money.
Professor Kim was recognized for his achievements and contributions in studying herbivorous insects ‘rice weevils’ and their host plant ‘wild tobacco’, especially for having explored the known facts in traditional ecology at the molecular level. His findings are presented in his paper titled ‘Trichobaris weevils distinguish amongst toxic host plants by sensing volatiles that do not affect larval performance’ published in Molecular Ecology in July 2016.
Furthermore, Professor Kim’s research team is continuing their work to identify the ecological functions of plant metabolites as well as interactions between flowers and insect vectors at the molecular level. In doing so, the team edits genes in various plant species using the latest gene editing technology.
The Yeochon Award for Ecology was first established in 2005 with funds donated by a senior ecologist, the late Honorary Professor Joon-Ho Kim of Seoul National University. The award is named after the professor’s pen name “Yeochon” and is intended to encourage promising next-generation ecologists to produce outstanding research achievements in the field of basic ecology.
Professor Kim said, “I will take this award as encouragement to continue taking challenging risks to observe ecological phenomenon from a new perspective. I will continue my research with my students with joy and enthusiasm.”
2019.08.14 View 3867
Professor Sang Gyu Kim Receives Yeochon Award for Ecology
Professor Sang-Gyu Kim from the Department of Biological Sciences was selected as the winner of the 12th Yeochon Award for Ecology presented by the Yeochon Association for Ecological Research.
The award was conferred on August 13 in Jeju at the annual conference co-hosted by the Ecological Society of Korea and the Yeochon Association for Ecological Research. Professor Kim received 10 million KRW in prize money.
Professor Kim was recognized for his achievements and contributions in studying herbivorous insects ‘rice weevils’ and their host plant ‘wild tobacco’, especially for having explored the known facts in traditional ecology at the molecular level. His findings are presented in his paper titled ‘Trichobaris weevils distinguish amongst toxic host plants by sensing volatiles that do not affect larval performance’ published in Molecular Ecology in July 2016.
Furthermore, Professor Kim’s research team is continuing their work to identify the ecological functions of plant metabolites as well as interactions between flowers and insect vectors at the molecular level. In doing so, the team edits genes in various plant species using the latest gene editing technology.
The Yeochon Award for Ecology was first established in 2005 with funds donated by a senior ecologist, the late Honorary Professor Joon-Ho Kim of Seoul National University. The award is named after the professor’s pen name “Yeochon” and is intended to encourage promising next-generation ecologists to produce outstanding research achievements in the field of basic ecology.
Professor Kim said, “I will take this award as encouragement to continue taking challenging risks to observe ecological phenomenon from a new perspective. I will continue my research with my students with joy and enthusiasm.”
2019.08.14 View 3867 -
 Distinguished Professor Sang Yup Lee Honored with the 23rd NAEK Award
(Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering)
Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering was honored to be the laureate of the 23rd NAEK Award.
The NAEK (National Academy of Engineering of Korea) Award was instituted in 1997 to honor and recognize engineers who have made significant contributions to the development of the engineering and technology field at universities, industries, and institutions. Every year, it is conferred to only one person who has achieved original and world-leading research that has led to national development.
Distinguished Professor Lee is a pioneering scholar of the field of systems metabolic engineering and he was recognized for his significant achievements in the biochemical industry by developing novel microbial bioprocesses. In particular, he is globally renowned for biological plastic synthesis, making or decomposing polymers with microorganisms instead of using fossil resources.
He has produced numerous high-quality research breakthroughs in metabolic and systems engineering. In 2016, he produced an easily degradable plastic with Escherichia coli (E. coli). In 2018, he successfully produced aromatic polyesters, the main material for PET (poly ethylene terephthalate) from E. coli strains. He also identified microorganism structures for PET degradation and improved its degradability with a novel variant.
His research was ranked number one in the research and development division of Top Ten Science and Technology News 2018 announced by Korean Federation of Science & Technology Societies. He is one of highly cited researchers (HCR) ranked in the top 1% by citations for their field by the Clarivate Analytics.
2019.03.21 View 5024
Distinguished Professor Sang Yup Lee Honored with the 23rd NAEK Award
(Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering)
Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering was honored to be the laureate of the 23rd NAEK Award.
The NAEK (National Academy of Engineering of Korea) Award was instituted in 1997 to honor and recognize engineers who have made significant contributions to the development of the engineering and technology field at universities, industries, and institutions. Every year, it is conferred to only one person who has achieved original and world-leading research that has led to national development.
Distinguished Professor Lee is a pioneering scholar of the field of systems metabolic engineering and he was recognized for his significant achievements in the biochemical industry by developing novel microbial bioprocesses. In particular, he is globally renowned for biological plastic synthesis, making or decomposing polymers with microorganisms instead of using fossil resources.
He has produced numerous high-quality research breakthroughs in metabolic and systems engineering. In 2016, he produced an easily degradable plastic with Escherichia coli (E. coli). In 2018, he successfully produced aromatic polyesters, the main material for PET (poly ethylene terephthalate) from E. coli strains. He also identified microorganism structures for PET degradation and improved its degradability with a novel variant.
His research was ranked number one in the research and development division of Top Ten Science and Technology News 2018 announced by Korean Federation of Science & Technology Societies. He is one of highly cited researchers (HCR) ranked in the top 1% by citations for their field by the Clarivate Analytics.
2019.03.21 View 5024 -
 Distinguished Professor Lee Re-Appointed As Co-Chair of the Global Future Council on Biotechnology
(Distinguished Professor Sang Yup Lee)
Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering was re-appointed as the co-chair of the Global Future Council on Biotechnology at the World Economic Forum during the annual GFC meeting in Dubai, UAE last week. Elizabeth O’Day, founder and CEO of Olaris Therapeutics, will co-chair the council with him for two years.
Professor Lee served as the inaugural co-chair of the council for two years from 2016 with Professor Feng Zhang from the Broad Institute of MIT and Harvard.
The World Economic Forum’s Global Future Councils on Biotechnology is comprised of 14 members and aims to identify policy opportunities to help accelerate new biotechnological discoveries and applications while guiding dialogue with consumers about their implications.
Professor Lee, who has made significant research breakthroughs in the field of systems metabolic engineering, received the George Washington Carver Award, the PV Danckwerts Memorial Lecture Award, and the Eni Award this year.
2018.11.19 View 2534
Distinguished Professor Lee Re-Appointed As Co-Chair of the Global Future Council on Biotechnology
(Distinguished Professor Sang Yup Lee)
Distinguished Professor Sang Yup Lee from the Department of Chemical and Biomolecular Engineering was re-appointed as the co-chair of the Global Future Council on Biotechnology at the World Economic Forum during the annual GFC meeting in Dubai, UAE last week. Elizabeth O’Day, founder and CEO of Olaris Therapeutics, will co-chair the council with him for two years.
Professor Lee served as the inaugural co-chair of the council for two years from 2016 with Professor Feng Zhang from the Broad Institute of MIT and Harvard.
The World Economic Forum’s Global Future Councils on Biotechnology is comprised of 14 members and aims to identify policy opportunities to help accelerate new biotechnological discoveries and applications while guiding dialogue with consumers about their implications.
Professor Lee, who has made significant research breakthroughs in the field of systems metabolic engineering, received the George Washington Carver Award, the PV Danckwerts Memorial Lecture Award, and the Eni Award this year.
2018.11.19 View 2534